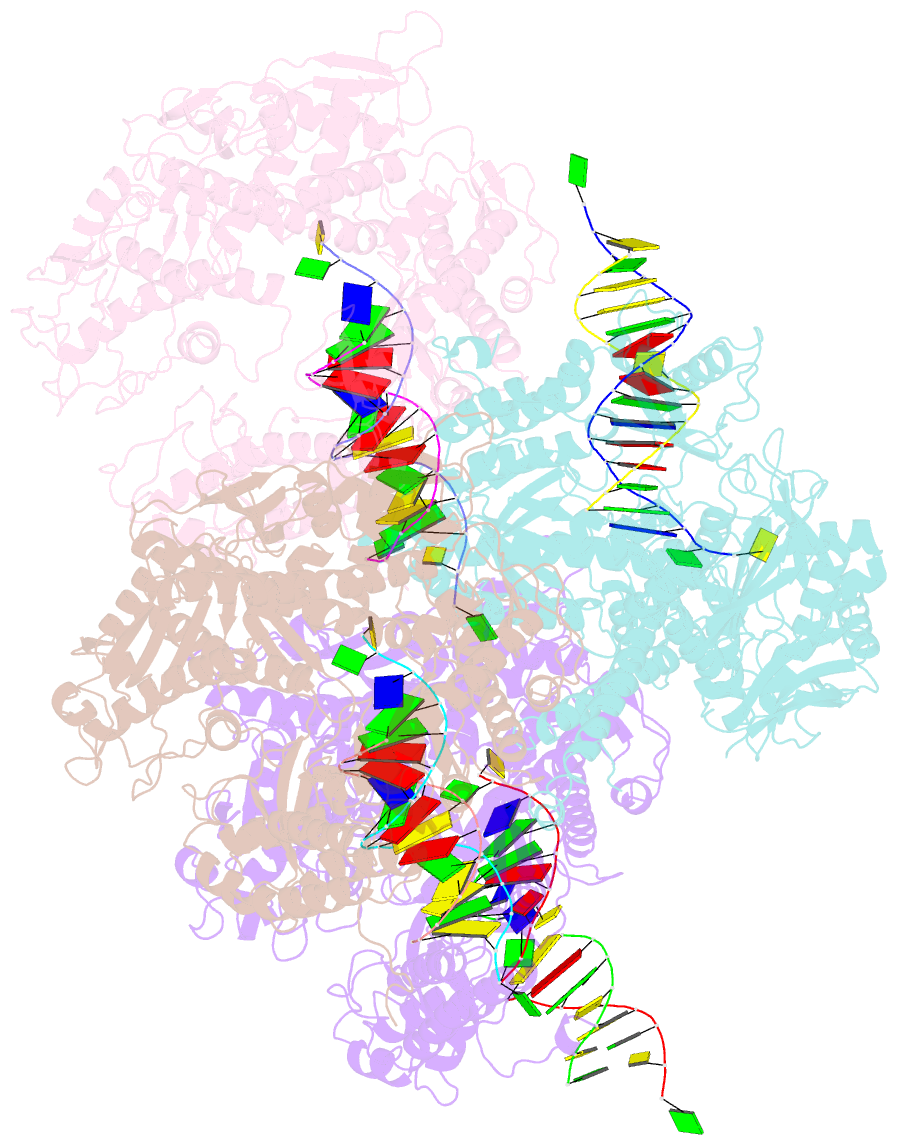
Summary information and primary citation
- PDB-id
- 2dy4; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transferase-DNA
- Method
- X-ray (2.65 Å)
- Summary
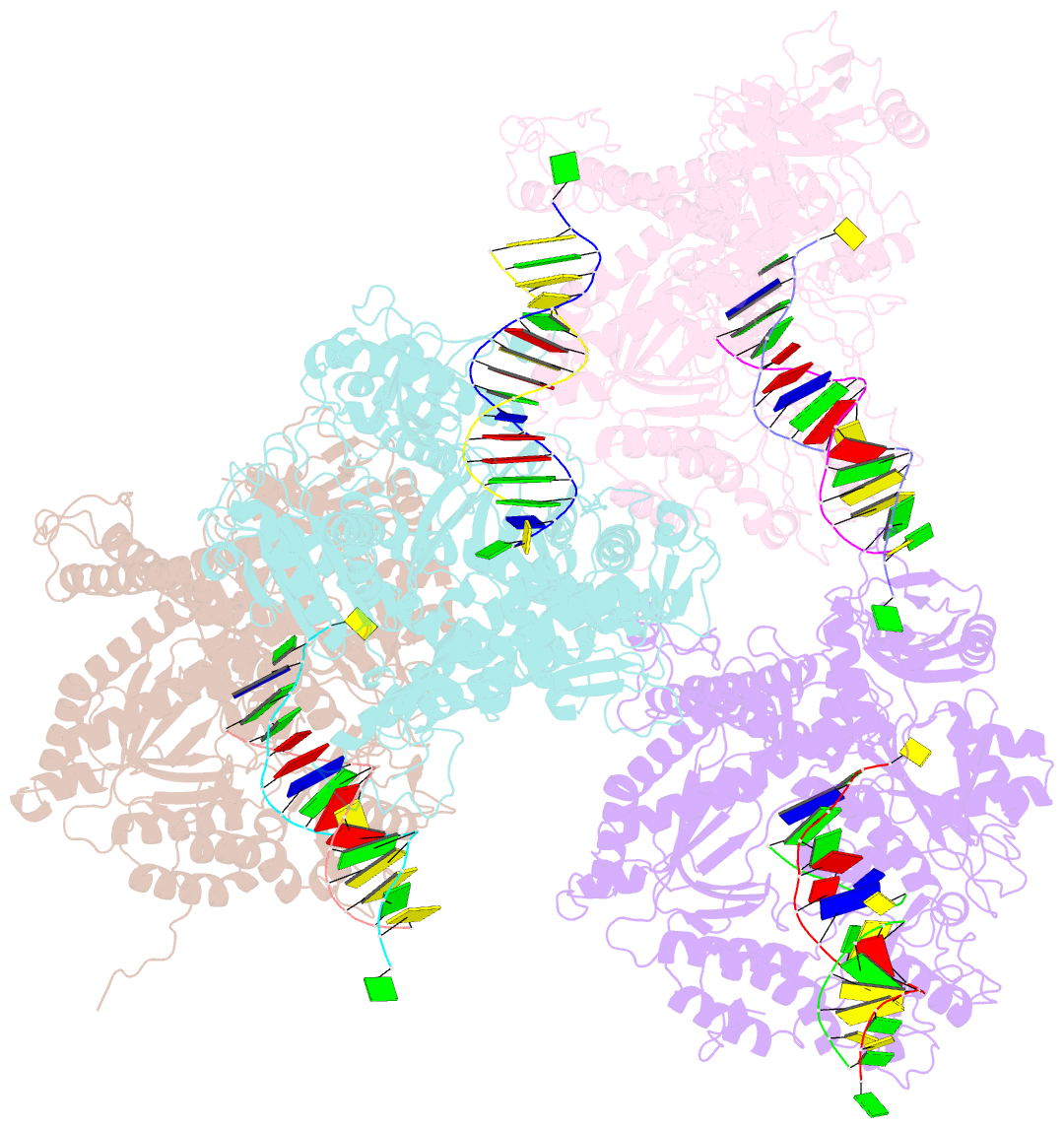
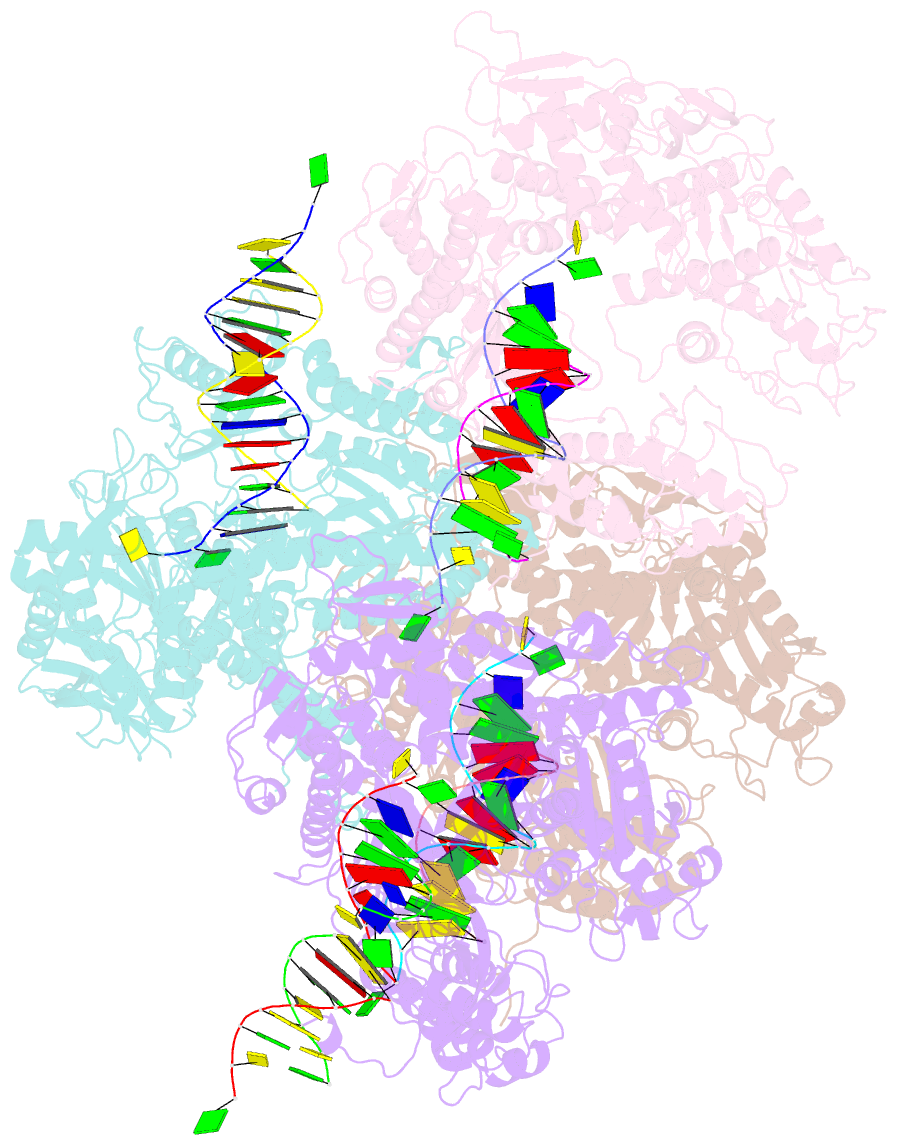
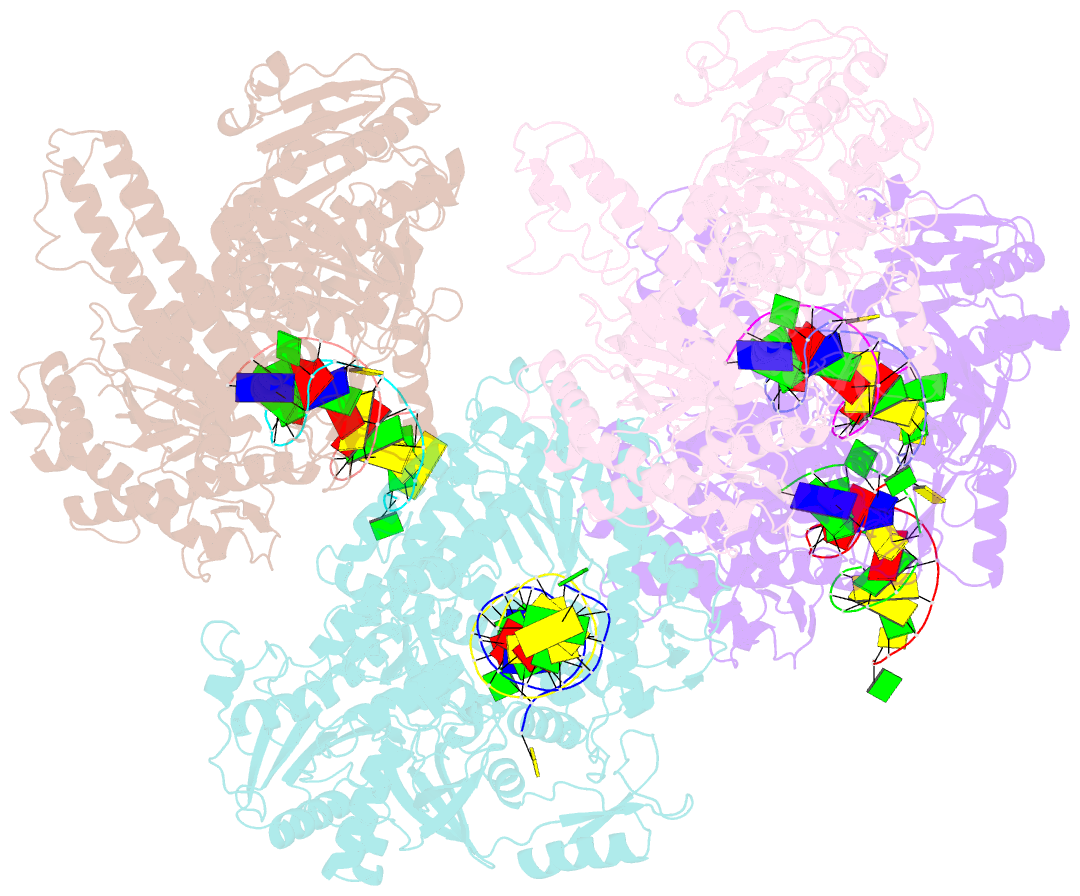
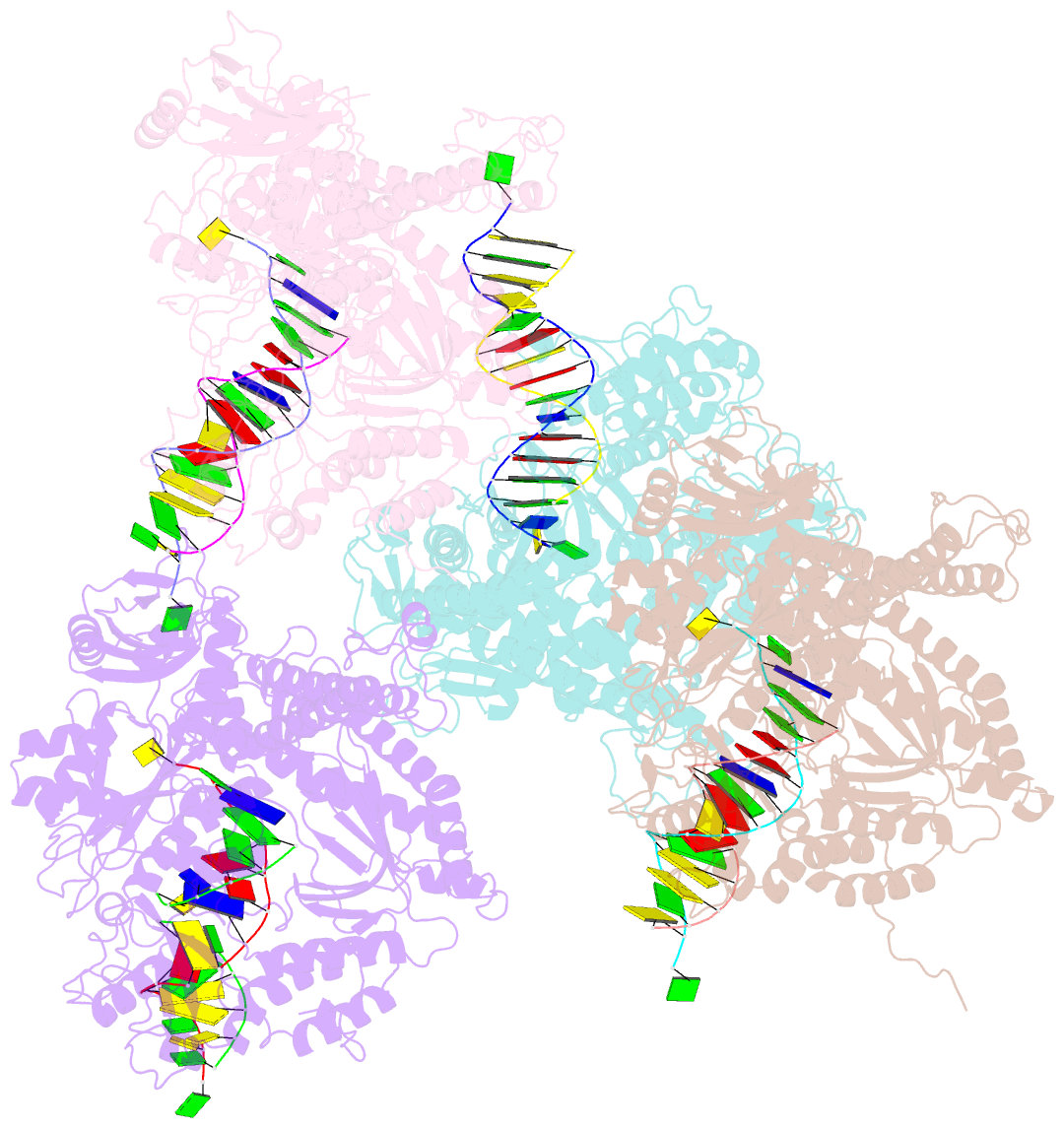
- Crystal structure of rb69 gp43 in complex with DNA containing thymine glycol
- Reference
- Aller P, Rould MA, Hogg M, Wallace SS, Doublie S (2007): "A structural rationale for stalling of a replicative DNA polymerase at the most common oxidative thymine lesion, thymine glycol." Proc.Natl.Acad.Sci.USA, 104, 814-818. doi: 10.1073/pnas.0606648104.
- Abstract
- Thymine glycol (Tg) is a common product of oxidation and ionizing radiation, including that used for cancer treatment. Although Tg is a poor mutagenic lesion, it has been shown to present a strong block to both repair and replicative DNA polymerases. The 2.65-A crystal structure of a binary complex of the replicative RB69 DNA polymerase with DNA shows that the templating Tg is intrahelical and forms a regular Watson-Crick base pair with the incorporated A. The C5 methyl group protrudes axially from the ring of the damaged pyrimidine and hinders stacking of the adjacent 5' template guanine. The position of the displaced 5' template guanine is such that the next incoming nucleotide cannot be incorporated into the growing primer strand, and it explains why primer extension past the lesion is prohibited even though DNA polymerases can readily incorporate an A across from the Tg lesion.