Summary information and primary citation
- PDB-id
- 1kdh; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transferase-DNA
- Method
- X-ray (3.0 Å)
- Summary
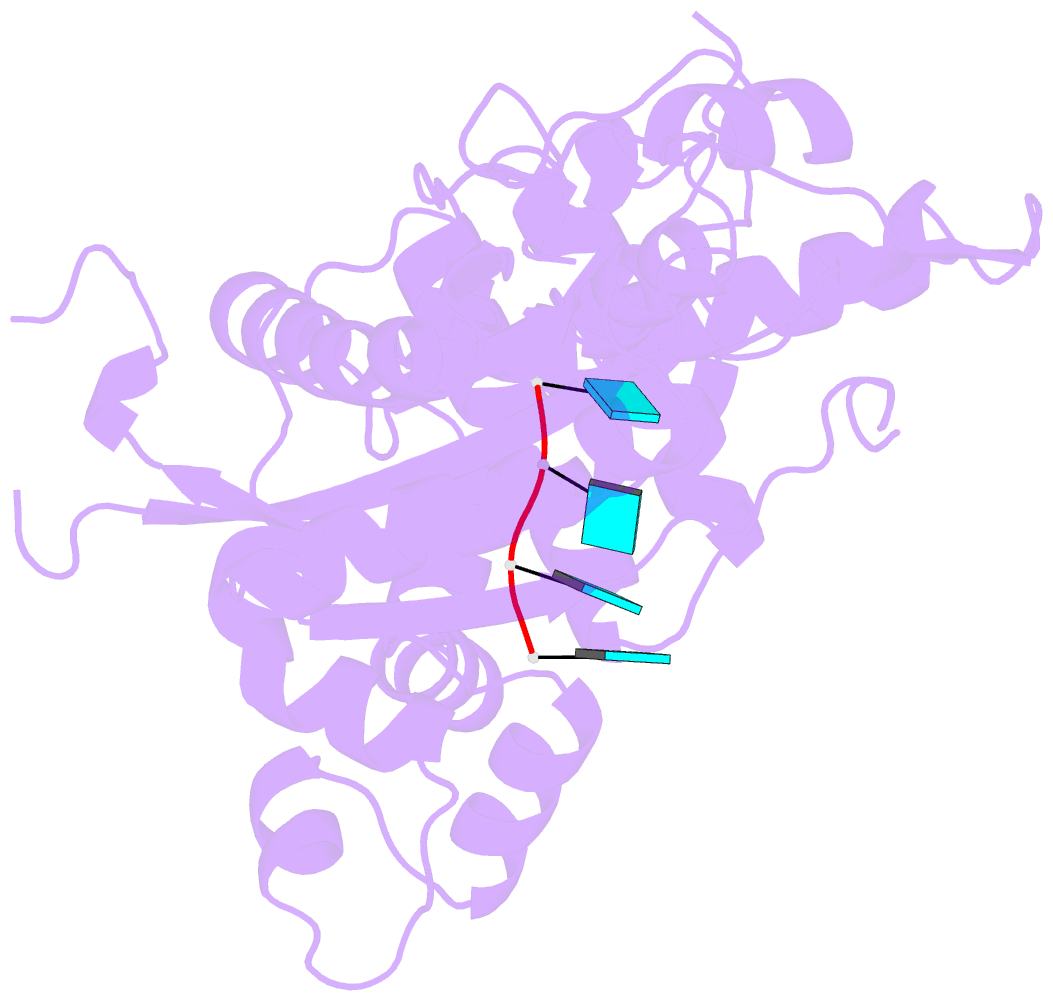
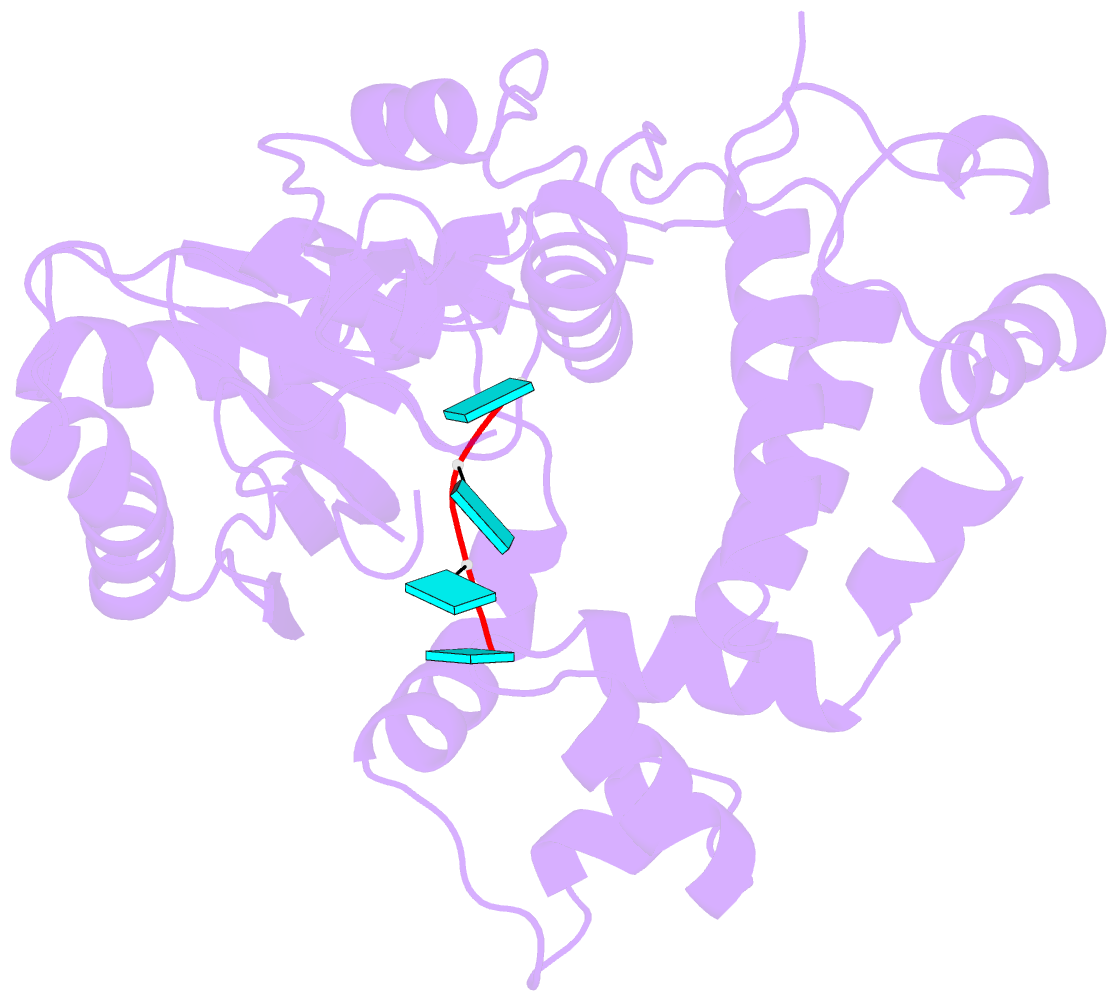
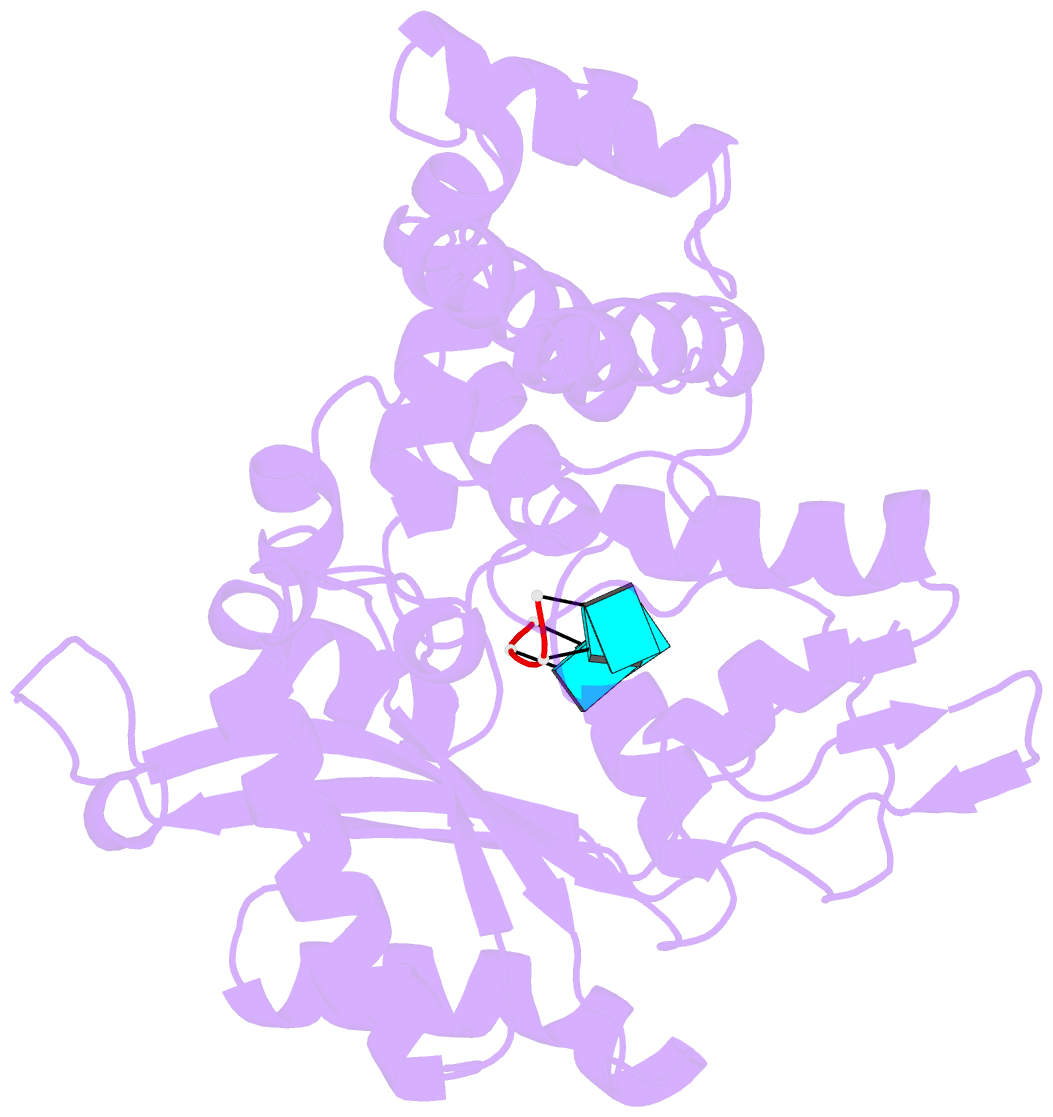
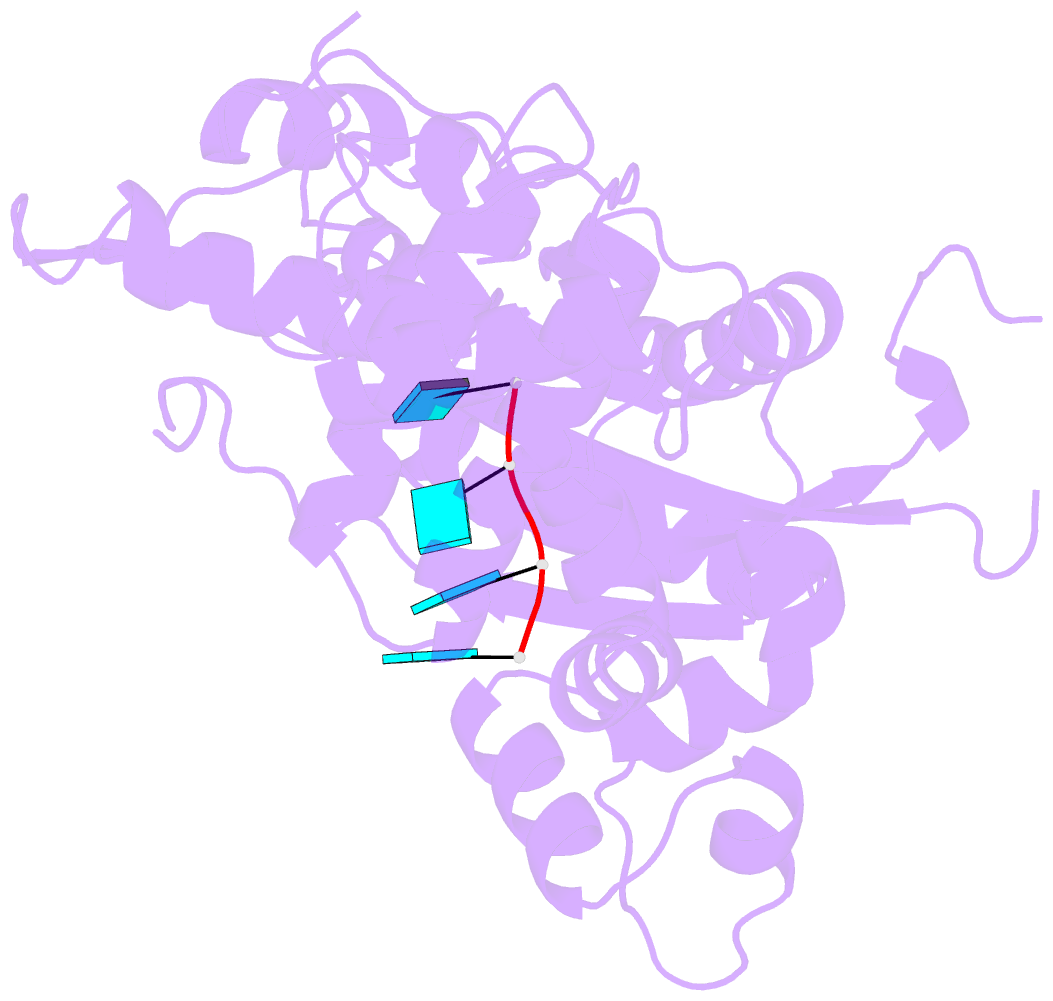
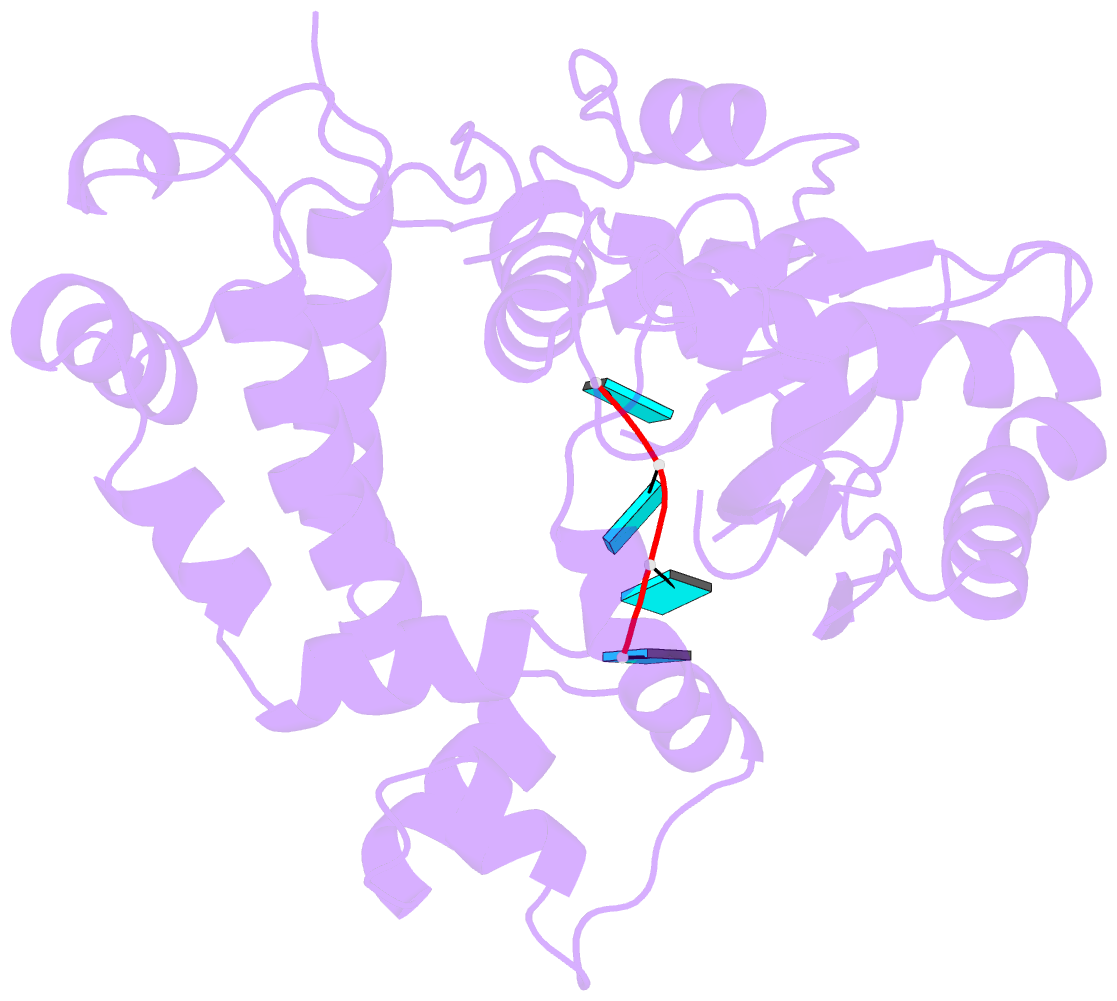
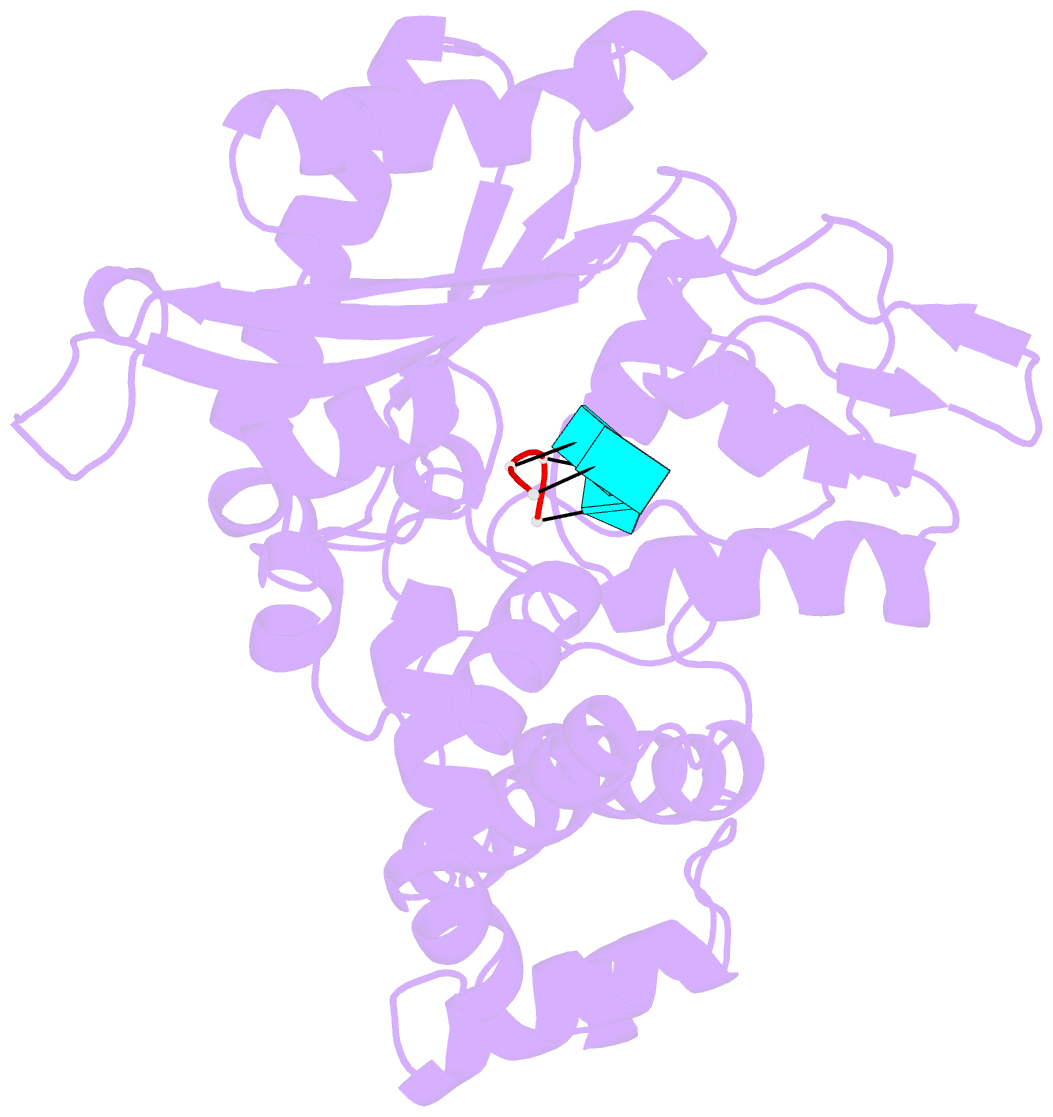
- Binary complex of murine terminal deoxynucleotidyl transferase with a primer single stranded DNA
- Reference
- Delarue M, Boule JB, Lescar J, Expert-Bezancon N, Jourdan N, Sukumar N, Rougeon F, Papanicolaou C (2002): "Crystal structures of a template-independent DNA polymerase: murine terminal deoxynucleotidyltransferase." EMBO J., 21, 427-439. doi: 10.1093/emboj/21.3.427.
- Abstract
- The crystal structure of the catalytic core of murine terminal deoxynucleotidyltransferase (TdT) at 2.35 A resolution reveals a typical DNA polymerase beta-like fold locked in a closed form. In addition, the structures of two different binary complexes, one with an oligonucleotide primer and the other with an incoming ddATP-Co(2+) complex, show that the substrates and the two divalent ions in the catalytic site are positioned in TdT in a manner similar to that described for the human DNA polymerase beta ternary complex, suggesting a common two metal ions mechanism of nucleotidyl transfer in these two proteins. The inability of TdT to accommodate a template strand can be explained by steric hindrance at the catalytic site caused by a long lariat-like loop, which is absent in DNA polymerase beta. However, displacement of this discriminating loop would be sufficient to unmask a number of evolutionarily conserved residues, which could then interact with a template DNA strand. The present structure can be used to model the recently discovered human polymerase mu, with which it shares 43% sequence identity.