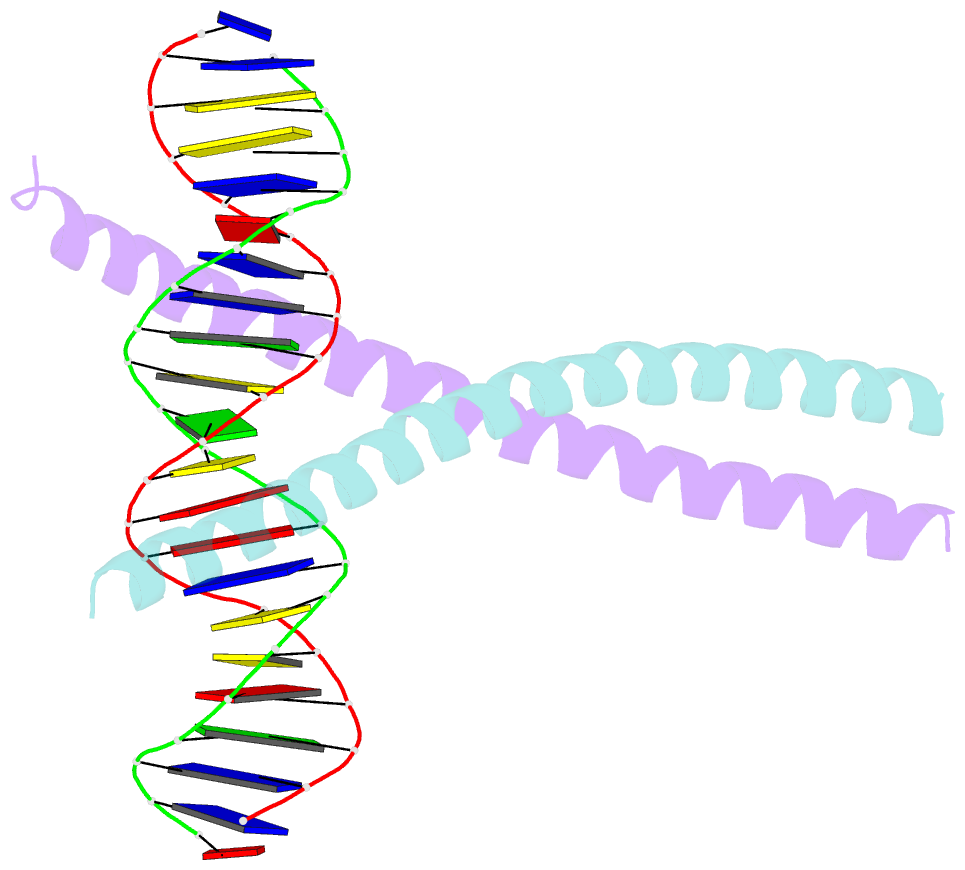
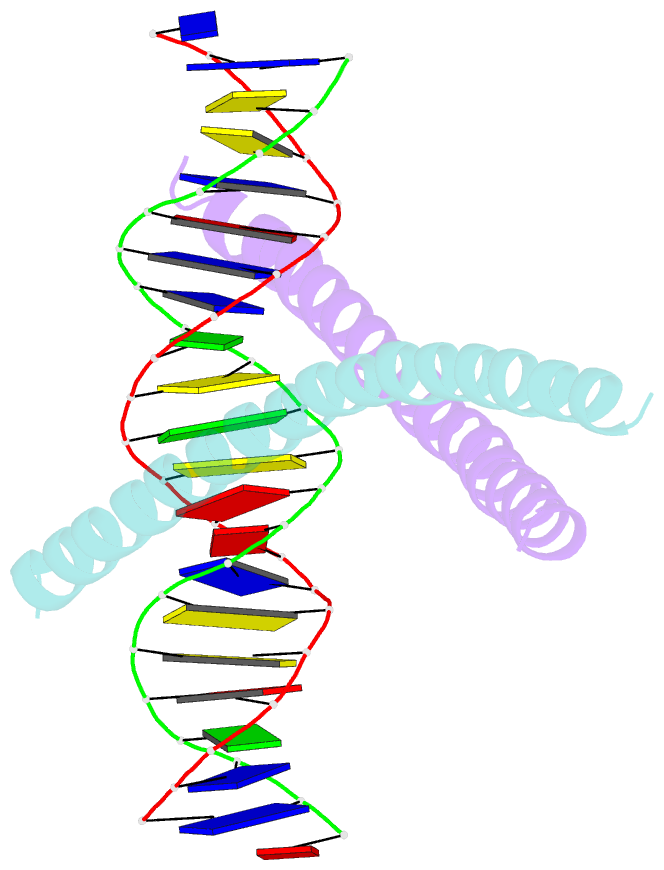
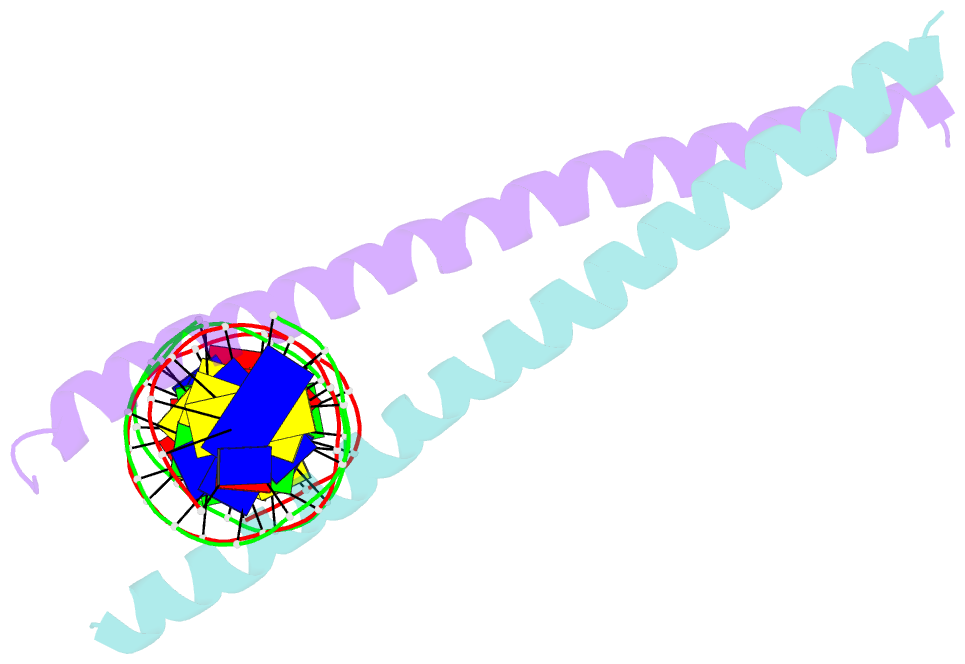
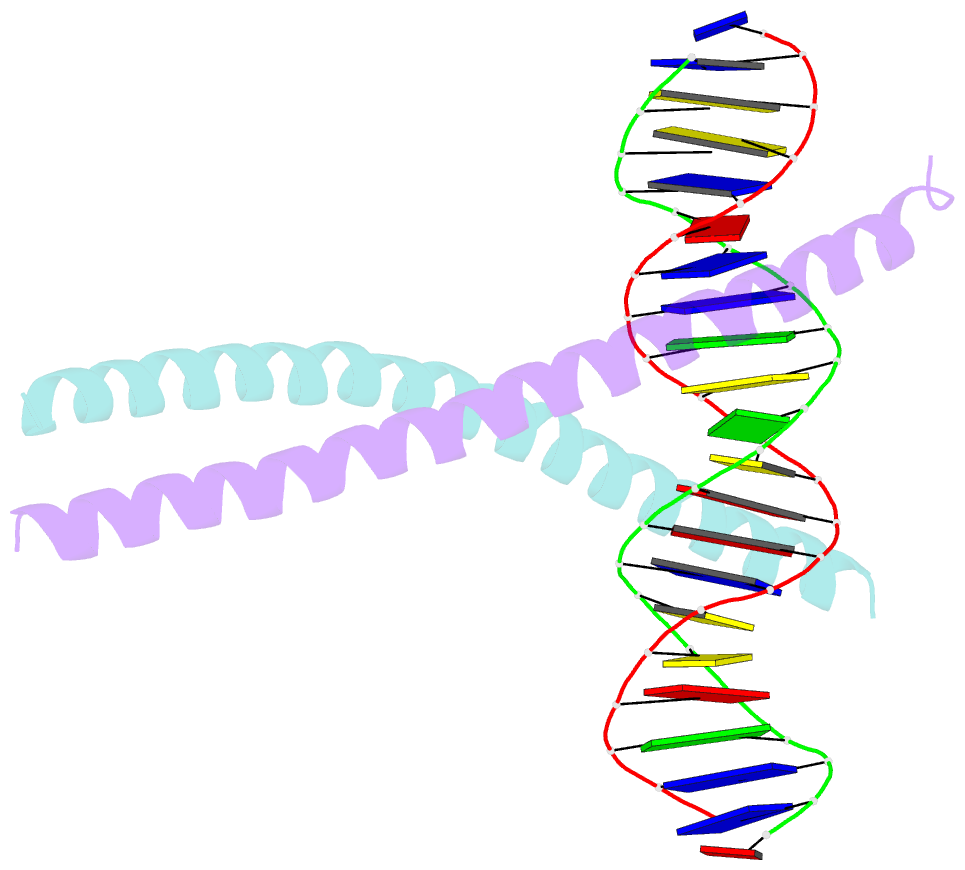
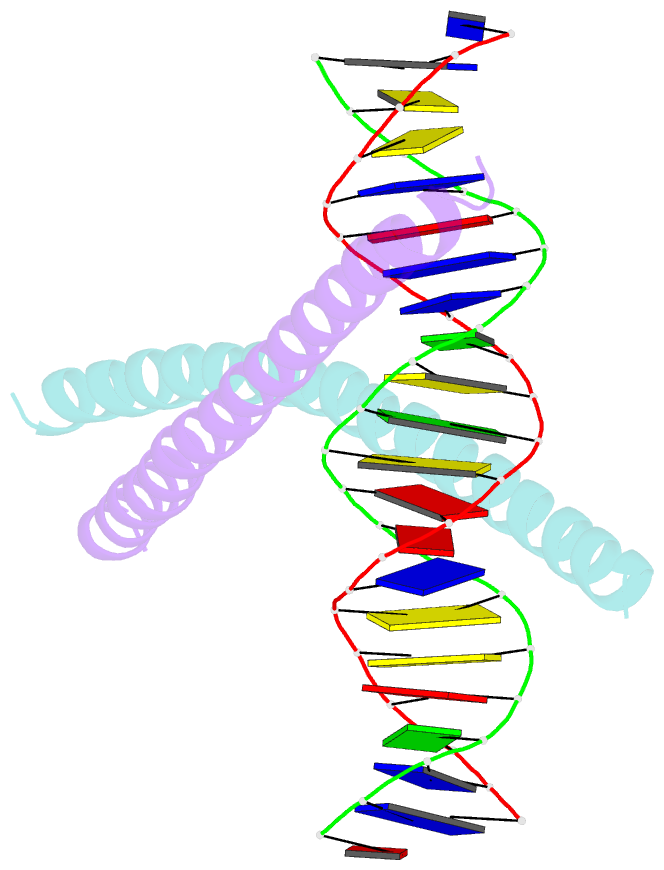
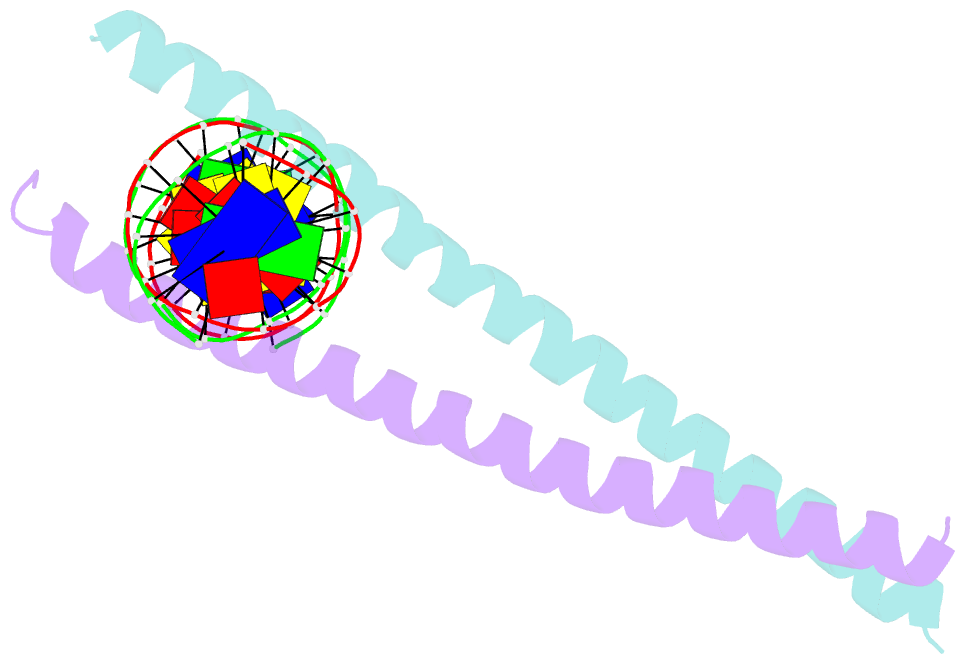
Summary information and primary citation
- PDB-id
- 1nwq; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transcription-DNA
- Method
- X-ray (2.8 Å)
- Summary
- Crystal structure of c-ebpalpha-DNA complex
- Reference
- Miller M, Shuman JD, Sebastian T, Dauter Z, Johnson PF (2003): "Structural Basis for DNA Recognition by the Basic Region Leucine Zipper
Transcription Factor CCAAT/enhancer Binding Protein Alpha." J.Biol.Chem., 278, 15178-15184. doi: 10.1074/jbc.M300417200.
- Abstract
- CCAAT/enhancer-binding proteins (C/EBPs) are basic region leucine zipper (bZIP) transcription factors that regulate cell differentiation, growth, survival, and inflammation. To understand the molecular basis of DNA recognition by the C/EBP family we determined the x-ray structure of a C/EBPalpha bZIP polypeptide bound to its cognate DNA site (A(-5)T(-4)T(-3)G(-2)C(-1)G(1)C(2)A(3)A(4)T(5)) and characterized several basic region mutants. Binding specificity is provided by interactions of basic region residues Arg(289), Asn(292), Ala(295), Val(296), Ser(299), and Arg(300) with DNA bases. A striking feature of the C/EBPalpha protein-DNA interface that distinguishes it from known bZIP-DNA complexes is the central role of Arg(289), which is hydrogen-bonded to base A(3), phosphate, Asn(292) (invariant in bZIPs), and Asn(293). The conformation of Arg(289) is also restricted by Tyr(285). In accordance with the structural model, mutation of Arg(289) or a pair of its interacting partners (Tyr(285) and Asn(293)) abolished C/EBPalpha binding activity. Val(296) (Ala in most other bZIPs) contributes to C/EBPalpha specificity by discriminating against purines at position -3 and imposing steric restraints on the invariant Arg(300). Mutating Val(296) to Ala strongly enhanced C/EBPalpha binding to cAMP response element (CRE) sites while retaining affinity for C/EBP sites. Thus, Arg(289) is essential for formation of the complementary protein-DNA interface, whereas Val(296) functions primarily to restrict interactions with related sequences such as CRE sites rather than specifying binding to C/EBP sites. Our studies also help to explain the phenotypes of mice carrying targeted mutations in the C/EBPalpha bZIP region.