Summary information and primary citation
- PDB-id
- 1oe4; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- hydrolase-DNA
- Method
- X-ray (2.0 Å)
- Summary
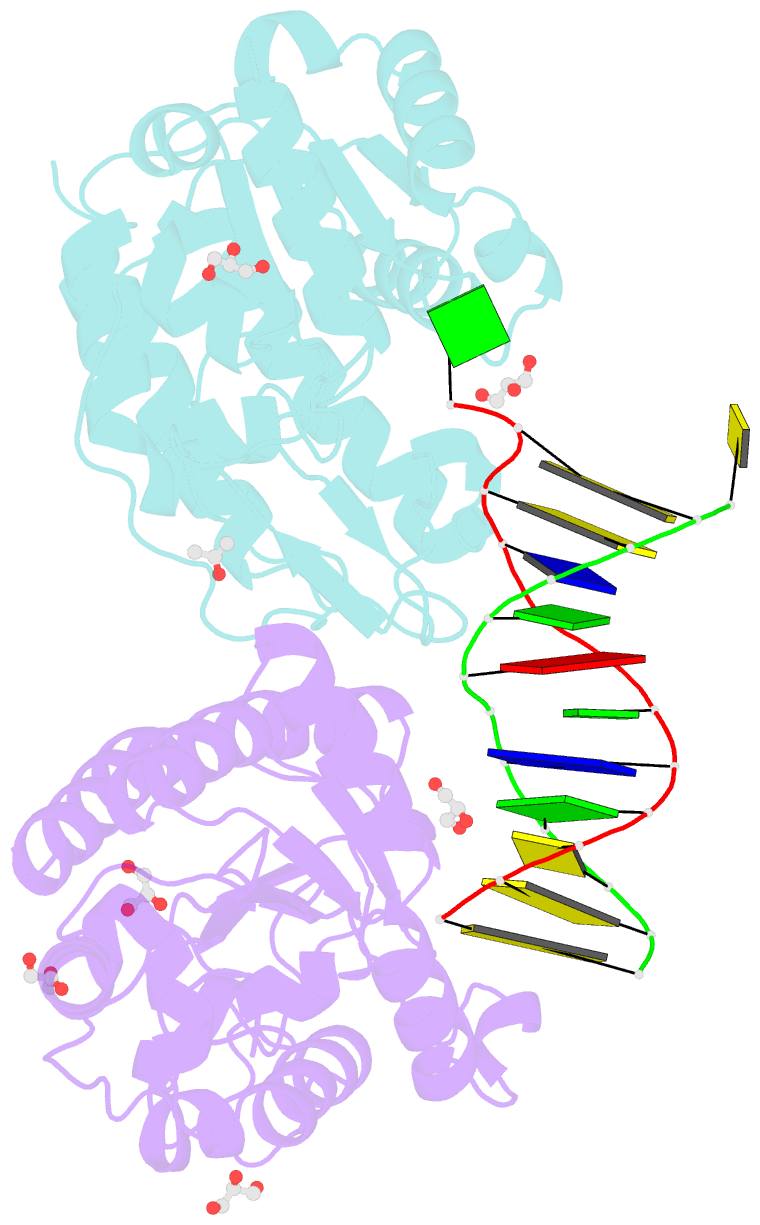
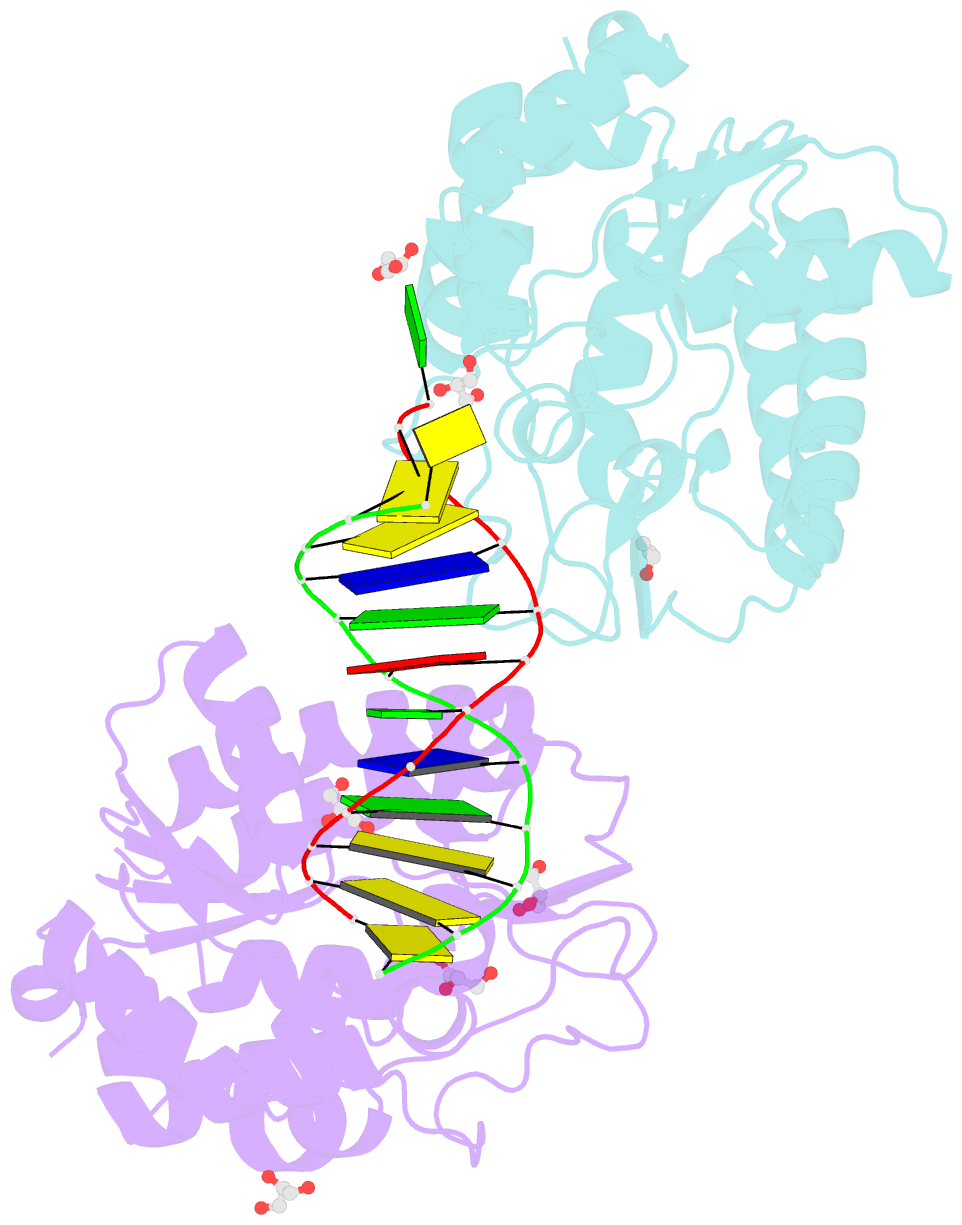
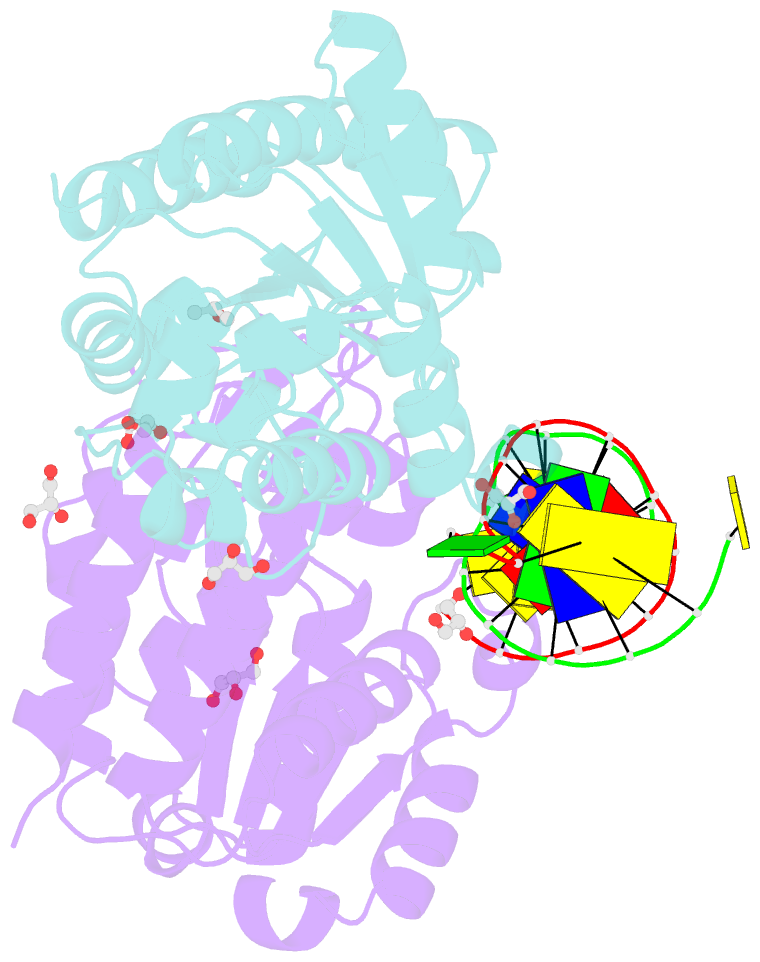
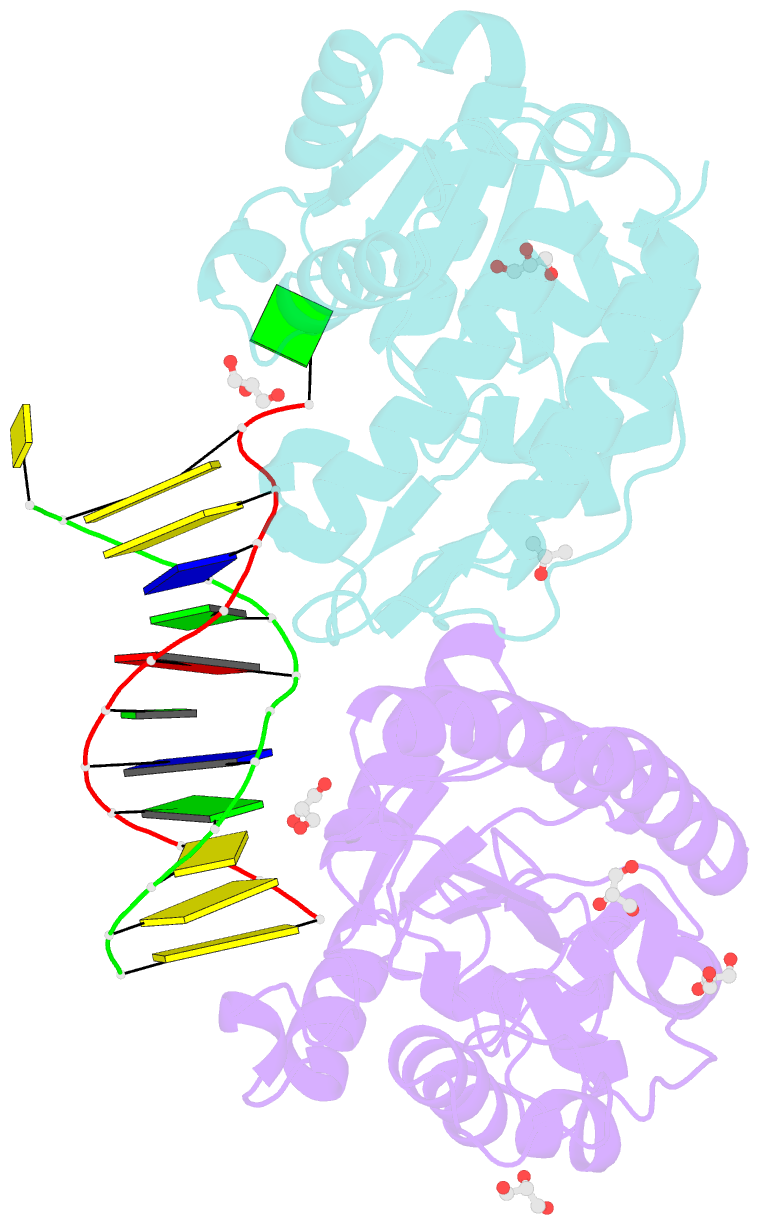
- Xenopus smug1, an anti-mutator uracil-DNA glycosylase
- Reference
- Wibley JEA, Waters TR, Haushalter K, Verdine GL, Pearl LH (2003): "Structure and Specificity of the Vertebrate Anti-Mutator Uracil-DNA Glycosylase Smug1." Mol.Cell, 11, 1647. doi: 10.1016/S1097-2765(03)00235-1.
- Abstract
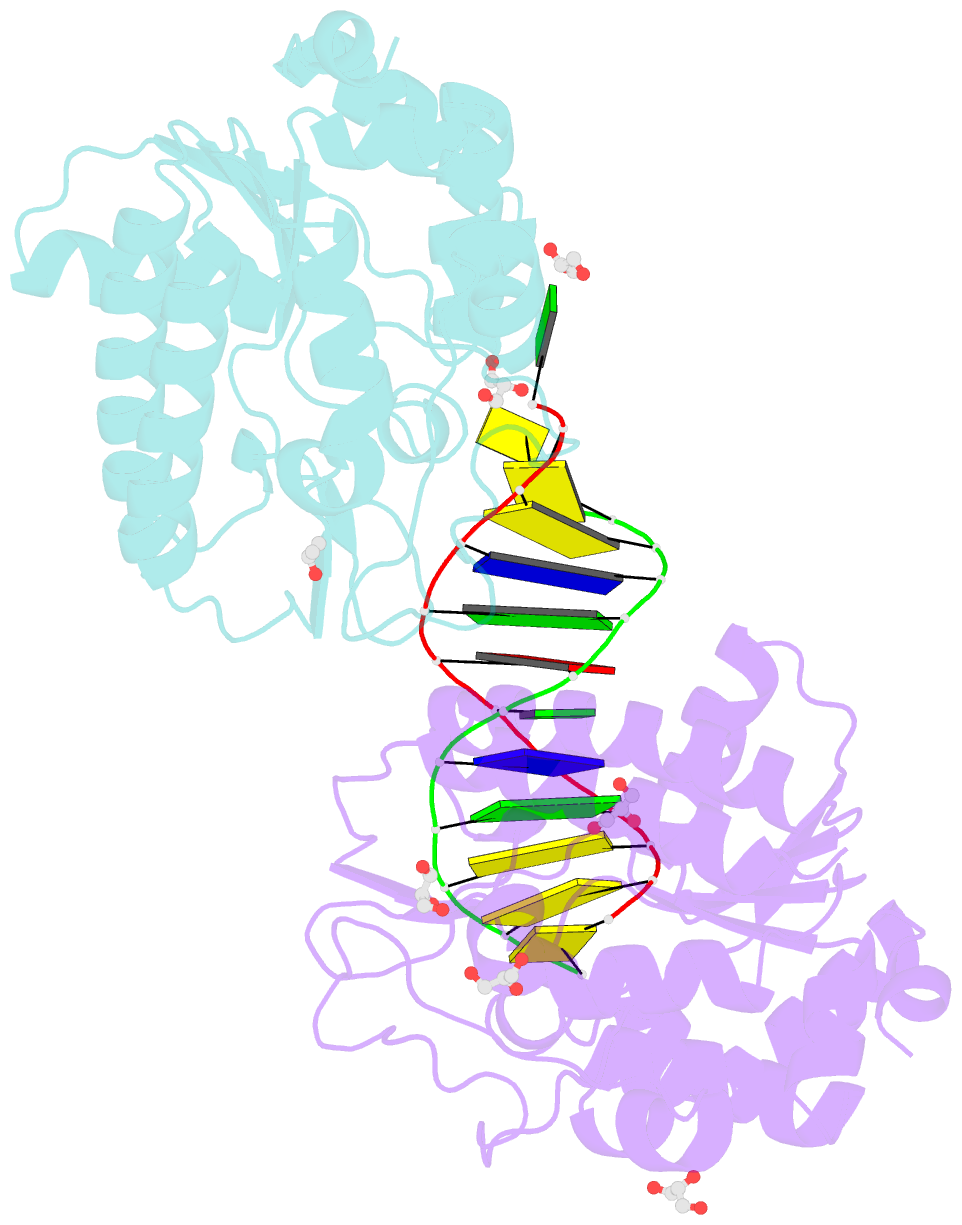
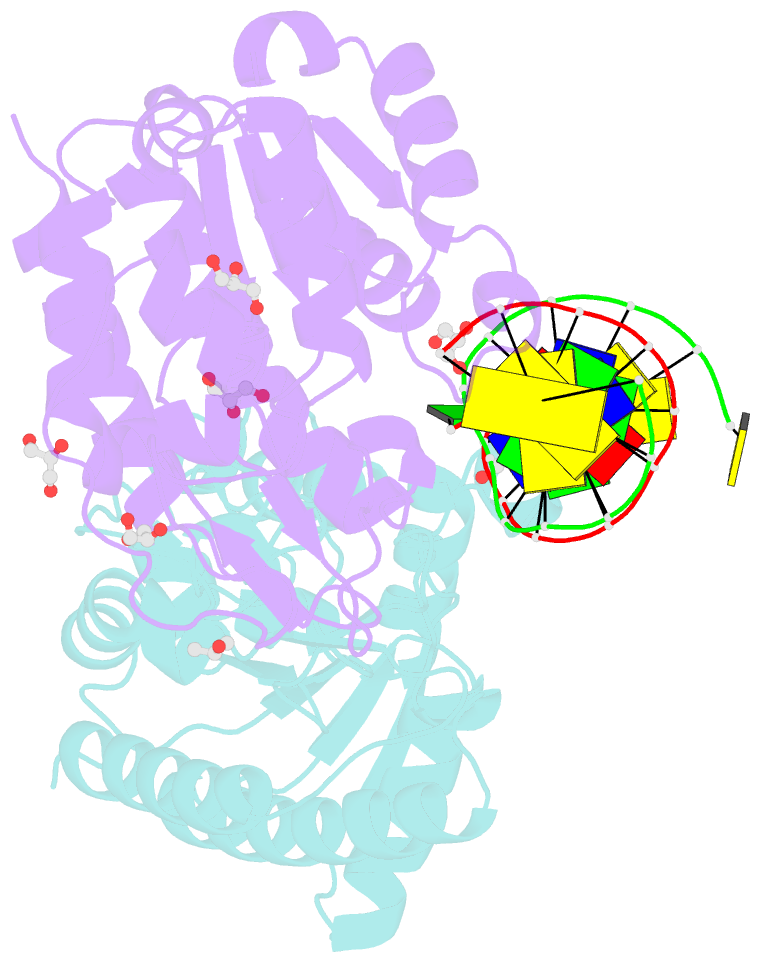
- Cytosine deamination is a major promutagenic process, generating G:U mismatches that can cause transition mutations if not repaired. Uracil is also introduced into DNA via nonmutagenic incorporation of dUTP during replication. In bacteria, uracil is excised by uracil-DNA glycosylases (UDG) related to E. coli UNG, and UNG homologs are found in mammals and viruses. Ung knockout mice display no increase in mutation frequency due to a second UDG activity, SMUG1, which is specialized for antimutational uracil excision in mammalian cells. Remarkably, SMUG1 also excises the oxidation-damage product 5-hydroxymethyluracil (HmU), but like UNG is inactive against thymine (5-methyluracil), a chemical substructure of HmU. We have solved the crystal structure of SMUG1 complexed with DNA and base-excision products. This structure indicates a more invasive interaction with dsDNA than observed with other UDGs and reveals an elegant water displacement/replacement mechanism that allows SMUG1 to exclude thymine from its active site while accepting HmU.