Summary information and primary citation
- PDB-id
- 1z63; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- hydrolase-DNA complex
- Method
- X-ray (3.0 Å)
- Summary
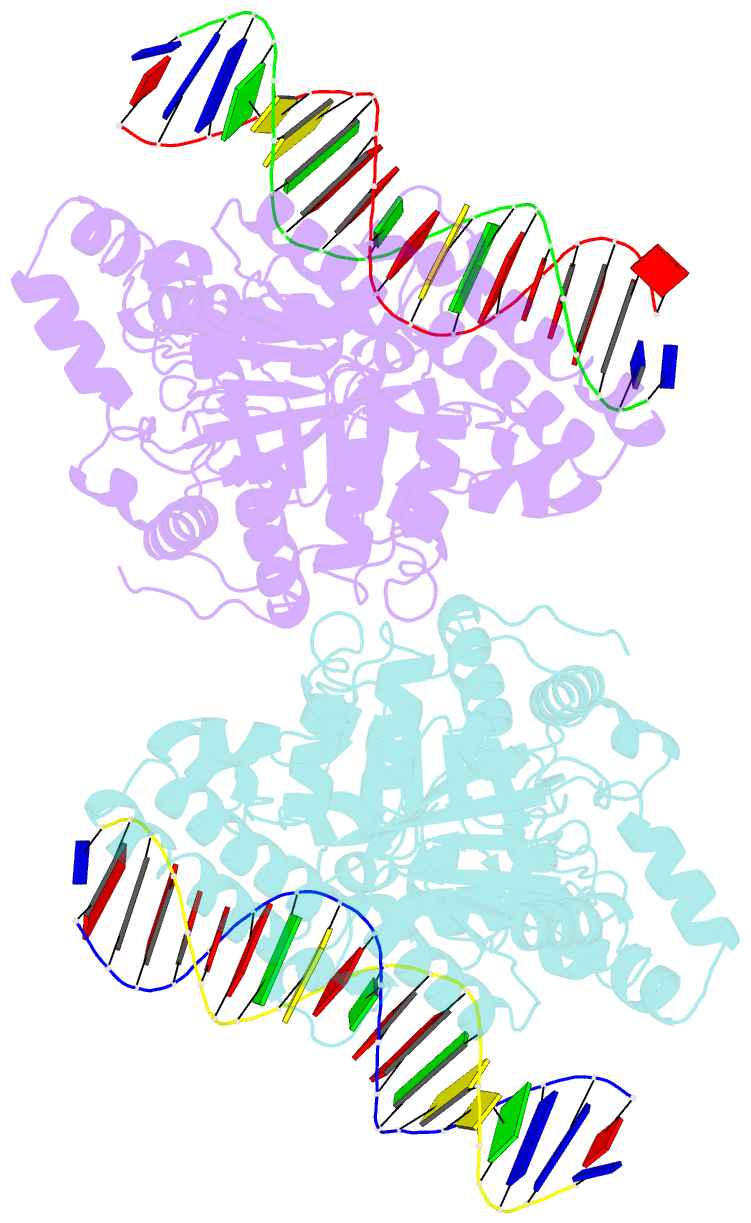
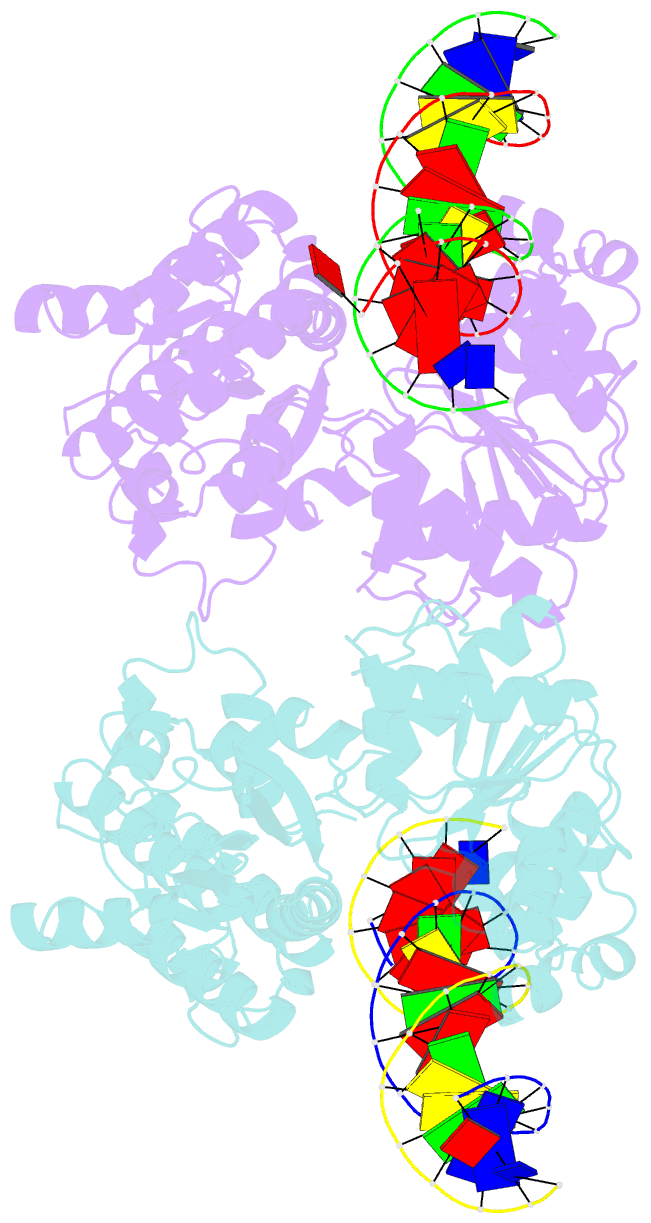
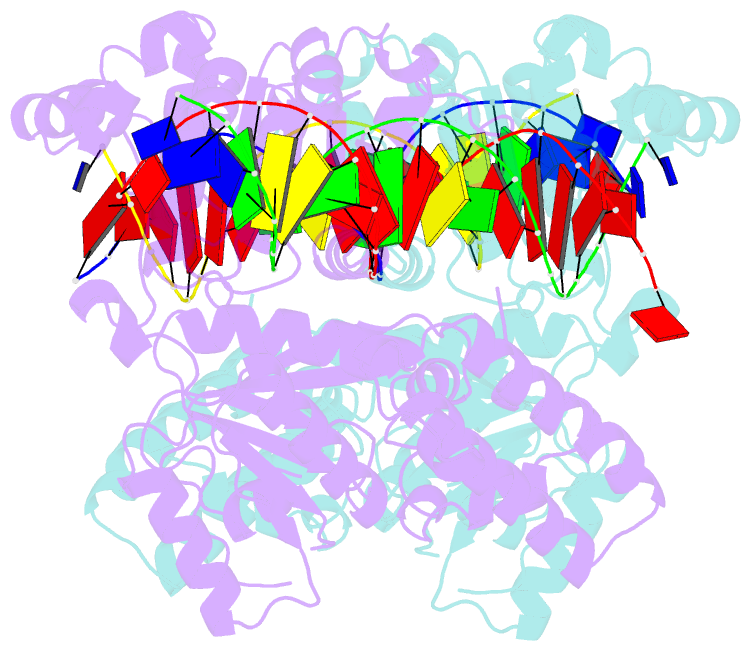
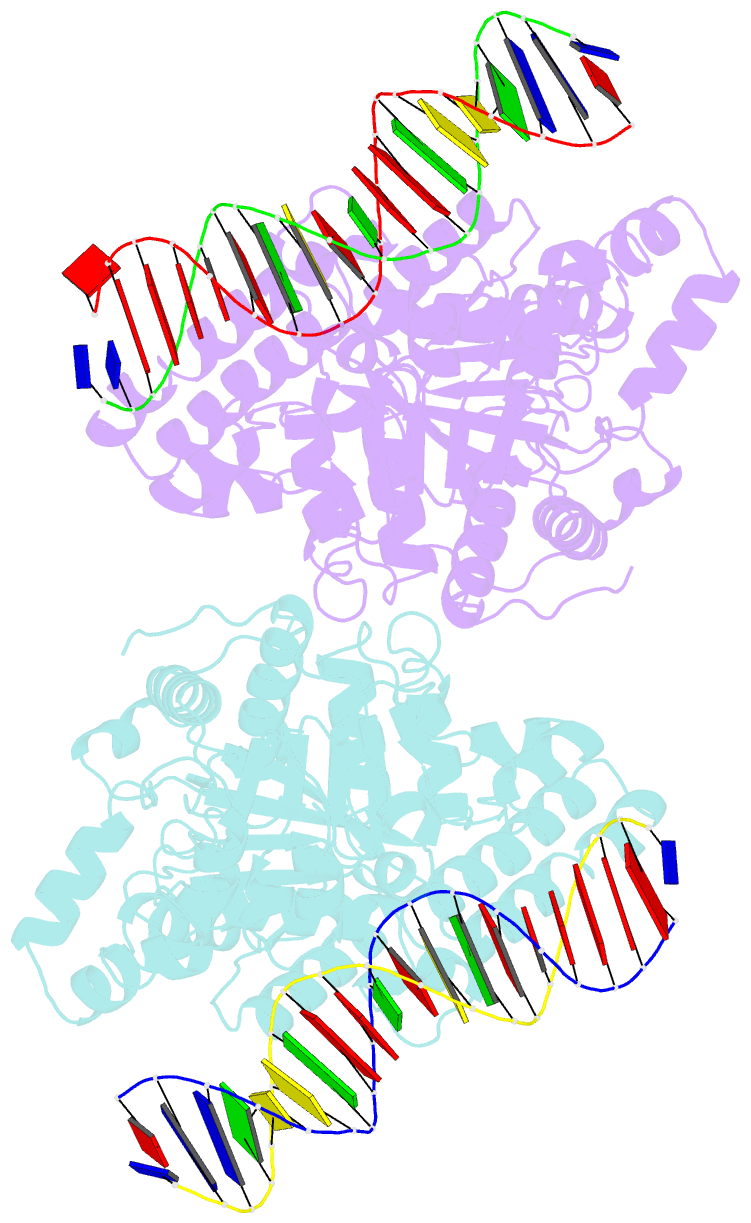
- Sulfolobus solfataricus swi2-snf2 atpase core in complex with dsDNA
- Reference
- Durr H, Korner C, Muller M, Hickmann V, Hopfner KP (2005): "X-ray structures of the Sulfolobus solfataricus SWI2/SNF2 ATPase core and its complex with DNA." Cell(Cambridge,Mass.), 121, 363-373. doi: 10.1016/j.cell.2005.03.026.
- Abstract
- SWI2/SNF2 ATPases remodel chromatin or other DNA:protein complexes by a poorly understood mechanism that involves ATP-dependent DNA translocation and generation of superhelical torsion. Crystal structures of a dsDNA-translocating SWI2/SNF2 ATPase core from Sulfolobus solfataricus reveal two helical SWI2/SNF2 specific subdomains, fused to a DExx box helicase-related ATPase core. Fully base paired duplex DNA binds along a central cleft via both minor groove strands, indicating that SWI2/SNF2 ATPases travel along the dsDNA minor groove without strand separation. A structural switch, linking DNA binding and the active site DExx motif, may account for the stimulation of ATPase activity by dsDNA. Our results suggest that torque in remodeling processes is generated by an ATP-driven screw motion of DNA along the active site cleft. The structures also redefine SWI2/SNF2 functional motifs and uncover unexpected structural correlation of mutations in Cockayne and X-linked mental retardation syndromes.