Summary information and primary citation
- PDB-id
- 1vfg; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transferase-RNA
- Method
- X-ray (2.8 Å)
- Summary
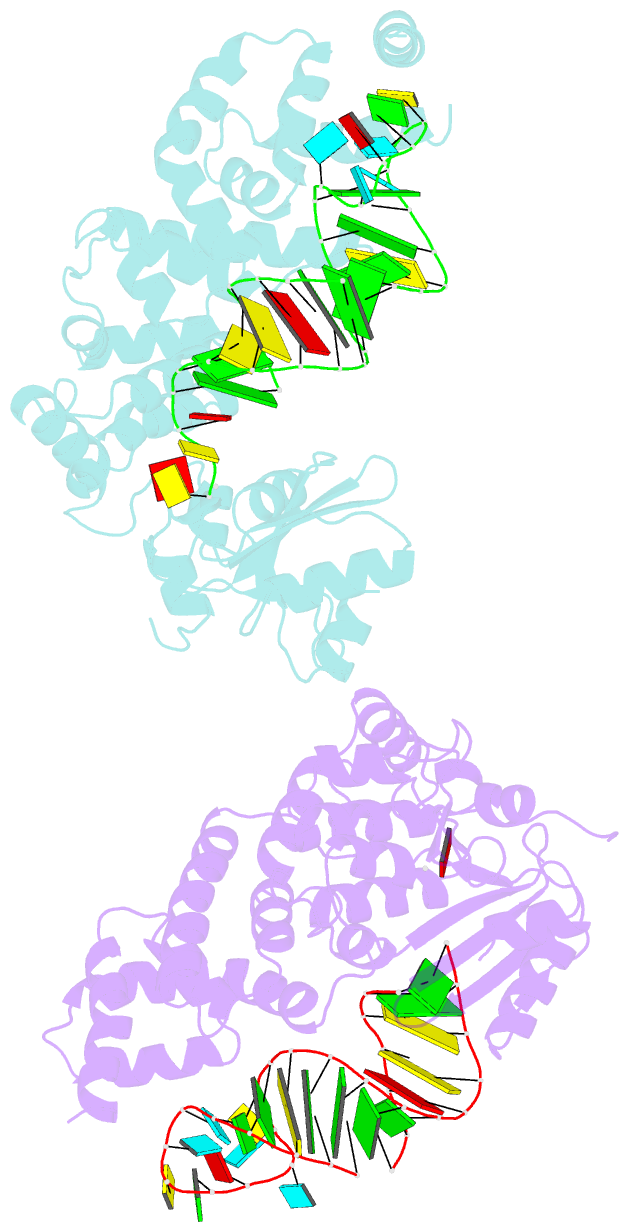
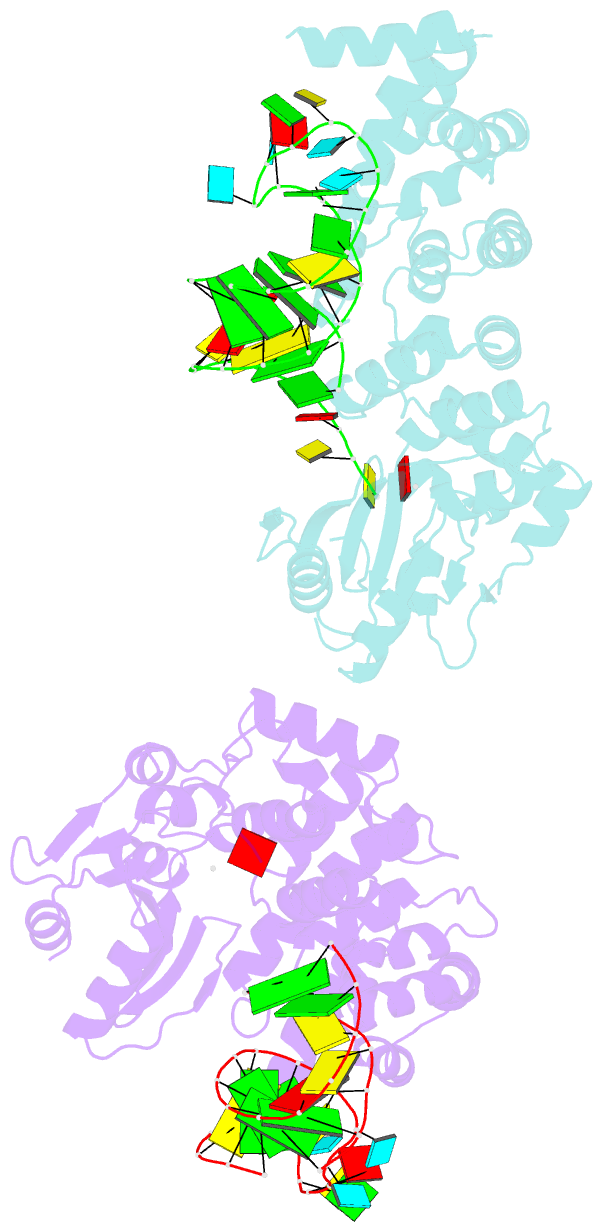
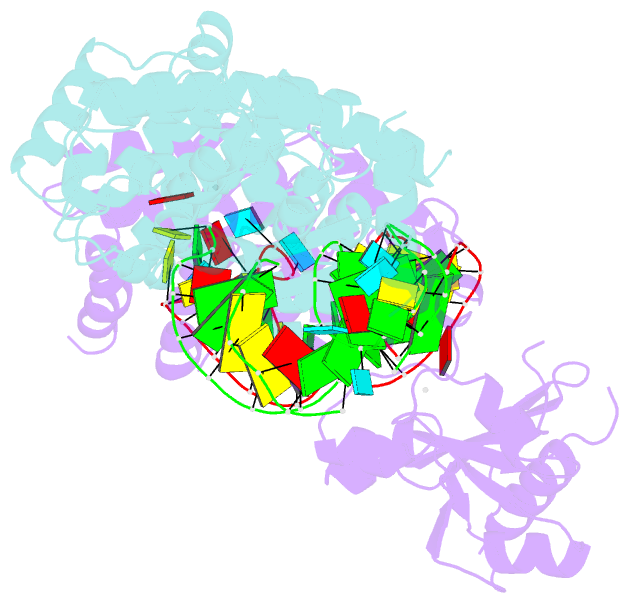
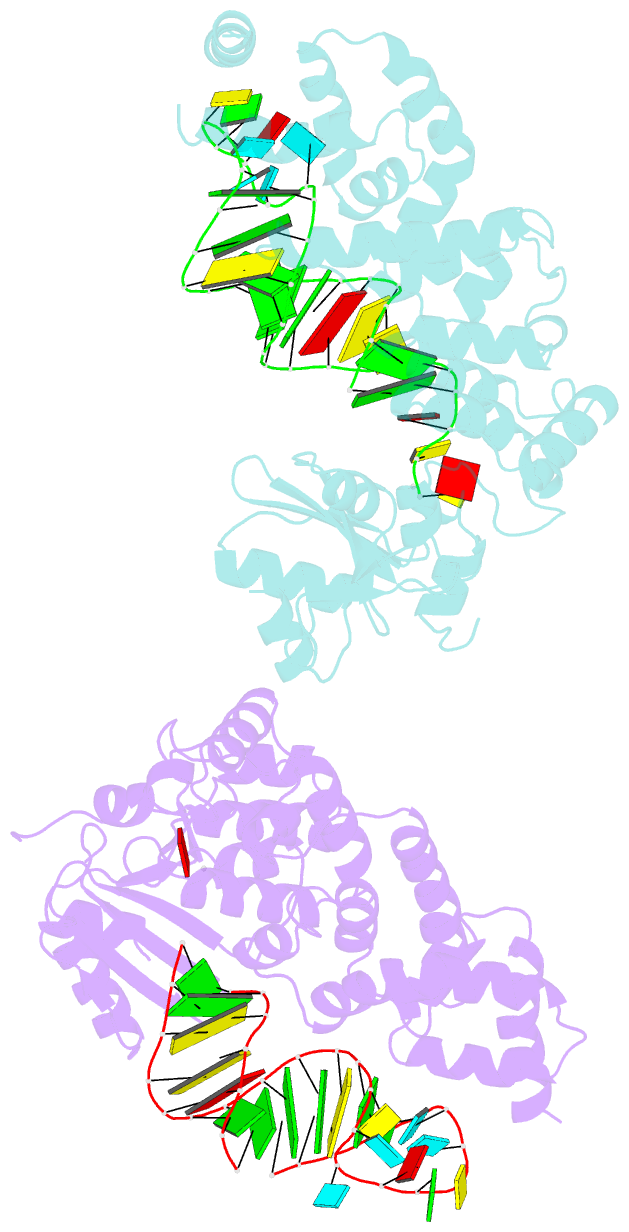
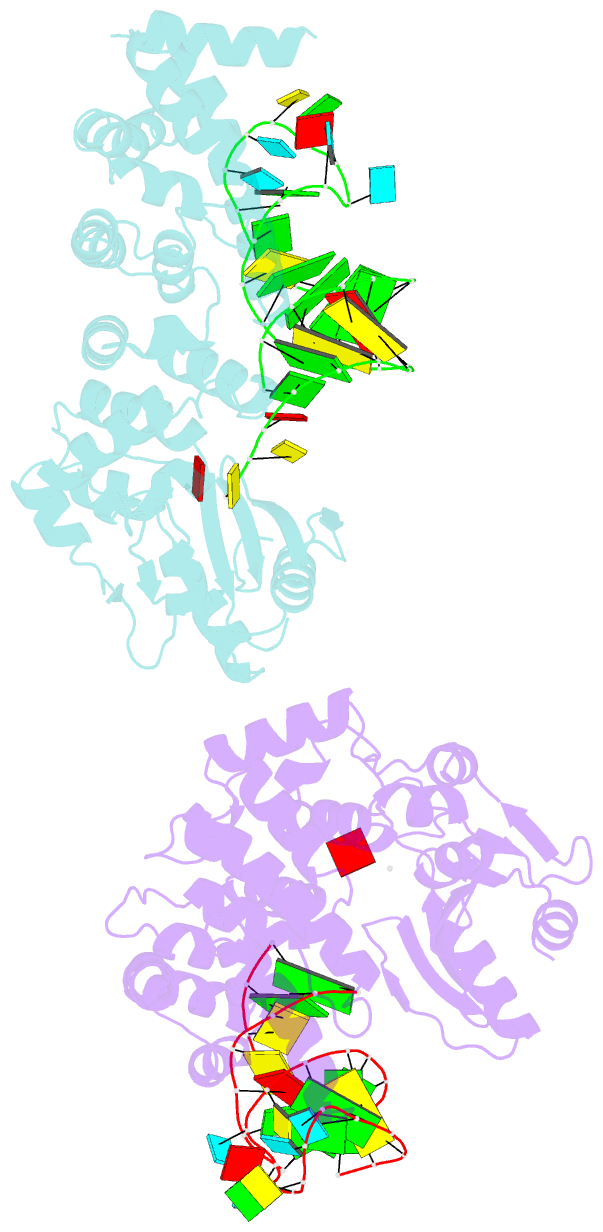
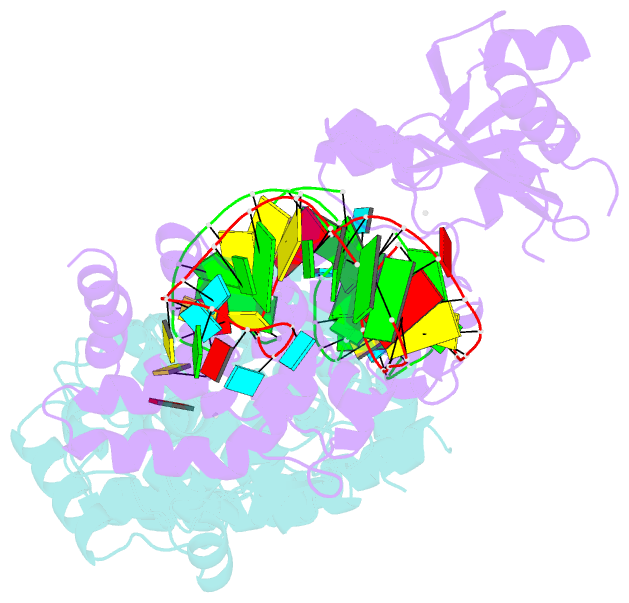
- Crystal structure of trna nucleotidyltransferase complexed with a primer trna and an incoming atp analog
- Reference
- Tomita K, Fukai S, Ishitani R, Ueda T, Takeuchi N, Vassylyev DG, Nureki O (2004): "Structural basis for template-independent RNA polymerization." Nature, 430, 700-704. doi: 10.1038/nature02712.
- Abstract
- The 3'-terminal CCA nucleotide sequence (positions 74-76) of transfer RNA is essential for amino acid attachment and interaction with the ribosome during protein synthesis. The CCA sequence is synthesized de novo and/or repaired by a template-independent RNA polymerase, 'CCA-adding enzyme', using CTP and ATP as substrates. Despite structural and biochemical studies, the mechanism by which the CCA-adding enzyme synthesizes the defined sequence without a nucleic acid template remains elusive. Here we present the crystal structure of Aquifex aeolicus CCA-adding enzyme, bound to a primer tRNA lacking the terminal adenosine and an incoming ATP analogue, at 2.8 A resolution. The enzyme enfolds the acceptor T helix of the tRNA molecule. In the catalytic pocket, C75 is adjacent to ATP, and their base moieties are stacked. The complementary pocket for recognizing C74-C75 of tRNA forms a 'protein template' for the penultimate two nucleotides, mimicking the nucleotide template used by template-dependent polymerases. These results are supported by systematic analyses of mutants. Our structure represents the 'pre-insertion' stage of selecting the incoming nucleotide and provides the structural basis for the mechanism underlying template-independent RNA polymerization.