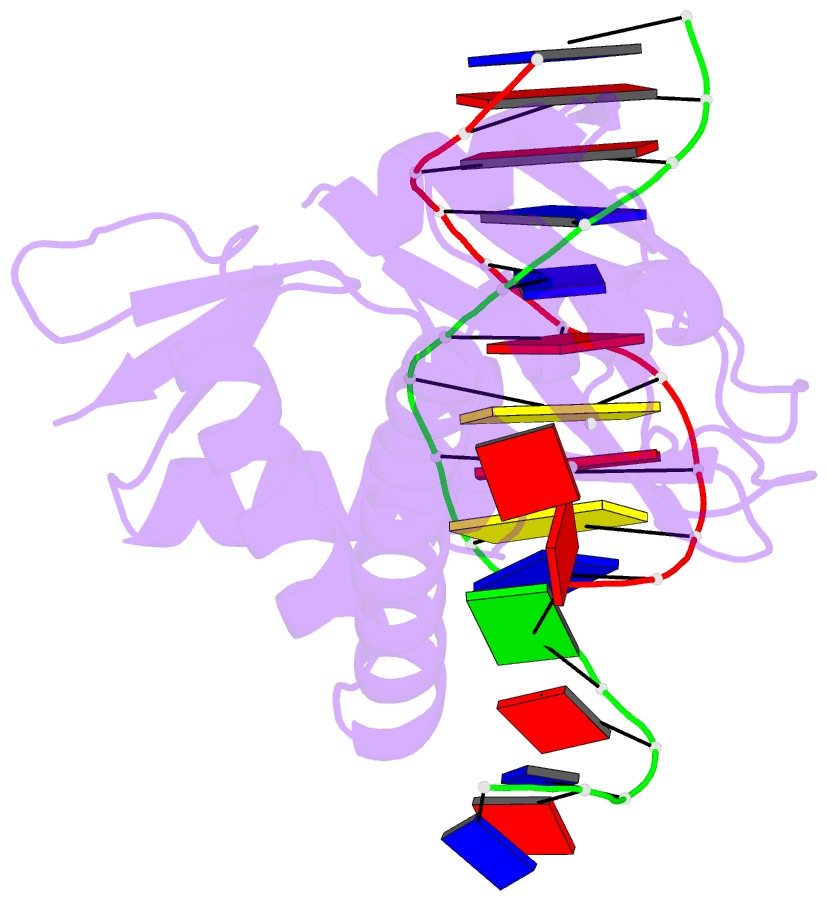
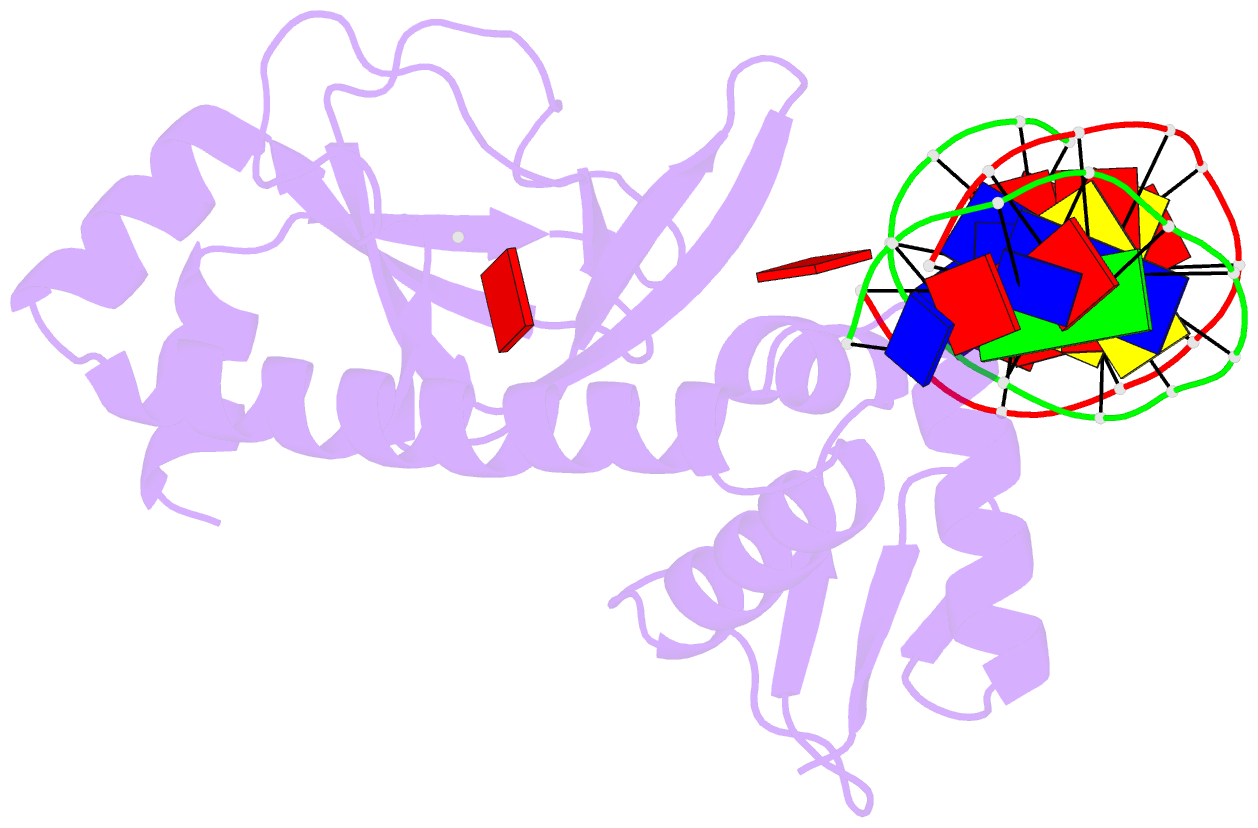
Summary information and primary citation
- PDB-id
- 2cgp; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transcription-DNA
- Method
- X-ray (2.2 Å)
- Summary
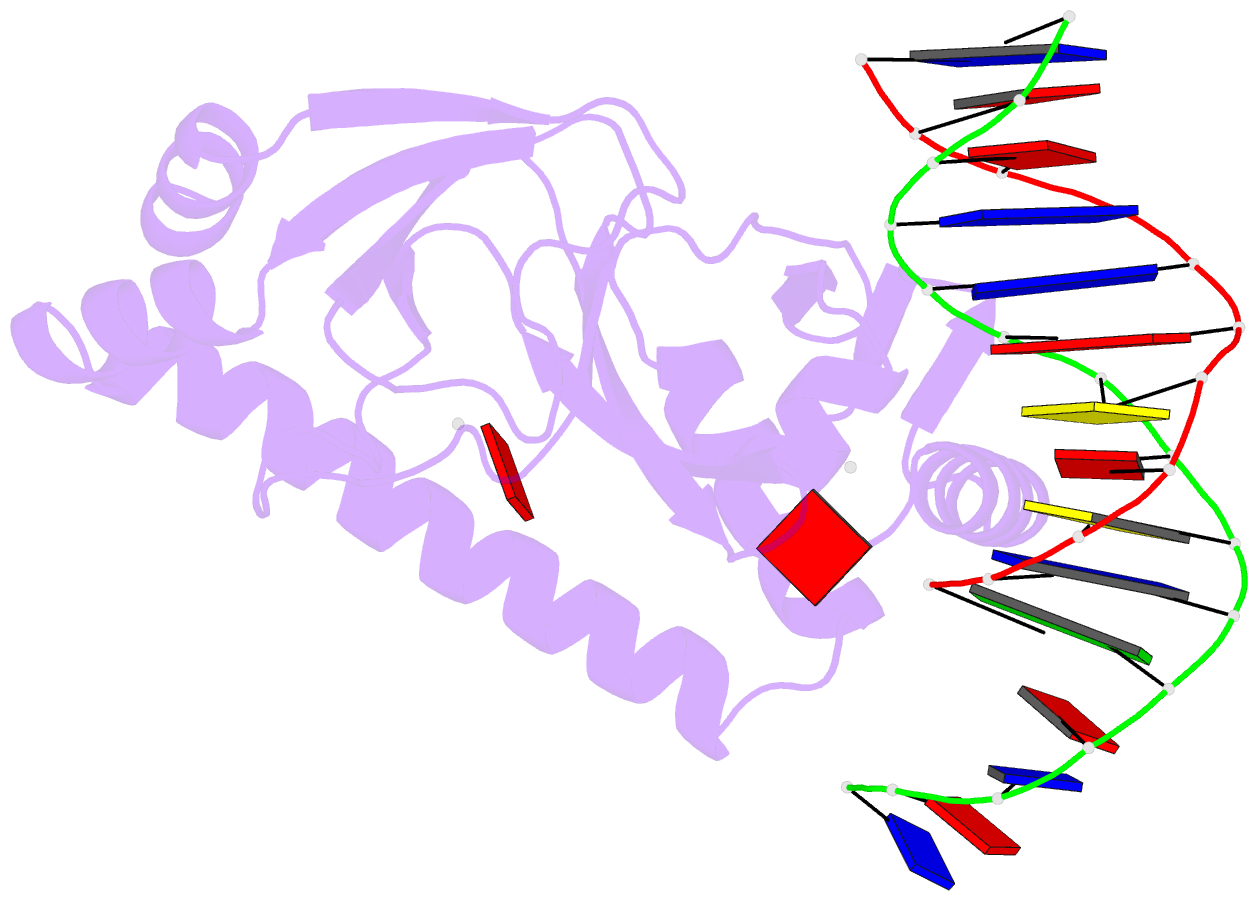
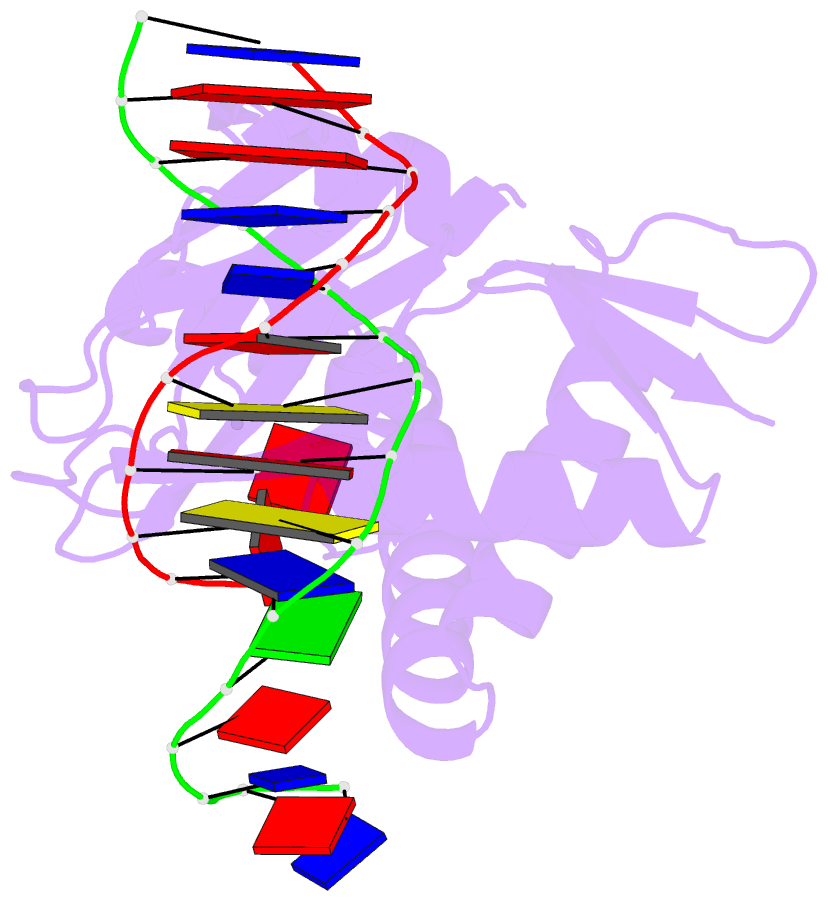
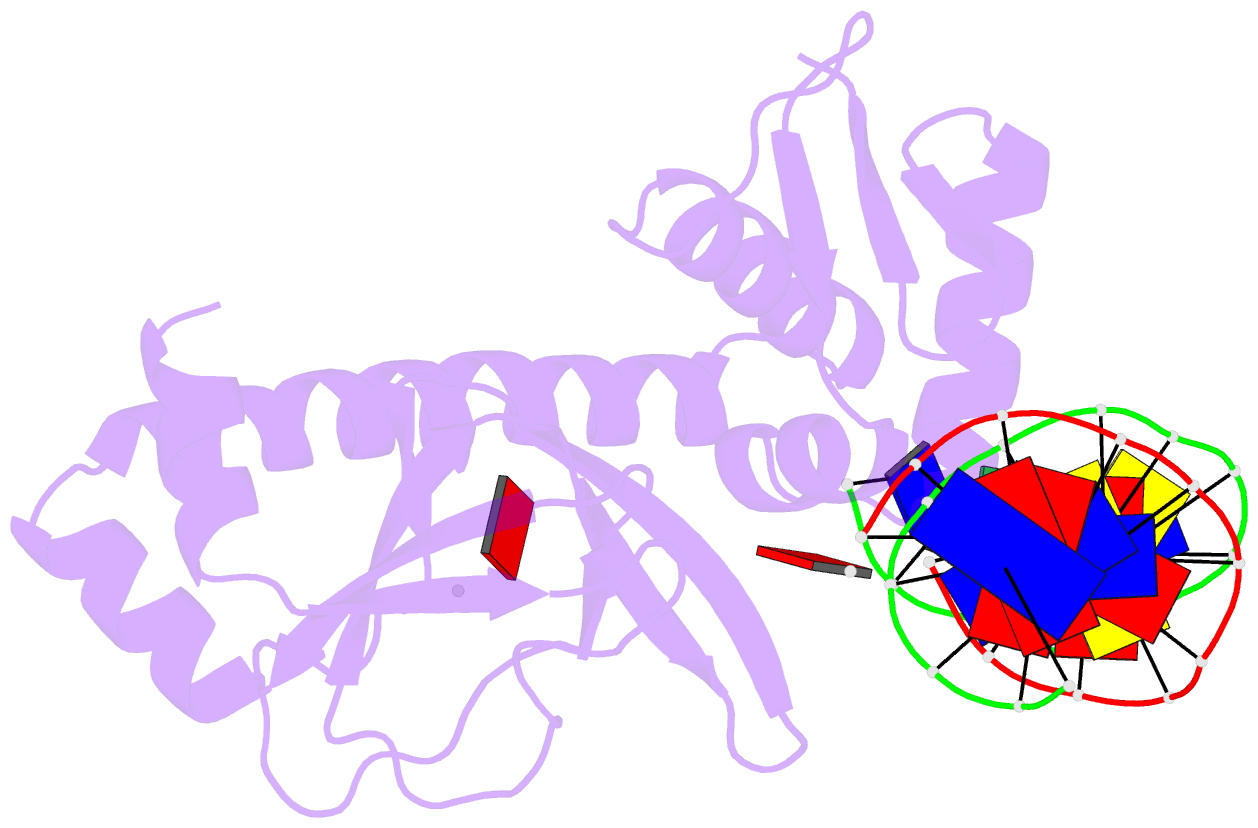
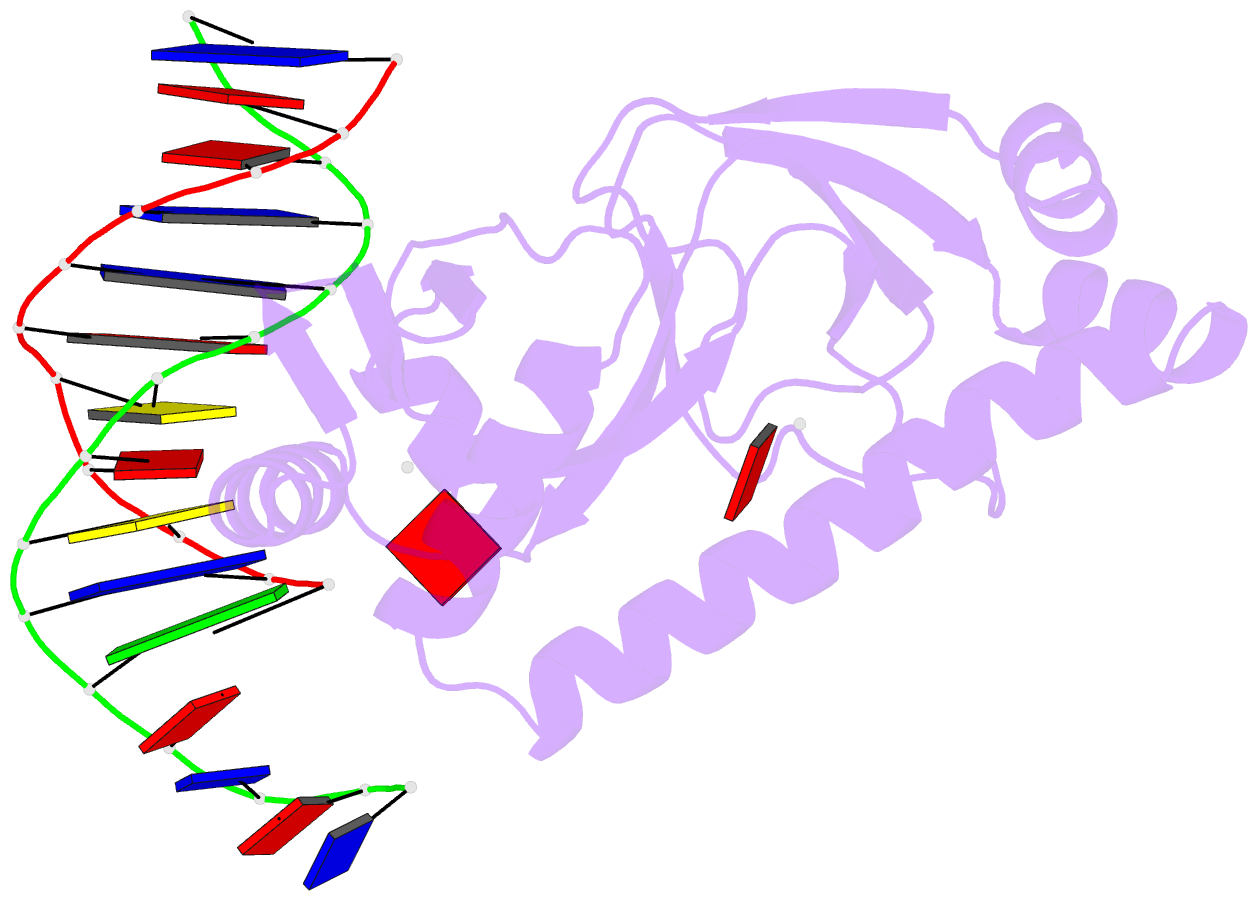
- Catabolite gene activator protein-DNA complex, adenosine-3',5'-cyclic-monophosphate
- Reference
- Passner JM, Steitz TA (1997): "The structure of a CAP-DNA complex having two cAMP molecules bound to each monomer." Proc.Natl.Acad.Sci.USA, 94, 2843-2847. doi: 10.1073/pnas.94.7.2843.
- Abstract
- The 2.2 A resolution crystal structure of the Escherichia coli catabolite gene activator protein (CAP) complexed with cAMP and a 46-bp DNA fragment reveals a second cAMP molecule bound to each protein monomer. The second cAMP is in the syn conformation and is located on the DNA binding domain interacting with the helix-turn-helix, a beta-hairpin from the regulatory domain and the DNA (via water molecules). The presence of this second cAMP site resolves the apparent discrepancy between the NMR and x-ray data on the conformation of cAMP, and explains the cAMP concentration-dependent behaviors of the protein. In addition, this site's close proximity to mutations affecting transcriptional activation and its water-mediated interactions with a DNA recognition residue (E181) and DNA raise the possibility that this site has biological relevance.