Summary information and primary citation
- PDB-id
- 1eiy; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- ligase-RNA
- Method
- X-ray (3.3 Å)
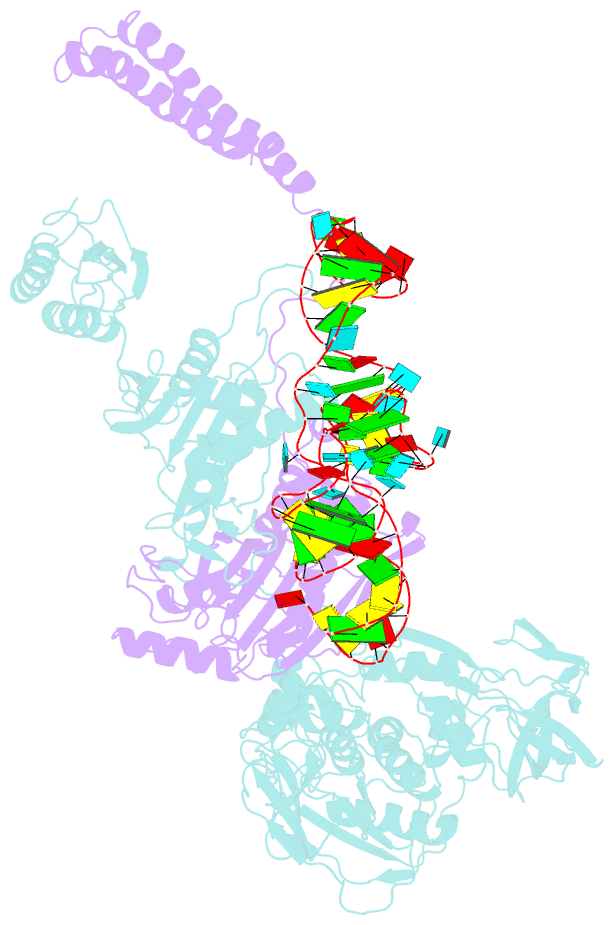
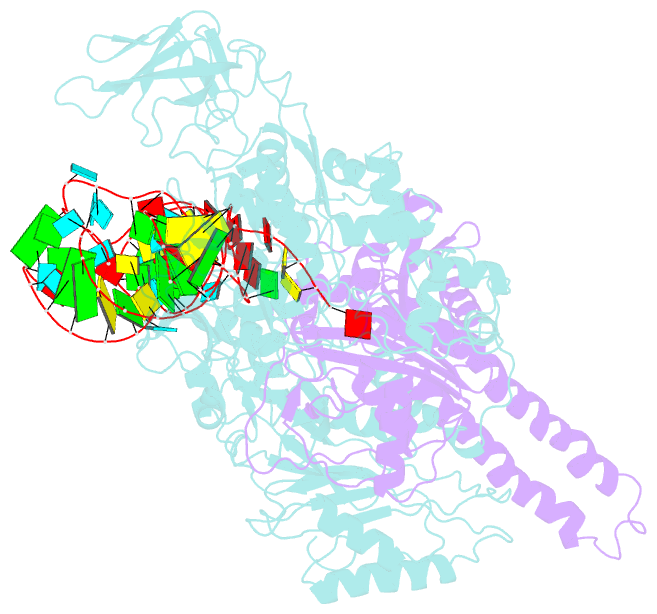
- Summary
- The crystal structure of phenylalanyl-trna synthetase from thermus thermophilus complexed with cognate trnaphe
- Reference
- Goldgur Y, Mosyak L, Reshetnikova L, Ankilova V, Lavrik O, Khodyreva S, Safro M (1997): "The crystal structure of phenylalanyl-tRNA synthetase from thermus thermophilus complexed with cognate tRNAPhe." Structure, 5, 59-68. doi: 10.1016/S0969-2126(97)00166-4.
- Abstract
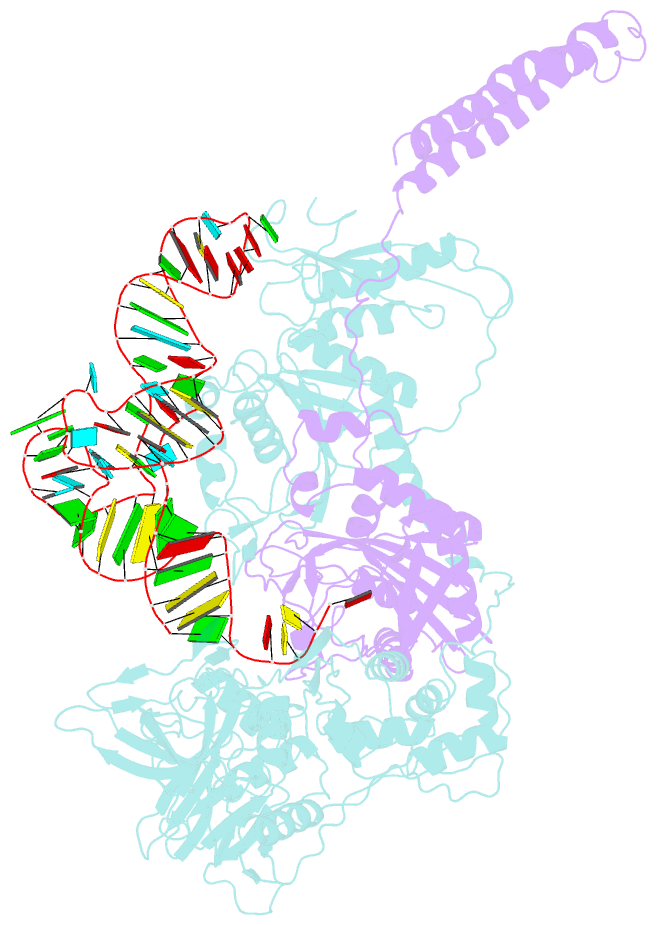
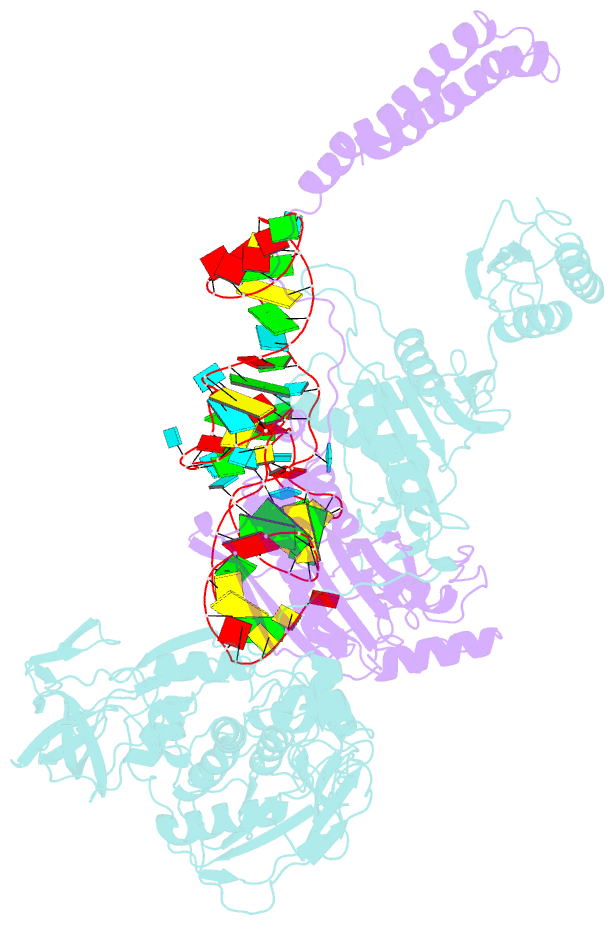
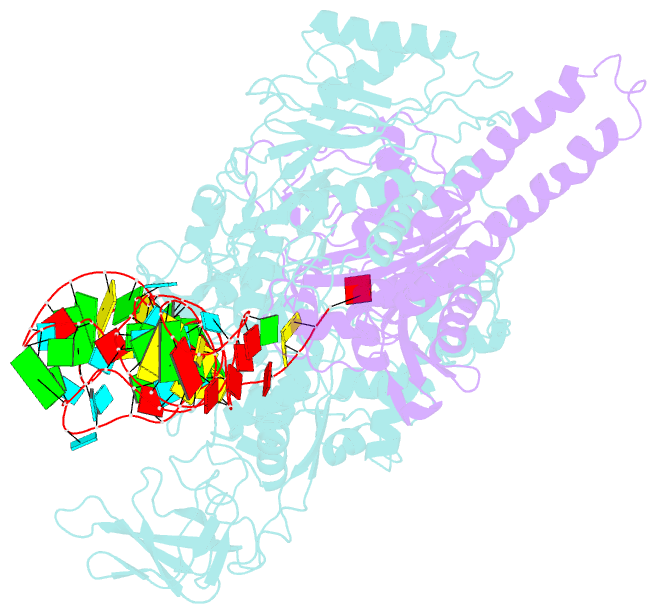
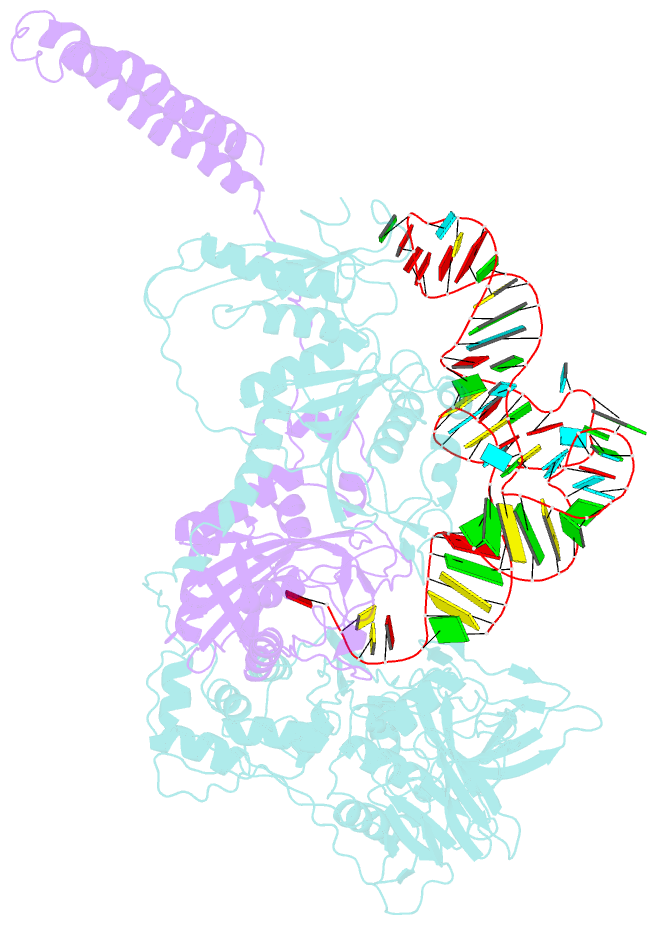
- Background: In the translation of the genetic code each aminoacyl-tRNA synthetase (aaRS) must recognize its own (cognate) tRNA and attach the corresponding amino acid to the acceptor end of tRNA, discriminating all the others. The(alphabeta)2 phenylalanyl-tRNA synthetase (PheRS) is one of the most complex enzymes in the aaRS family and is characterized by anomalous charging properties. Structurally, the enzyme belongs to class II aaRSs, as its catalytic domain is built around an antiparallel beta sheet, but functionally it resembles class I as it aminoacylates the 2'OH of the terminal ribose of tRNA (class II aaRSs aminoacylate the 3'OH). With the availability of the three-dimensional structure of the complex between multisubunit PheRS and tRNAPhe, a fuller picture of the specific tRNA-aaRS interactions is beginning to emerge.
Results: The crystal structure of Thermus thermophilus PheRS complexed with cognate tRNA has been solved at 3.28 A resolution. It reveals that one tRNAPhe molecule binds across all four PheRS subunits. The interactions of PheRS with tRNA stabilize the flexible N-terminal part of the alpha subunit, which appeared to form the enzyme's 11th domain, comprising a coiled-coil structure (helical arm) built up of two long antiparallel alpha helices. The helical arms are similar to those observed in SerRS and are in the same relative orientation with respect to the catalytic domain. Anticodon recognition upon tRNA binding is performed by the B8 domain, the structure of which is similar to that of the RNA-binding domain (RBD) of the small spliceosomal protein U1A. The Th. thermophilus PheRS approaches the anticodon loop from the minor groove side.
Conclusions: The mode of interactions with tRNA explains the absolute necessity for the (alphabeta)2 architecture of PheRS. The interactions of tRNAPhe with PheRS and particularly with the coiled-coil domain of the alpha subunit result in conformational changes in TPsiC and D loops seen by comparison with uncomplexed yeast tRNAPhe. The tRNAPhe is a newly recognized type of RNA molecule specifically interacting with the RBD fold. In addition, a new type of anticodon-binding domain emerges in the aaRS family. The uniqueness of PheRS in charging 2'OH of tRNA is dictated by the size of its adenine-binding pocket and by the local conformation of the tRNA's CCA end.