Summary information and primary citation
- PDB-id
- 1pvo; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transcription-RNA
- Method
- X-ray (3.0 Å)
- Summary
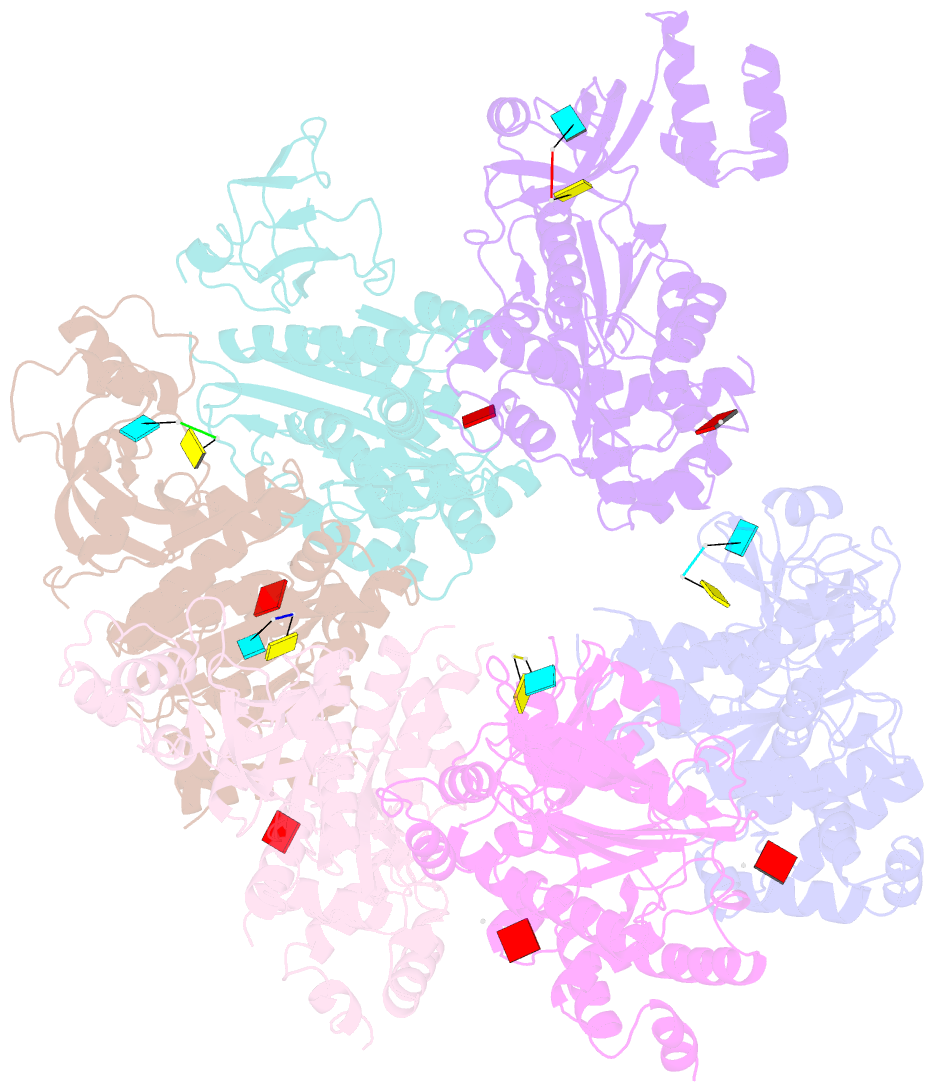
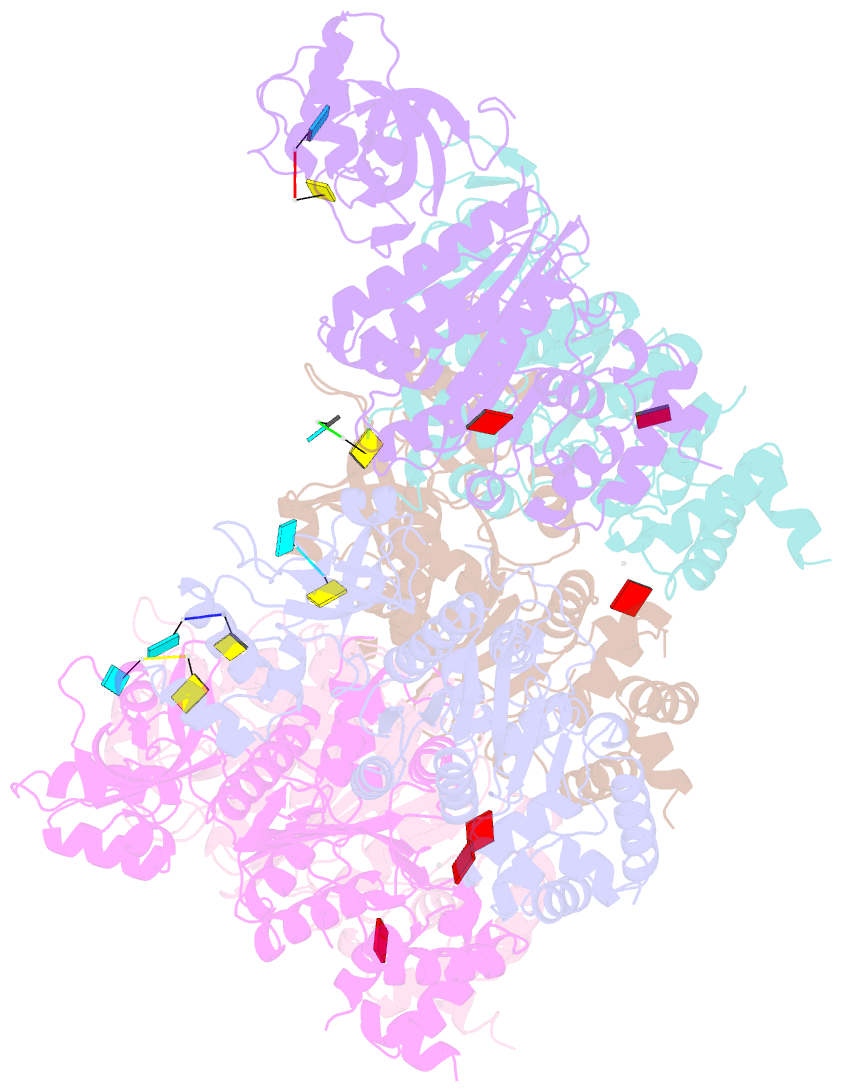
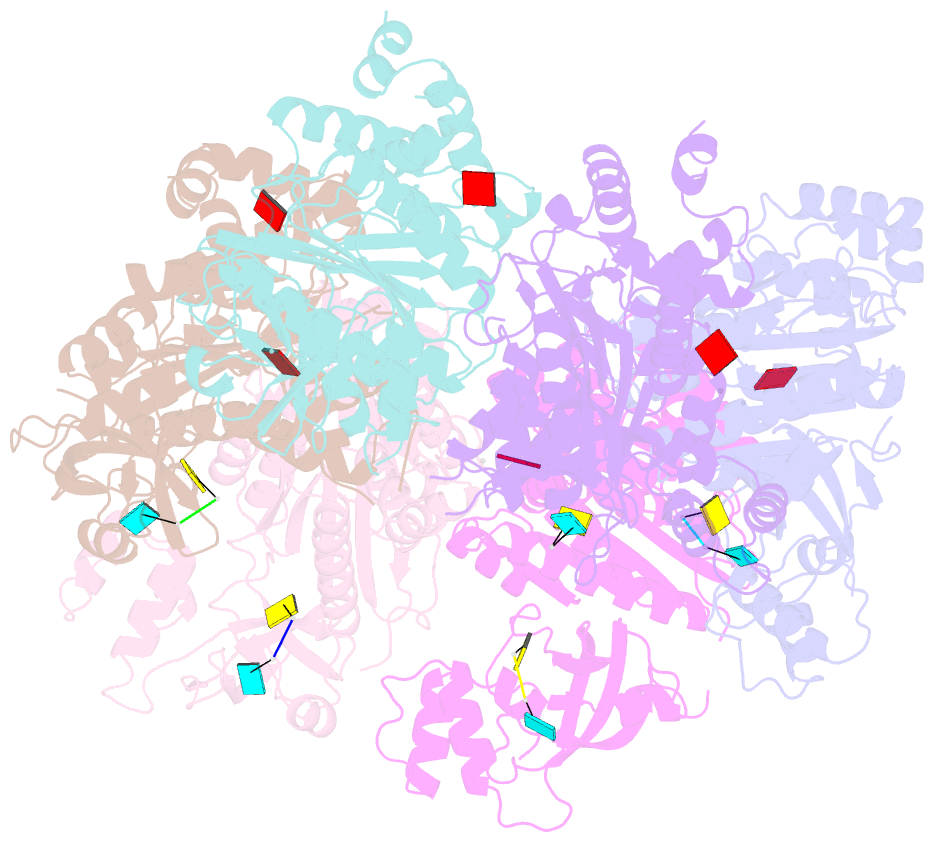
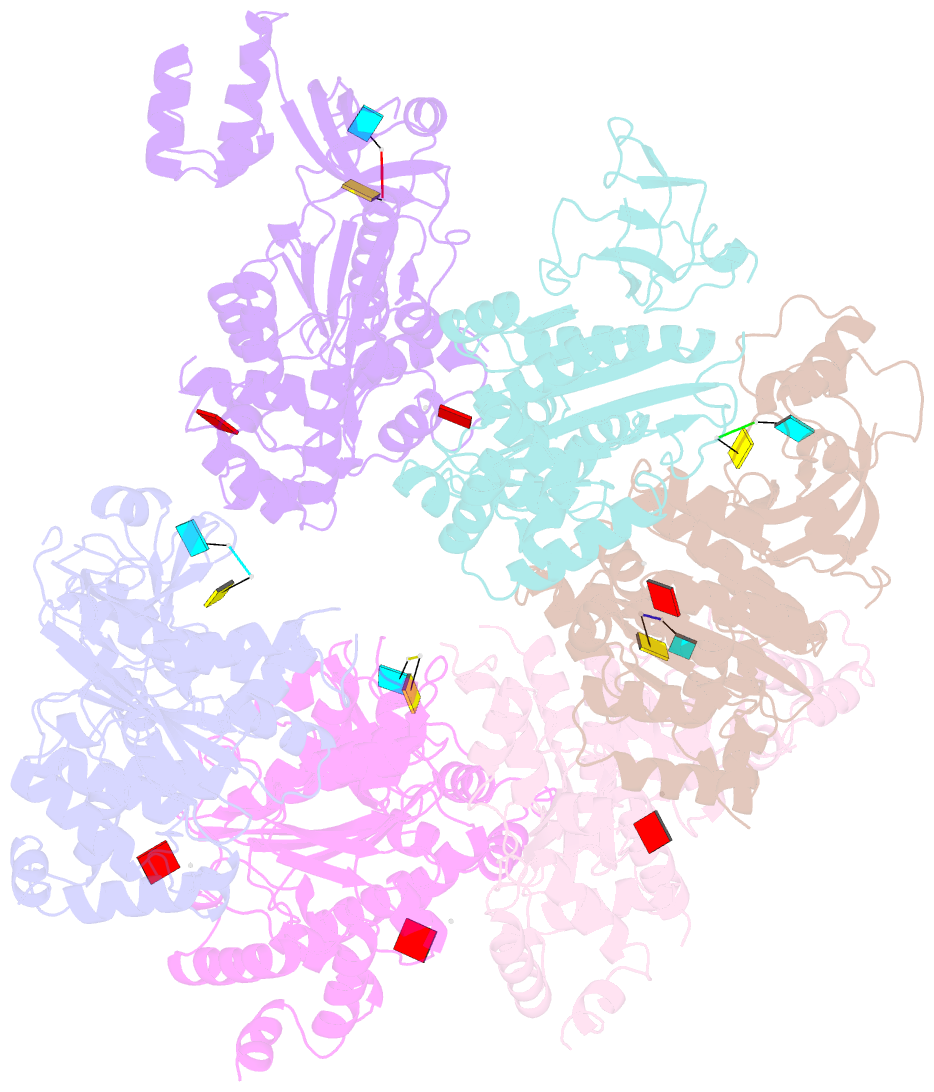
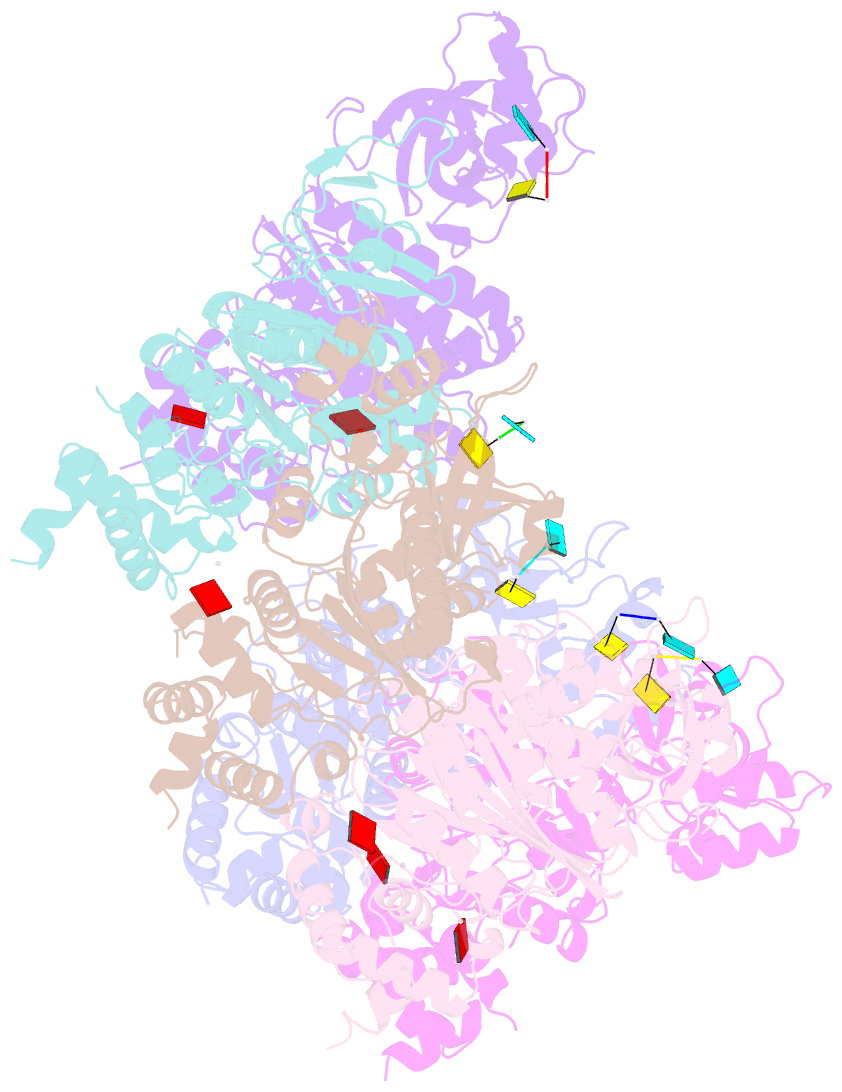
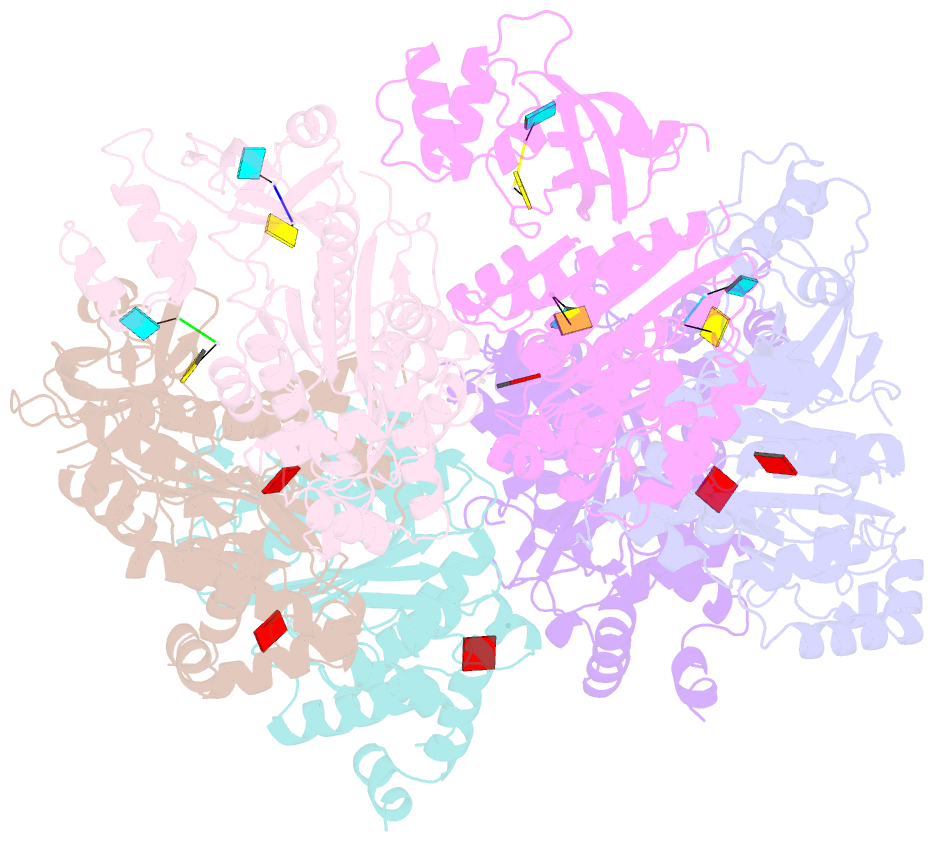
- X-ray crystal structure of rho transcription termination factor in complex with ssrna substrate and anppnp
- Reference
- Skordalakes E, Berger JM (2003): "Structure of the Rho transcription terminator: mechanism of mRNA recognition and helicase loading." Cell(Cambridge,Mass.), 114, 135-146. doi: 10.1016/S0092-8674(03)00512-9.
- Abstract
- In bacteria, one of the major transcriptional termination mechanisms requires a RNA/DNA helicase known as the Rho factor. We have determined two structures of Rho complexed with nucleic acid recognition site mimics in both free and nucleotide bound states to 3.0 A resolution. Both structures show that Rho forms a hexameric ring in which two RNA binding sites--a primary one responsible for target mRNA recognition and a secondary one required for mRNA translocation and unwinding--point toward the center of the ring. Rather than forming a closed ring, the Rho hexamer is split open, resembling a "lock washer" in its global architecture. The distance between subunits at the opening is sufficiently wide (12 A) to accommodate single-stranded RNA. This open configuration most likely resembles a state poised to load onto mRNA and suggests how related ring-shaped enzymes may be breached to bind nucleic acids.