Summary information and primary citation
- PDB-id
- 2li8; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transcription-RNA
- Method
- NMR
- Summary
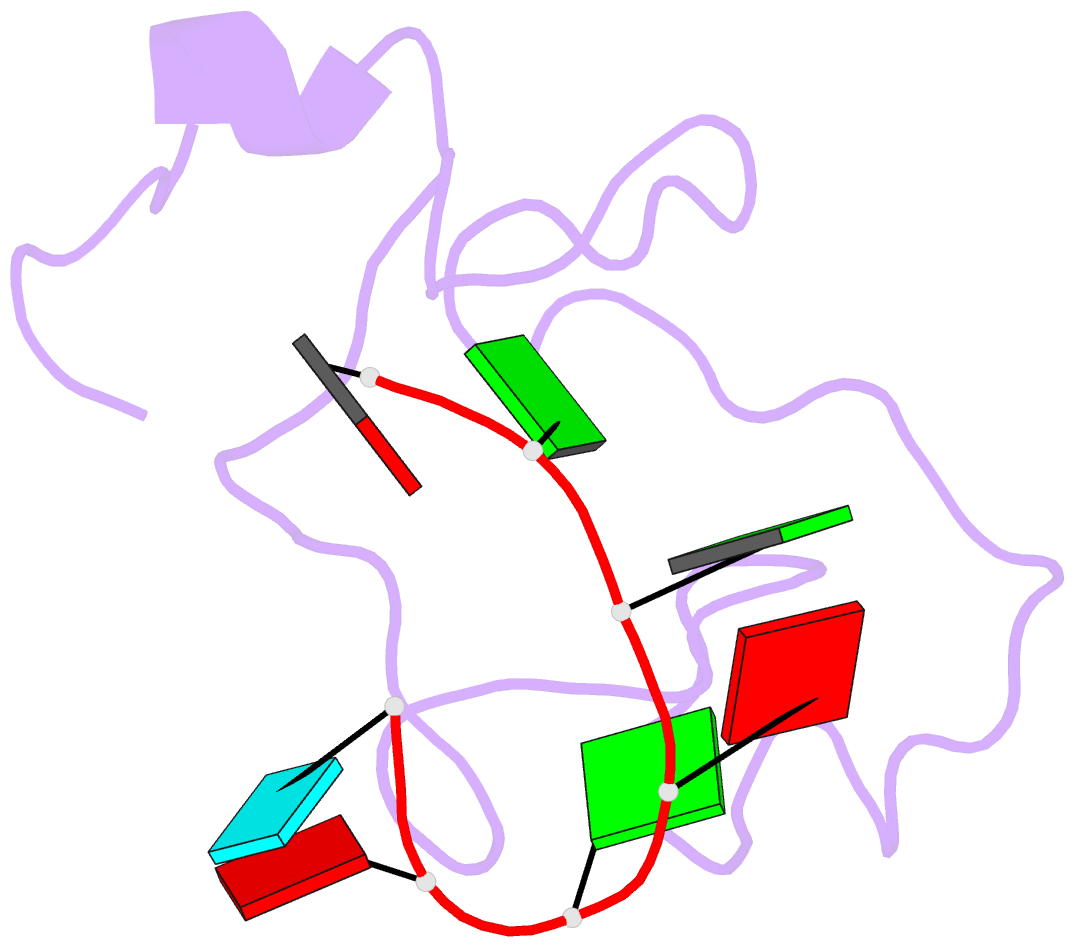
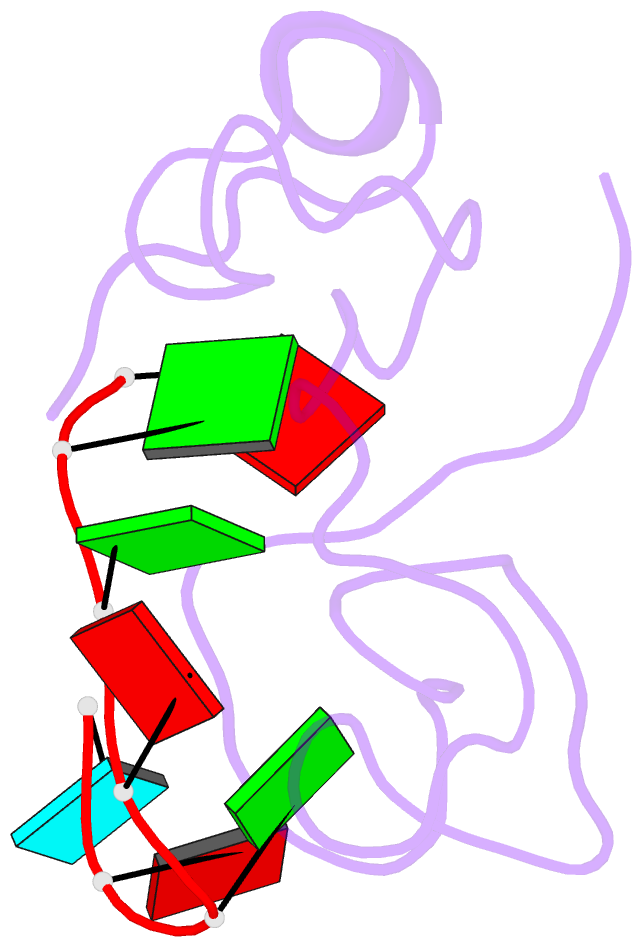
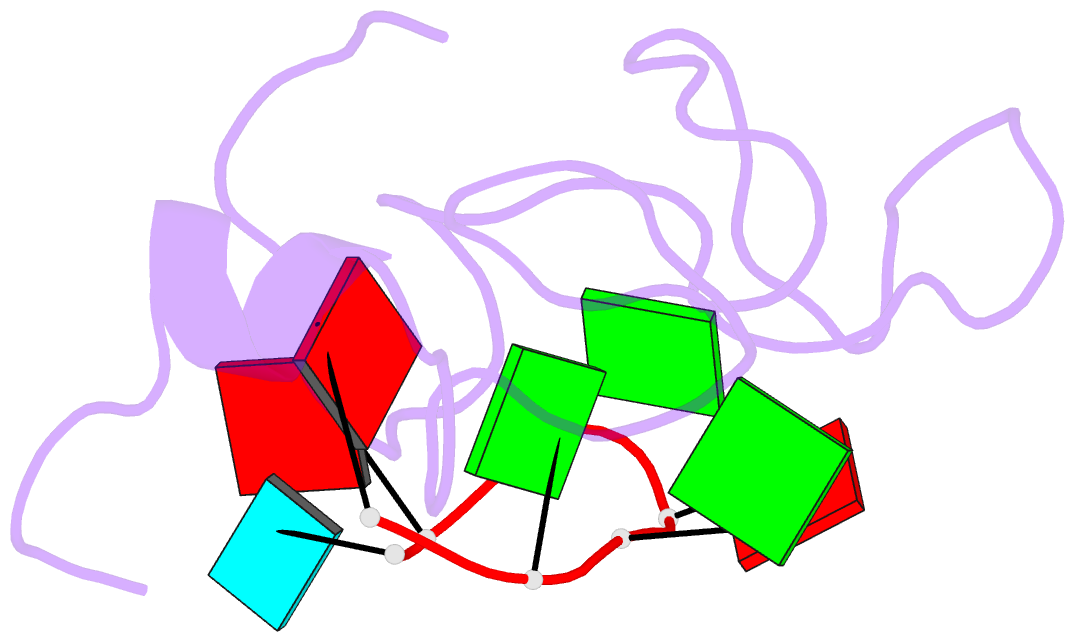
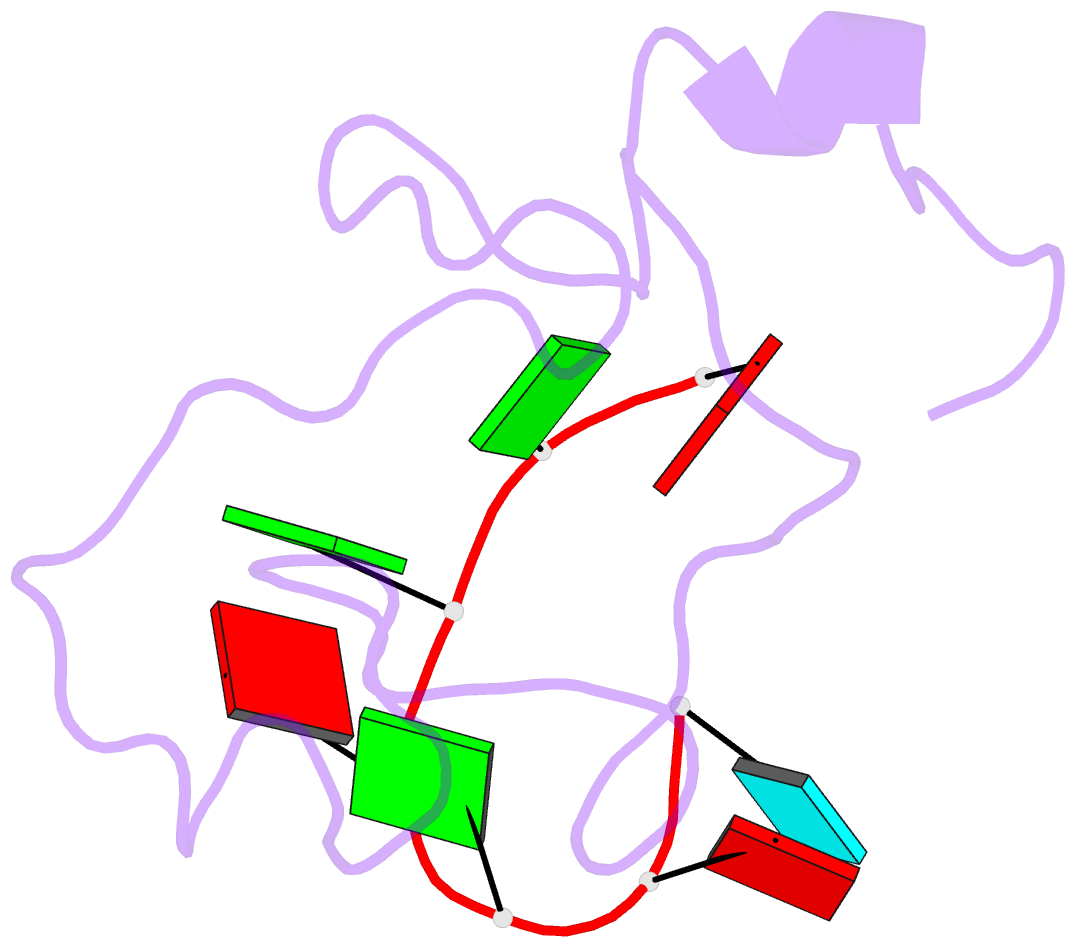
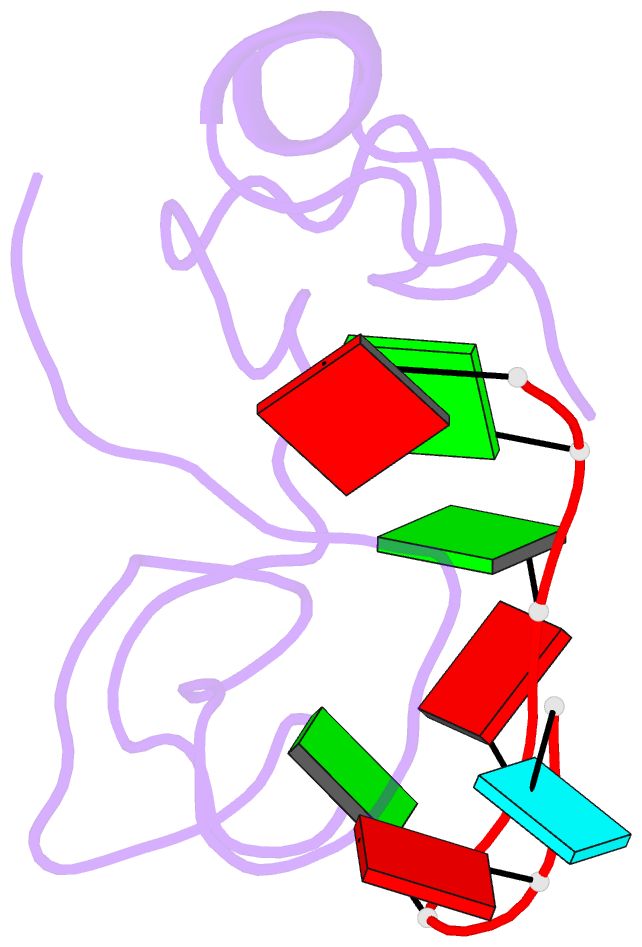
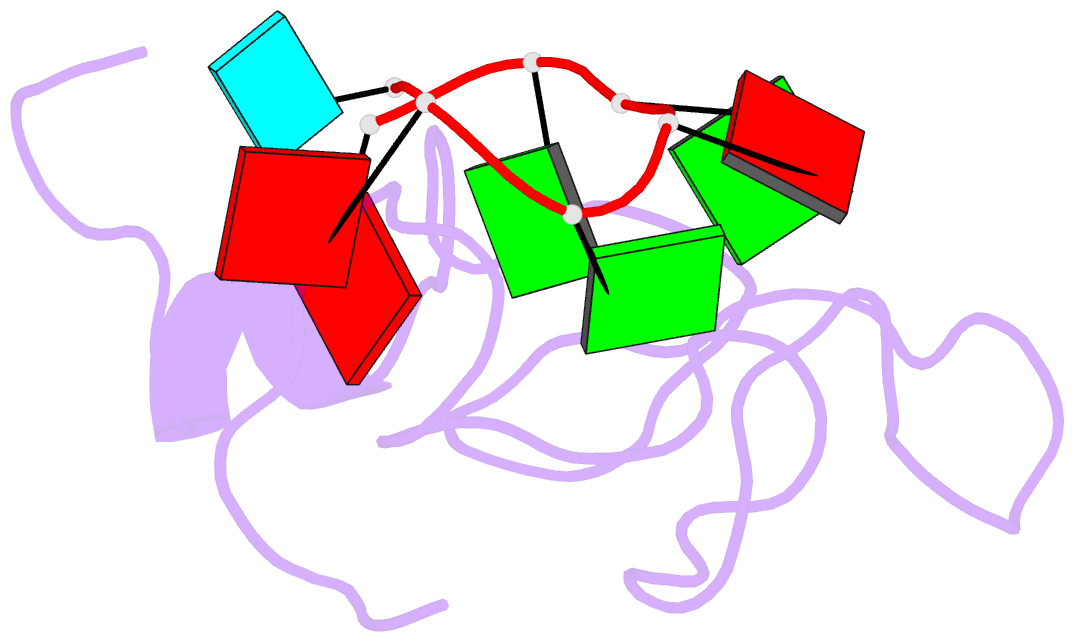
- The solution structure of the lin28-znf domains bound to aggagau of pre-let-7 mirna
- Reference
- Loughlin FE, Gebert LF, Towbin H, Brunschweiger A, Hall J, Allain FH (2011): "Structural basis of pre-let-7 miRNA recognition by the zinc knuckles of pluripotency factor Lin28." Nat.Struct.Mol.Biol., 19, 84-89. doi: 10.1038/nsmb.2202.
- Abstract
- Lin28 inhibits the biogenesis of let-7 miRNAs through a direct interaction with the terminal loop of pre-let-7. This interaction requires the zinc-knuckle domains of Lin28. We show that the zinc knuckle domains of Lin28 are sufficient to provide binding selectivity for pre-let-7 miRNAs and present the NMR structure of human Lin28 zinc knuckles bound to the short sequence 5'-AGGAGAU-3'. The structure reveals that each zinc knuckle recognizes an AG dinucleotide separated by a single nucleotide spacer. This defines a new 5'-NGNNG-3' consensus motif that explains how Lin28 selectively recognizes pre-let-7 family members. Binding assays in cell lysates and functional assays in cultured cells demonstrate that the interactions observed in the solution structure also occur between the full-length protein and members of the pre-let-7 family. The consensus sequence explains several seemingly disparate previously published observations on the binding properties of Lin28.