Summary information and primary citation
- PDB-id
- 1k82; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- hydrolase-DNA
- Method
- X-ray (2.1 Å)
- Summary
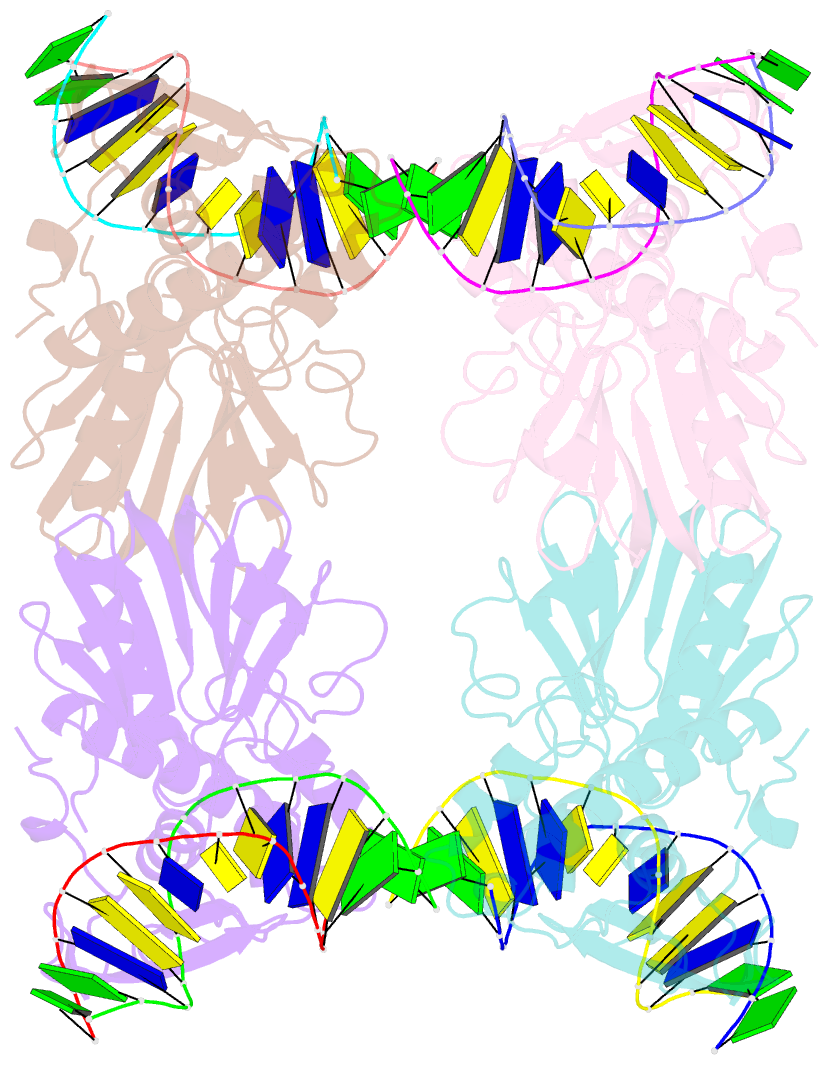
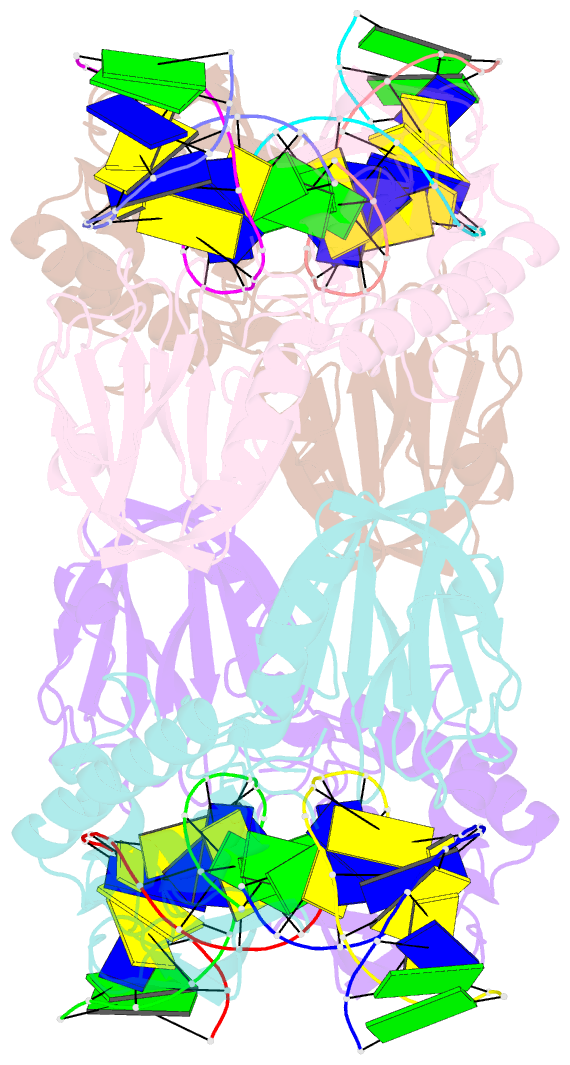
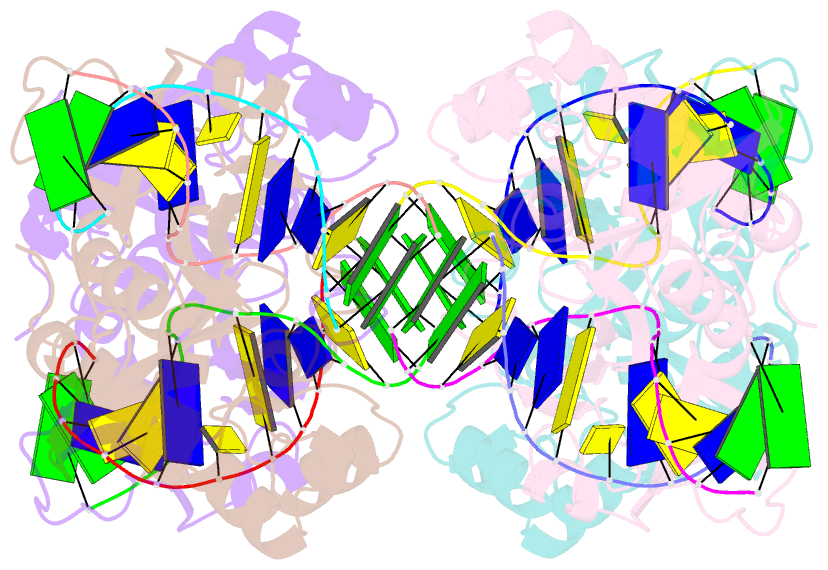
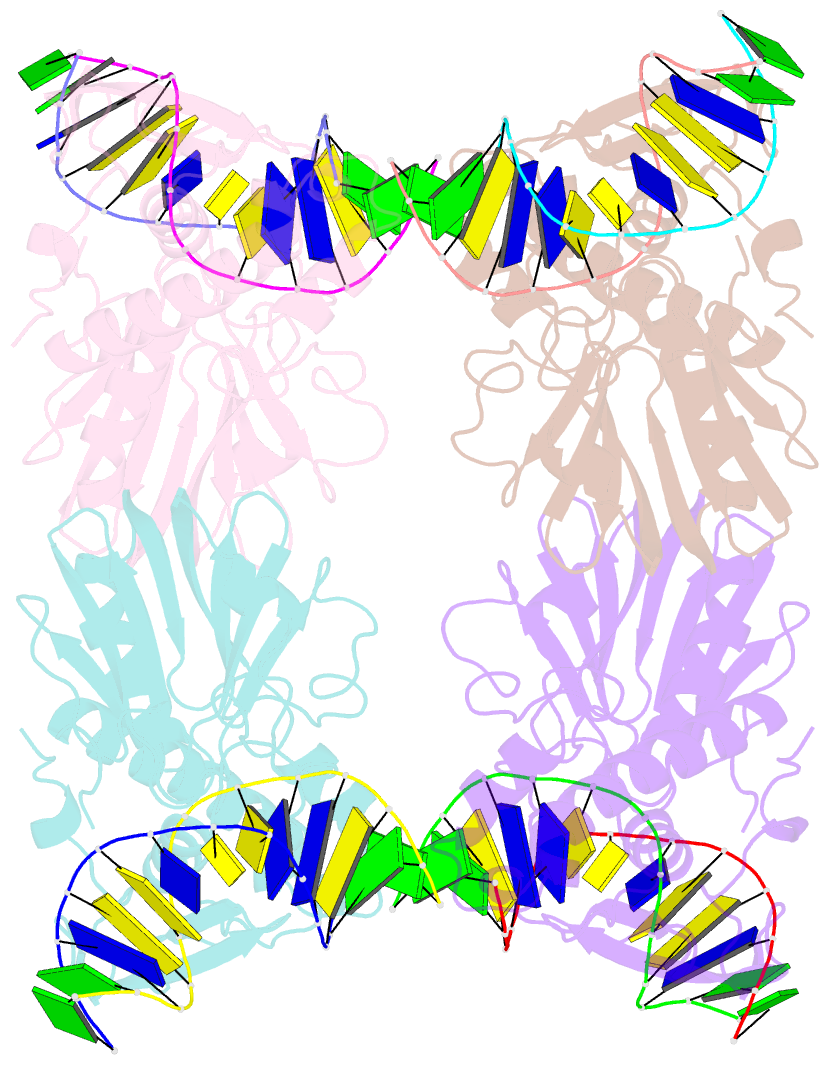
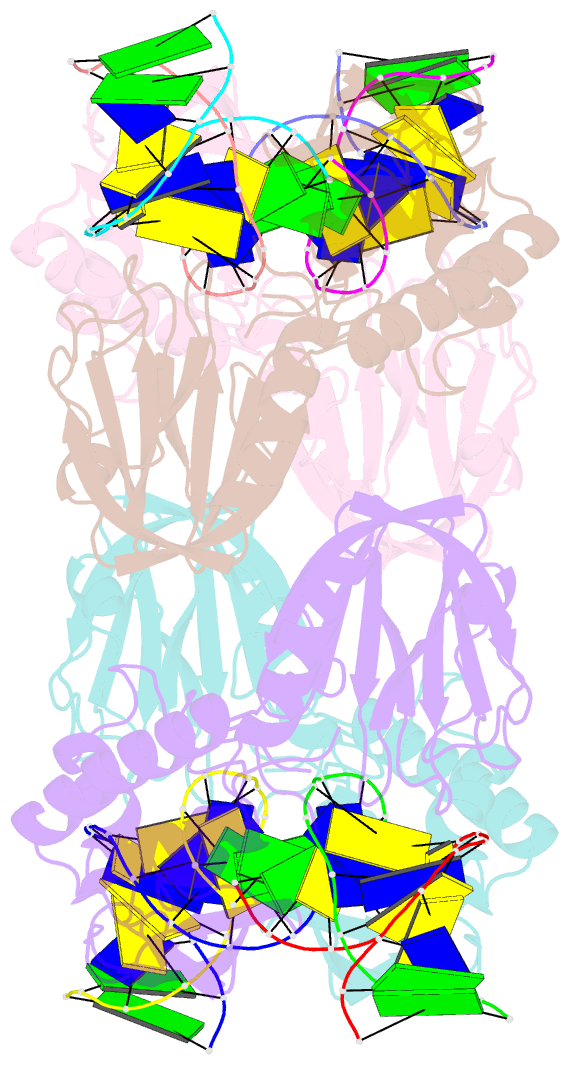
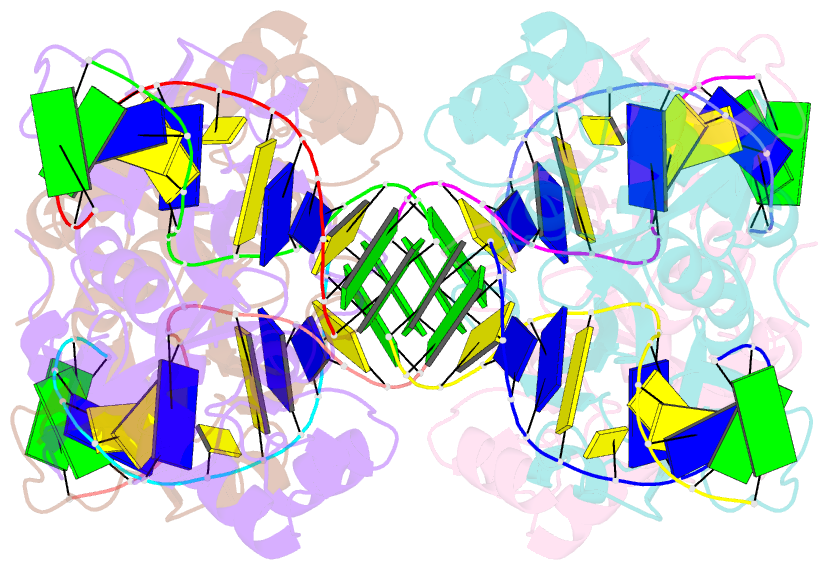
- Crystal structure of e.coli formamidopyrimidine-DNA glycosylase (fpg) covalently trapped with DNA
- Reference
- Gilboa R, Zharkov DO, Golan G, Fernandes AS, Gerchman SE, Matz E, Kycia JH, Grollman AP, Shoham G (2002): "Structure of formamidopyrimidine-DNA glycosylase covalently complexed to DNA." J.Biol.Chem., 277, 19811-19816. doi: 10.1074/jbc.M202058200.
- Abstract
- Formamidopyrimidine-DNA glycosylase (Fpg) is a DNA repair enzyme that excises oxidized purines from damaged DNA. The Schiff base intermediate formed during this reaction between Escherichia coli Fpg and DNA was trapped by reduction with sodium borohydride, and the structure of the resulting covalently cross-linked complex was determined at a 2.1-A resolution. Fpg is a bilobal protein with a wide, positively charged DNA-binding groove. It possesses a conserved zinc finger and a helix-two turn-helix motif that participate in DNA binding. The absolutely conserved residues Lys-56, His-70, Asn-168, and Arg-258 form hydrogen bonds to the phosphodiester backbone of DNA, which is sharply kinked at the lesion site. Residues Met-73, Arg-109, and Phe-110 are inserted into the DNA helix, filling the void created by nucleotide eversion. A deep hydrophobic pocket in the active site is positioned to accommodate an everted base. Structural analysis of the Fpg-DNA complex reveals essential features of damage recognition and the catalytic mechanism of Fpg.