Summary information and primary citation
- PDB-id
- 1q3v; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- replication-DNA
- Method
- X-ray (2.91 Å)
- Summary
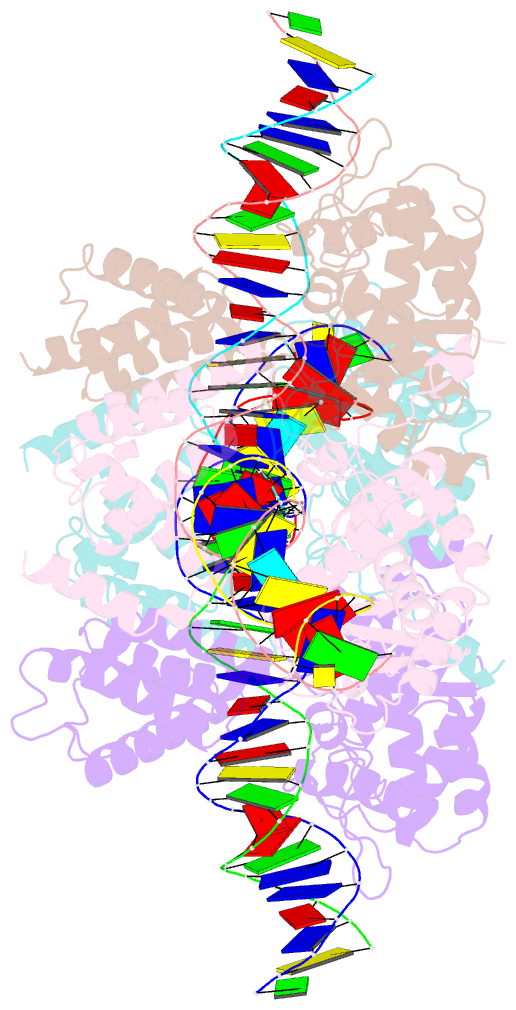
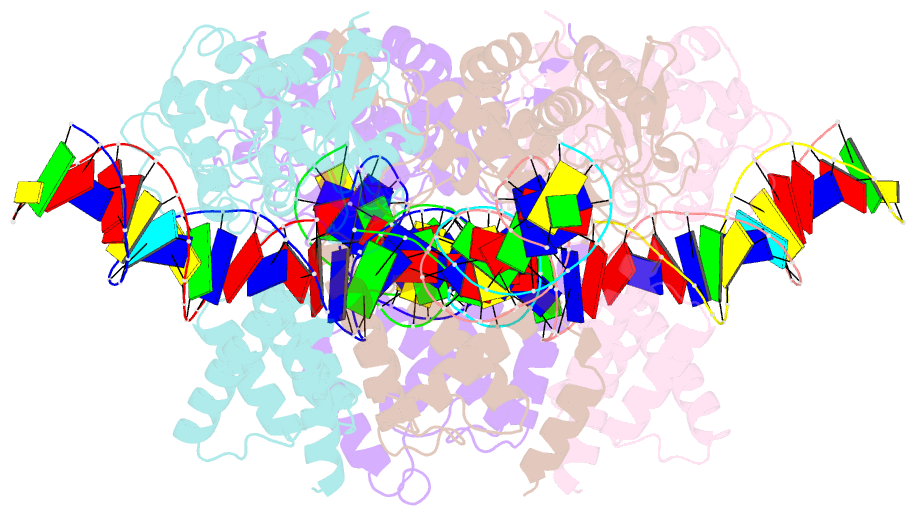
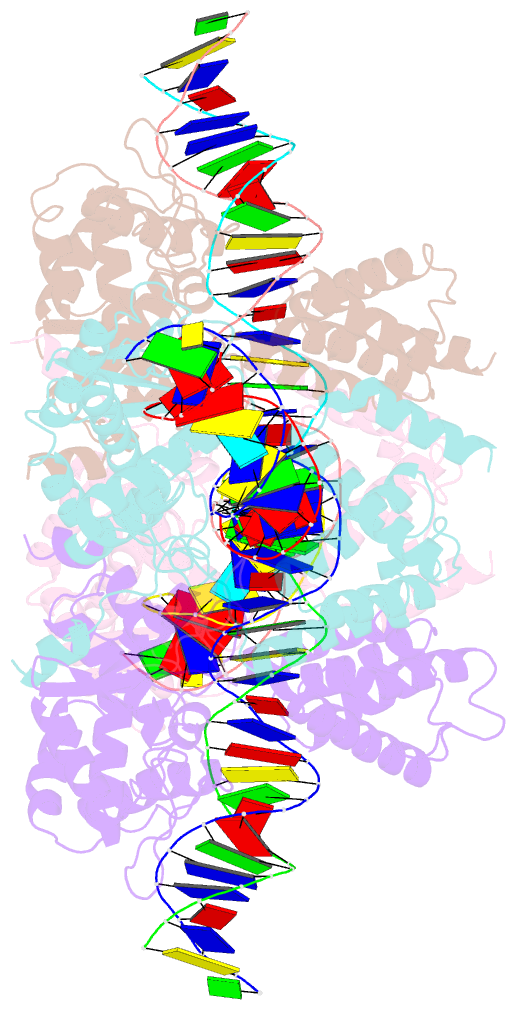
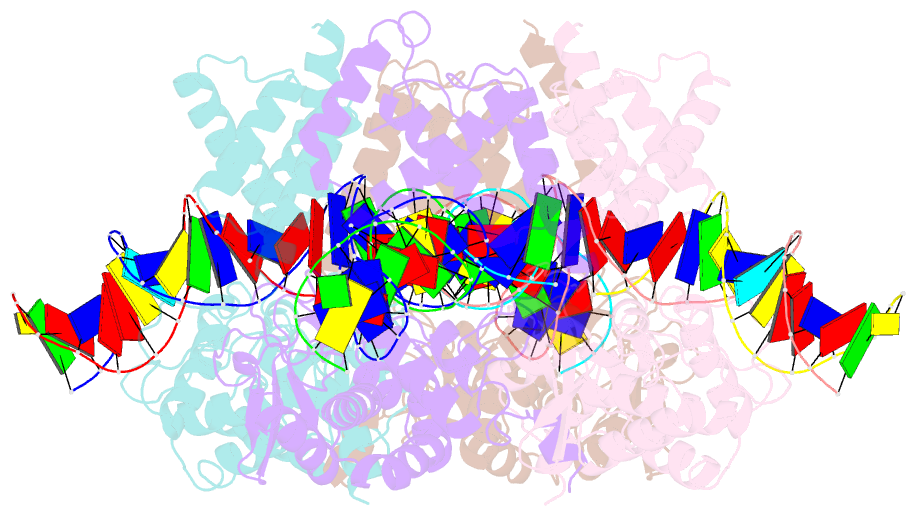
- Crystal structure of a wild-type cre recombinase-loxp synapse: phosphotyrosine covalent intermediate
- Reference
- Ennifar E, Meyer JEW, Buchholz F, Stewart AF, Suck D (2003): "Crystal structure of a wild-type Cre recombinase-loxP synapse reveals a novel spacer conformation suggesting an alternative mechanism for DNA cleavage activation." Nucleic Acids Res., 31, 5449-5460. doi: 10.1093/nar/gkg732.
- Abstract
- Escherichia coli phage P1 Cre recombinase catalyzes the site-specific recombination of DNA containing loxP sites. We report here two crystal structures of a wild-type Cre recombinase-loxP synaptic complex corresponding to two distinct reaction states: an initial pre-cleavage complex, trapped using a phosphorothioate modification at the cleavable scissile bond that prevents the recombination reaction, and a 3'-phosphotyrosine protein-DNA intermediate resulting from the first strand cleavage. In contrast to previously determined Cre complexes, both structures contain a full tetrameric complex in the asymmetric unit, unequivocally showing that the anti-parallel arrangement of the loxP sites is an intrinsic property of the Cre-loxP recombination synapse. The conformation of the spacer is different to the one observed for the symmetrized loxS site: a kink next to the scissile phosphate in the top strand of the pre-cleavage complex leads to unstacking of the TpG step and a widening of the minor groove. This side of the spacer is interacting with a 'cleavage-competent' Cre subunit, suggesting that the first cleavage occurs at the ApT step in the top strand. This is further confirmed by the structure of the 3'-phosphotyrosine intermediate, where the DNA is cleaved in the top strands and covalently linked to the 'cleavage-competent' subunits. The cleavage is followed by a movement of the C-terminal part containing the attacking Y324 and the helix N interacting with the 'non-cleaving' subunit. This rearrangement could be responsible for the interconversion of Cre subunits. Our results also suggest that the Cre-induced kink next to the scissile phosphodiester activates the DNA for cleavage at this position and facilitates strand transfer.