Summary information and primary citation
- PDB-id
- 1ig4; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transcription-DNA
- Method
- NMR
- Summary
- Solution structure of the methyl-cpg-binding domain of human mbd1 in complex with methylated DNA
- Reference
- Ohki I, Shimotake N, Fujita N, Jee J, Ikegami T, Nakao M, Shirakawa M (2001): "Solution structure of the methyl-CpG binding domain of human MBD1 in complex with methylated DNA." Cell(Cambridge,Mass.), 105, 487-497. doi: 10.1016/S0092-8674(01)00324-5.
- Abstract
- In vertebrates, the biological consequences of DNA methylation are often mediated by protein factors containing conserved methyl-CpG binding domains (MBDs). Mutations in the MBD protein MeCP2 cause the neurodevelopmental disease Rett syndrome. We report here the solution structure of the MBD of the human methylation-dependent transcriptional regulator MBD1 bound to methylated DNA. DNA binding causes a loop in MBD1 to fold into a major and novel DNA binding interface. Recognition of the methyl groups and CG sequence at the methylation site is due to five highly conserved residues that form a hydrophobic patch. The structure indicates how MBD may access nucleosomal DNA without encountering steric interference from core histones, and provides a basis to interpret mutations linked to Rett syndrome in MeCP2.
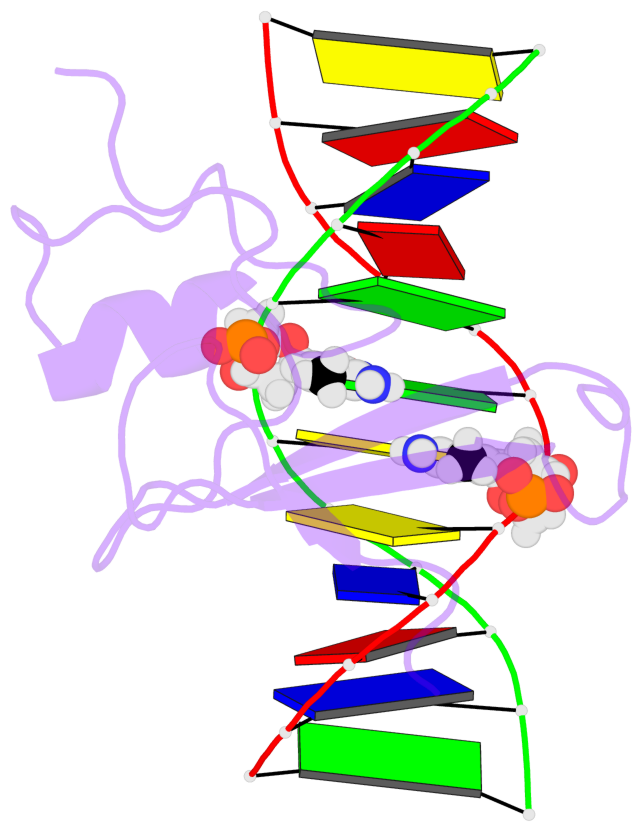
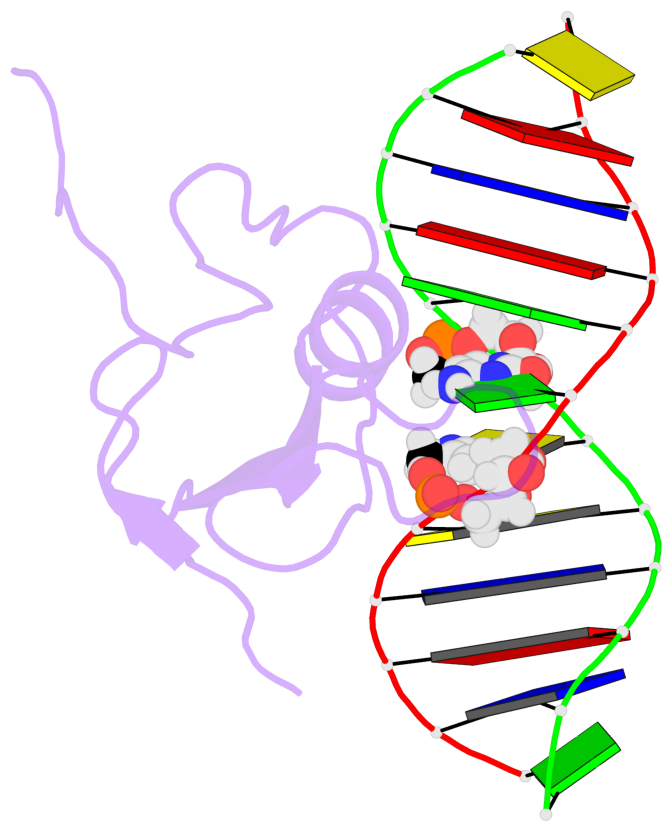
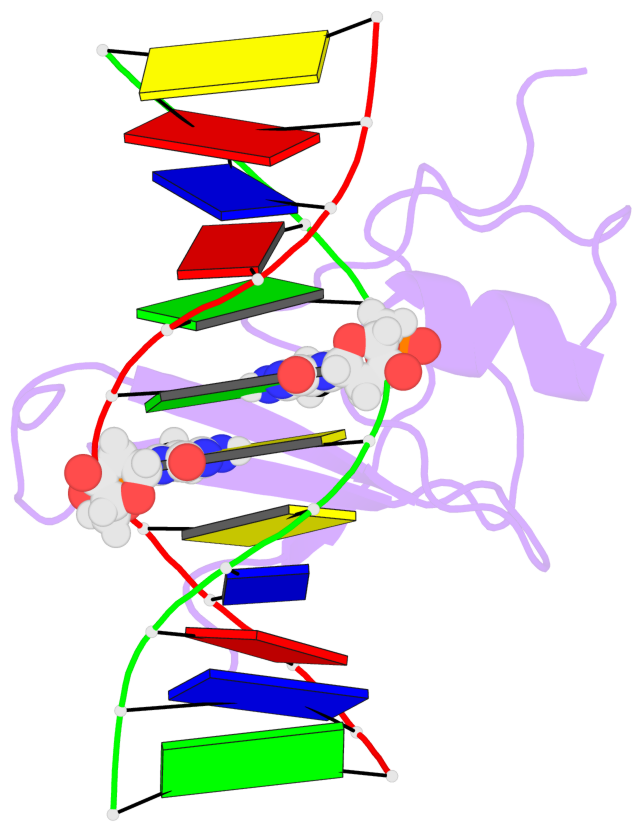
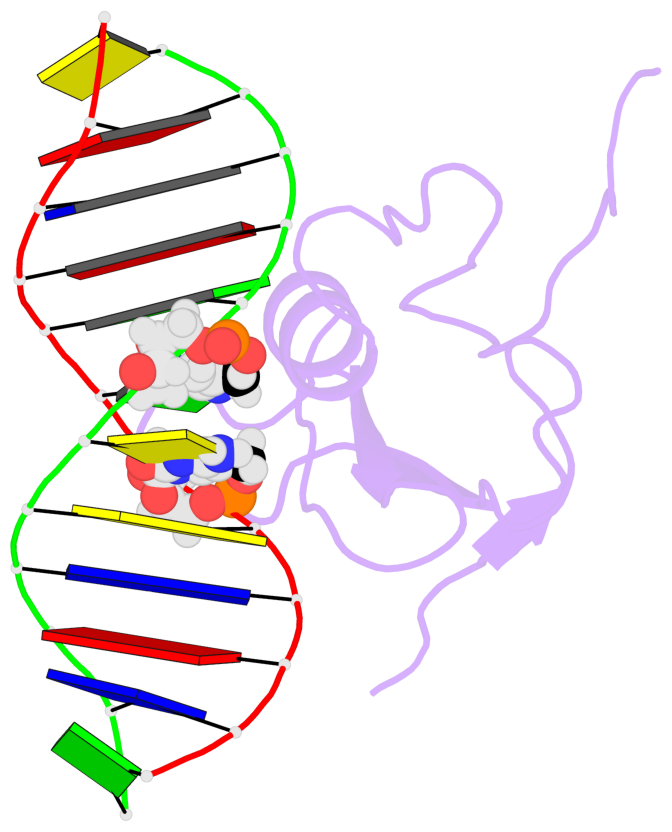
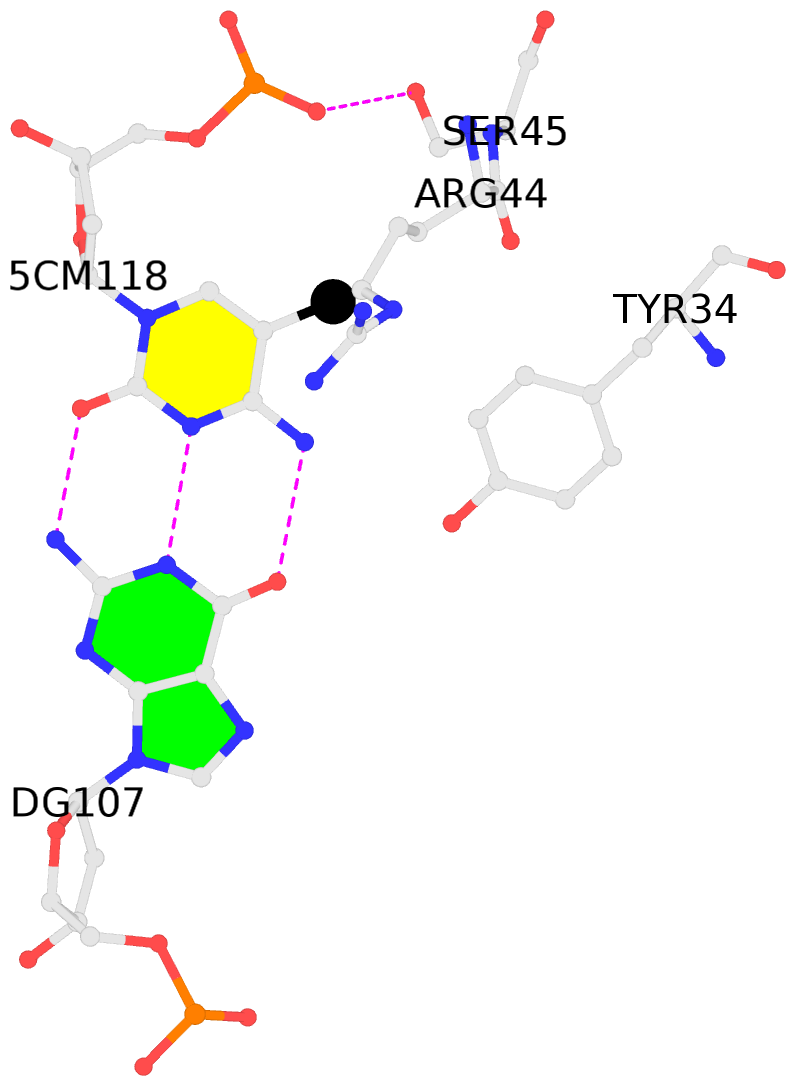
- The contacts include paired nucleotides (mostly a G in Watson-Crick G-C pairing), and
amino-acids within a 4.5-A distance cutoff to base atoms of 5mC.
- The structure is oriented in the base reference frame of 5mC, allowing for easy comparison
and direct superimposition between entries.
- The black sphere (•) denotes the 5-methyl carbon atom in 5mC.
No. 1 B.5CM106: hydrophobic-with-A.VAL20 is-WC-paired is-in-duplex [+]:CcG/cGG |
 |
|
No. 2 C.5CM118: other-contacts is-WC-paired is-in-duplex [-]:cGG/CcG |
 |
|