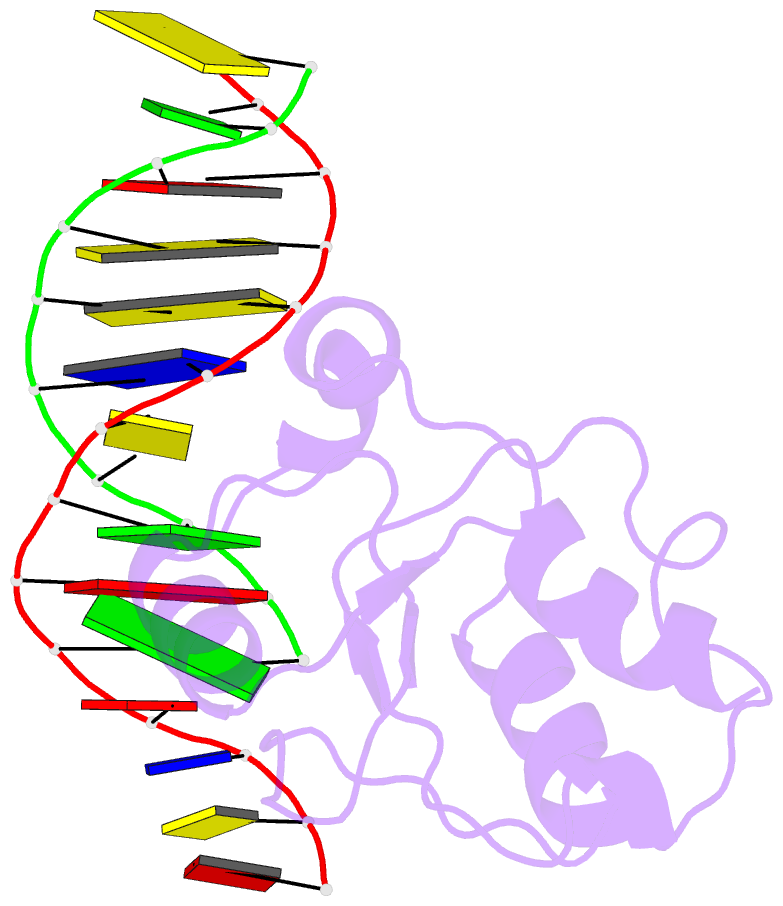
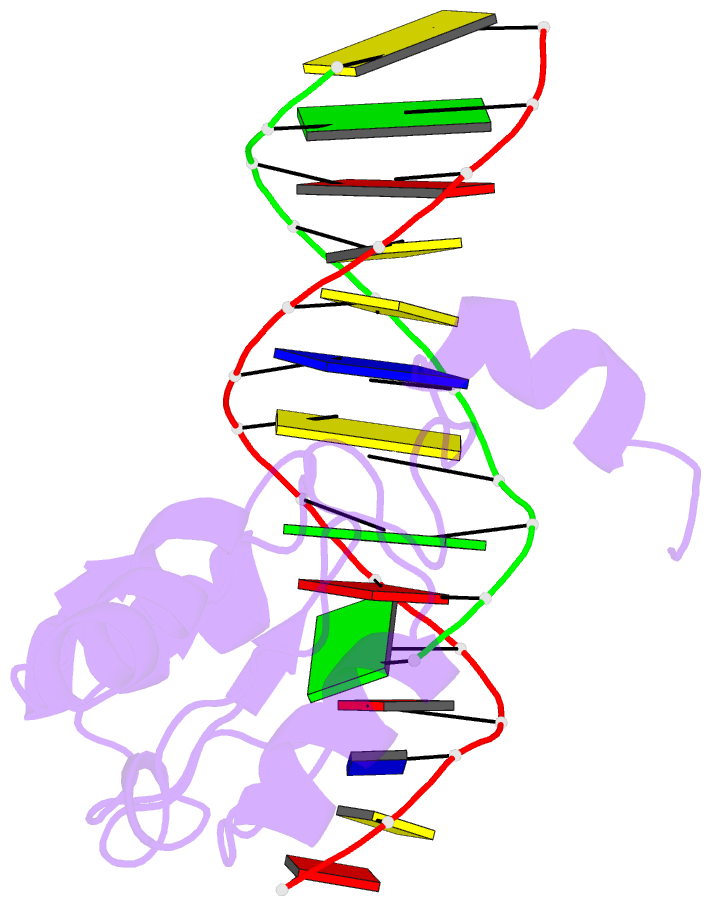
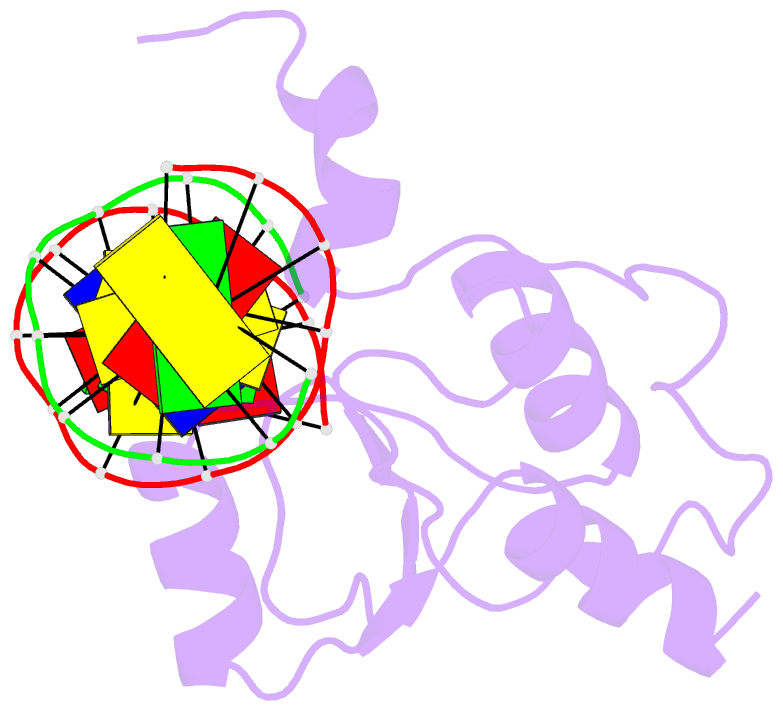
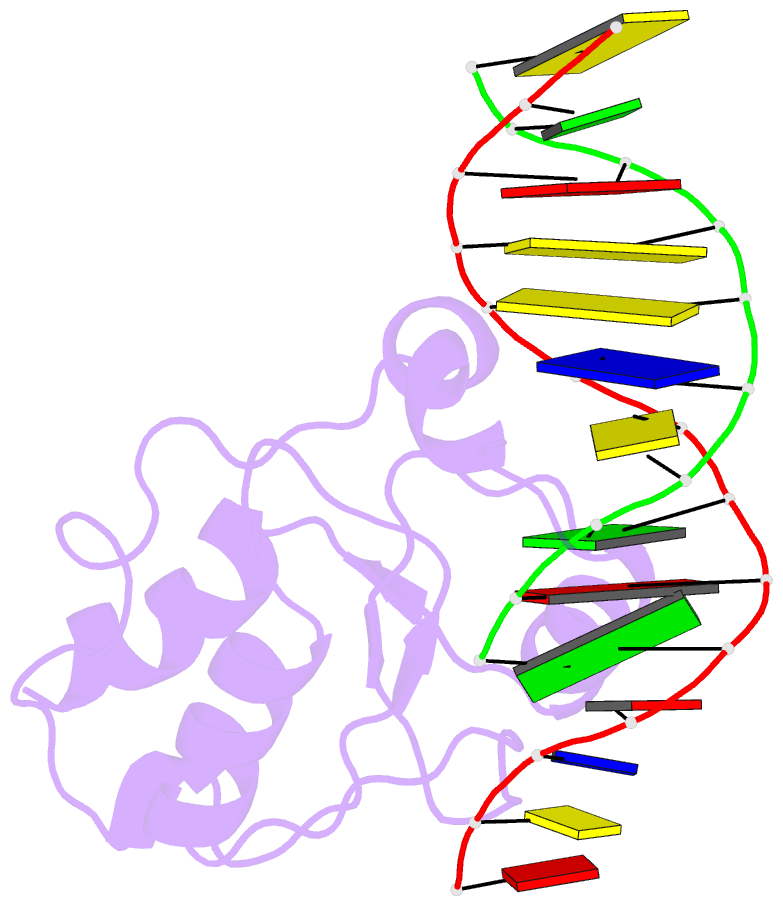
Summary information and primary citation
- PDB-id
- 2k7f; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- replication-DNA
- Method
- NMR
- Summary
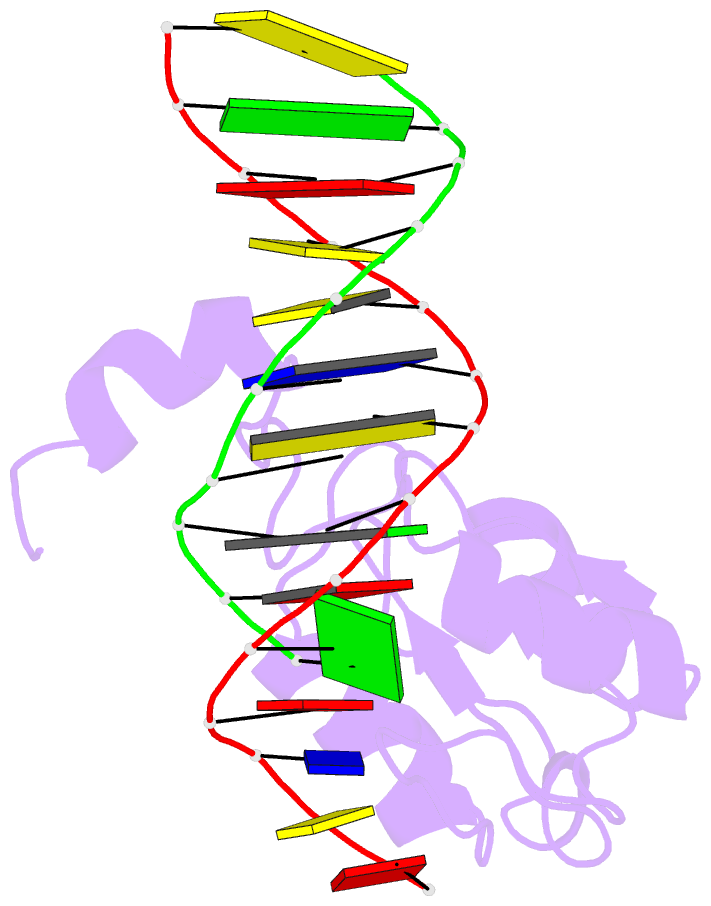
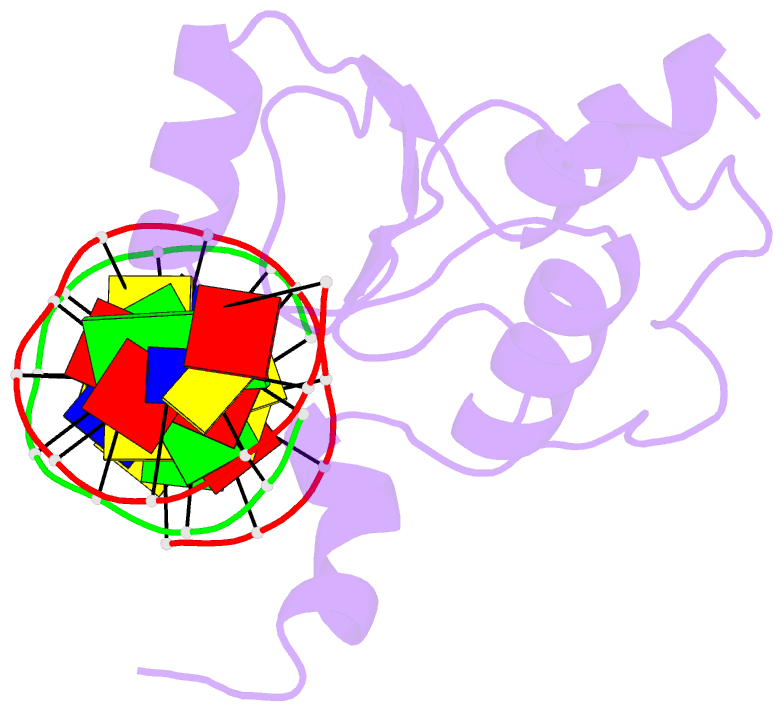
- Haddock calculated model of the complex between the brct region of rfc p140 and dsDNA
- Reference
- Kobayashi M, Ab E, Bonvin AM, Siegal G (2010): "Structure of the DNA-bound BRCA1 C-terminal region from human replication factor C p140 and model of the protein-DNA complex." J.Biol.Chem., 285, 10087-10097. doi: 10.1074/jbc.M109.054106.
- Abstract
- BRCA1 C-terminal domain (BRCT)-containing proteins are found widely throughout the animal and bacteria kingdoms where they are exclusively involved in cell cycle regulation and DNA metabolism. Whereas most BRCT domains are involved in protein-protein interactions, a small subset has bona fide DNA binding activity. Here, we present the solution structure of the BRCT region of the large subunit of replication factor C bound to DNA and a model of the structure-specific complex with 5'-phosphorylated double-stranded DNA. The replication factor C BRCT domain possesses a large basic patch on one face, which includes residues that are structurally conserved and ligate the phosphate in phosphopeptide binding BRCT domains. An extra alpha-helix at the N terminus, which is required for DNA binding, inserts into the major groove and makes extensive contacts to the DNA backbone. The model of the protein-DNA complex suggests 5'-phosphate recognition by the BRCT domains of bacterial NAD(+)-dependent ligases and a nonclamp loading role for the replication factor C complex in DNA transactions.