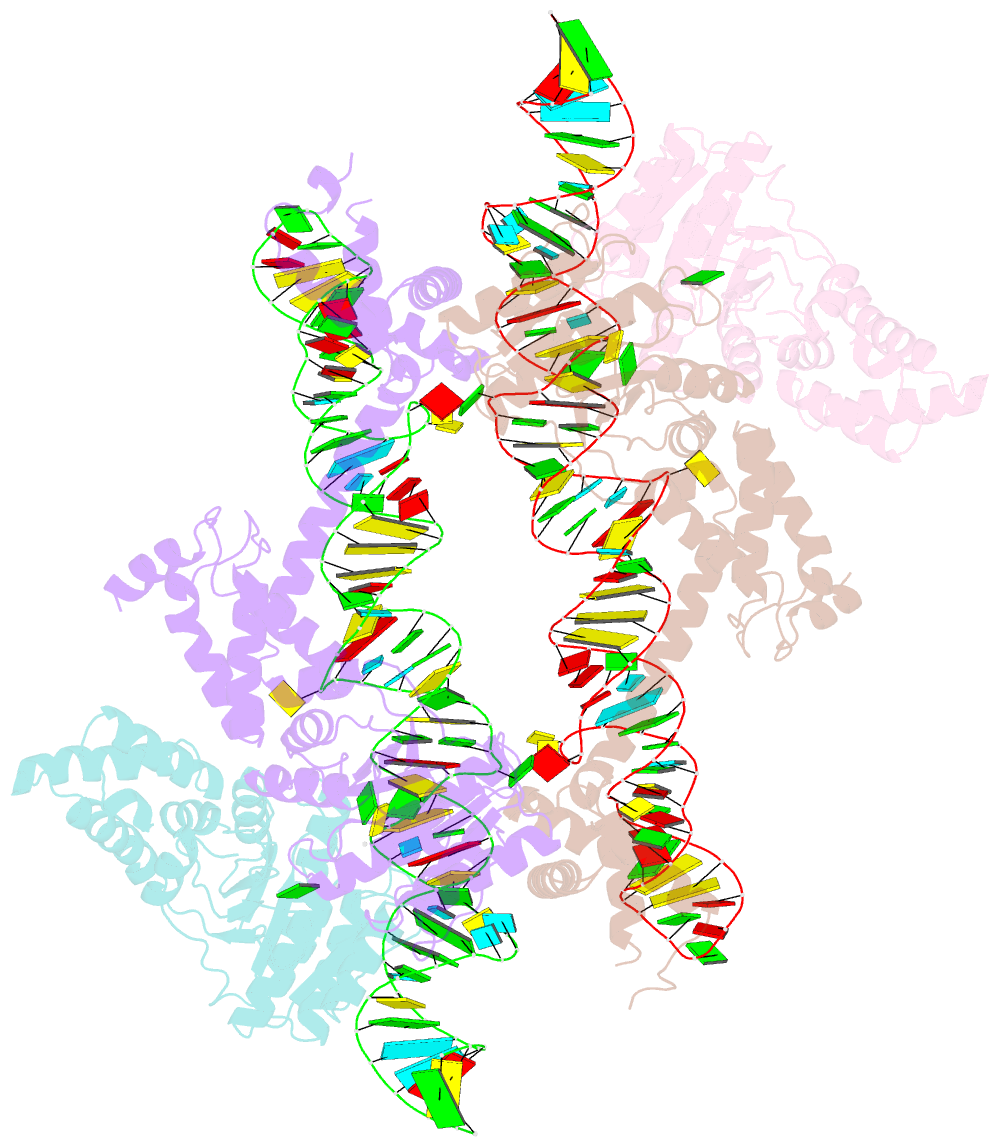
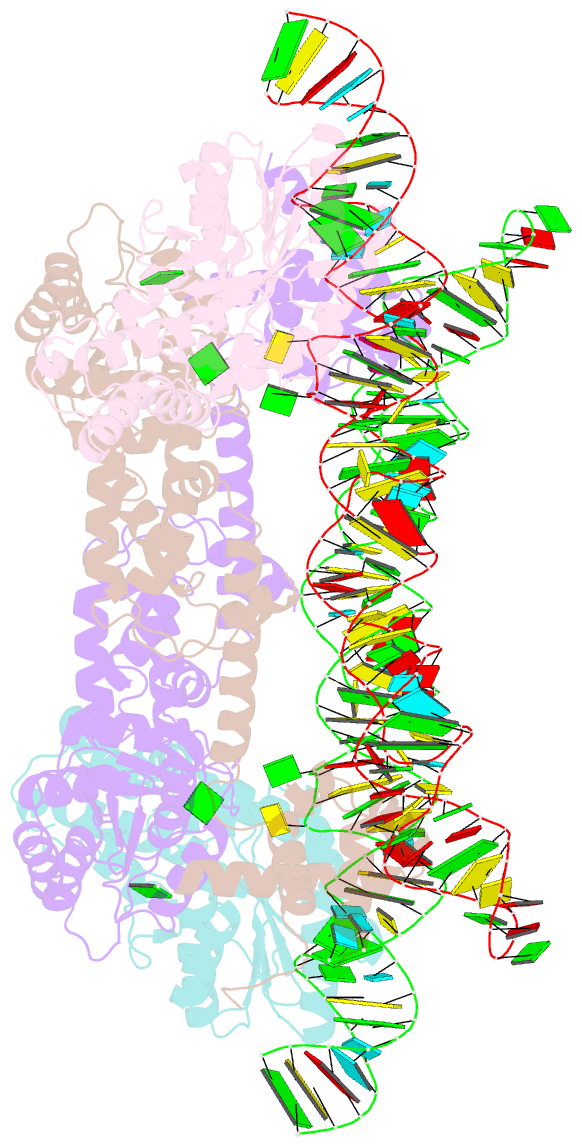
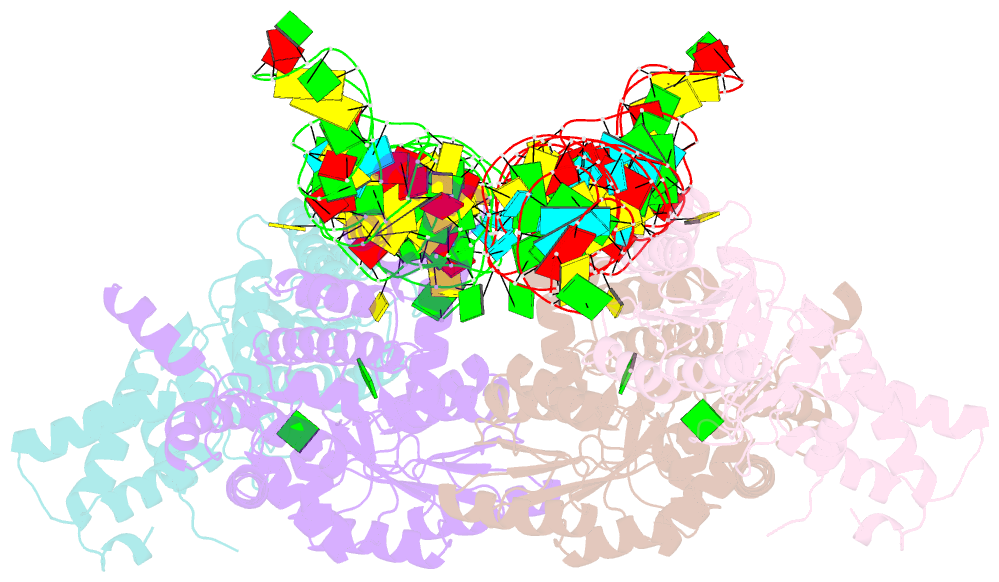
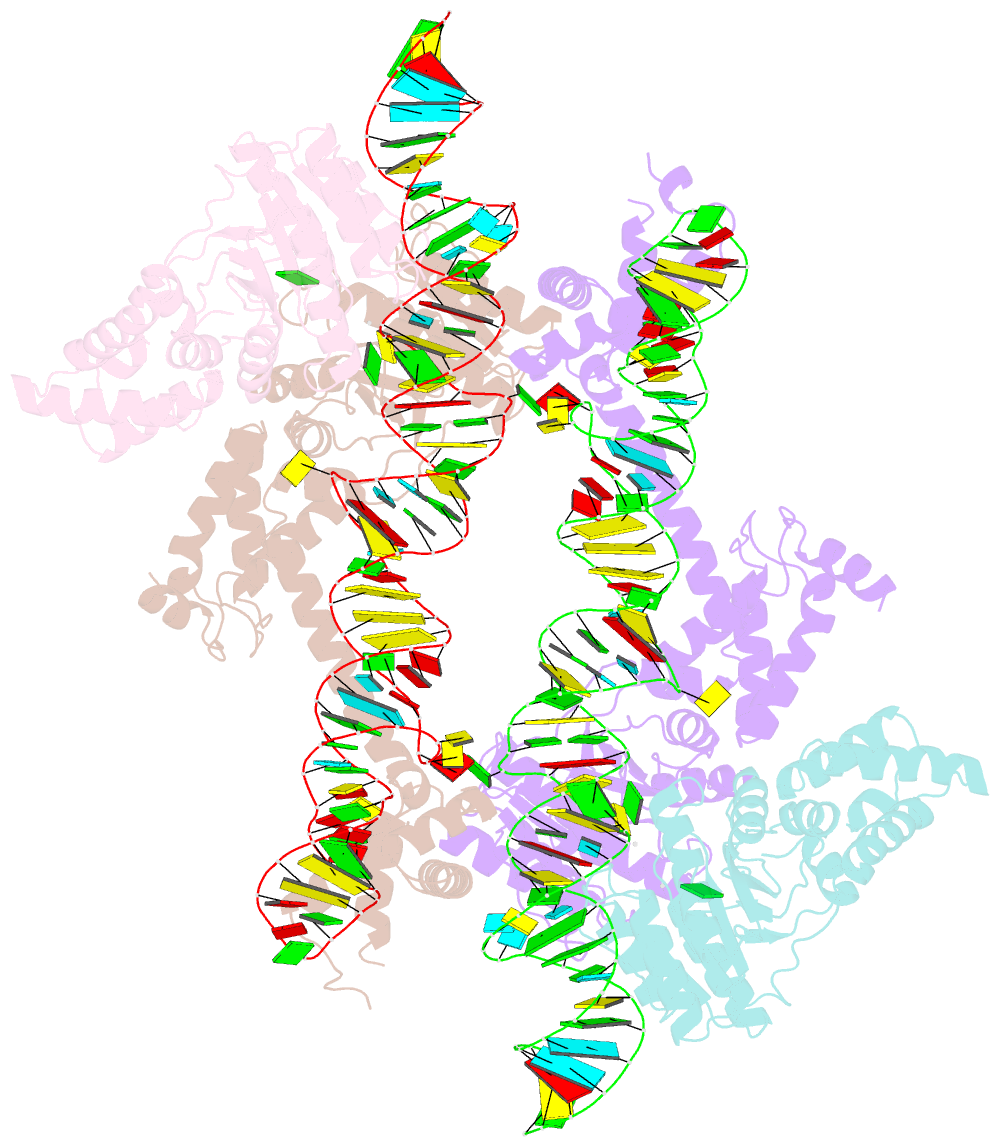
Summary information and primary citation
- PDB-id
- 2xxa; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- protein transport
- Method
- X-ray (3.94 Å)
- Summary
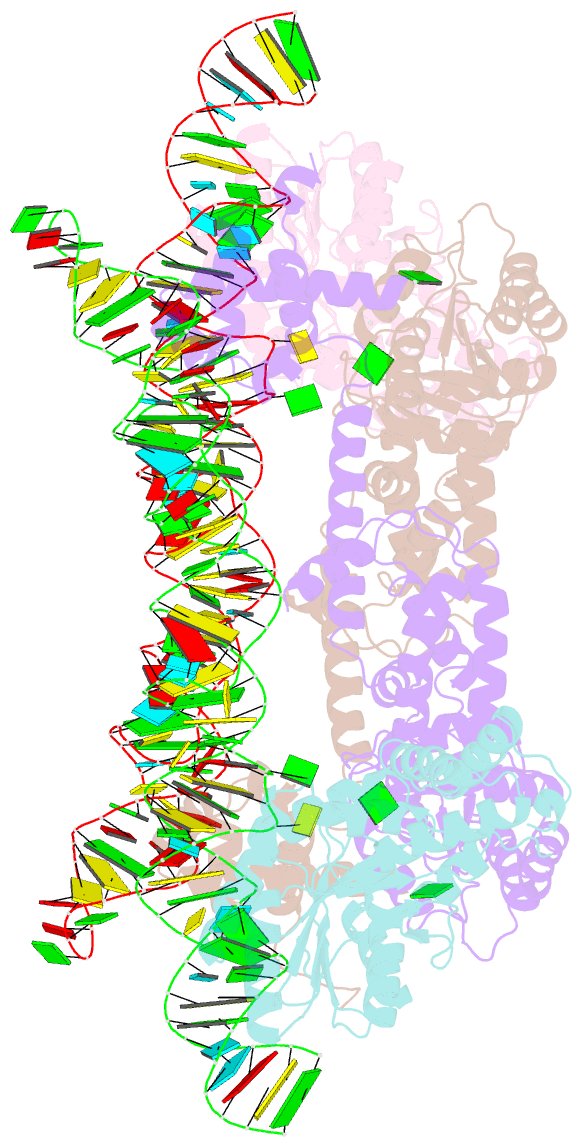
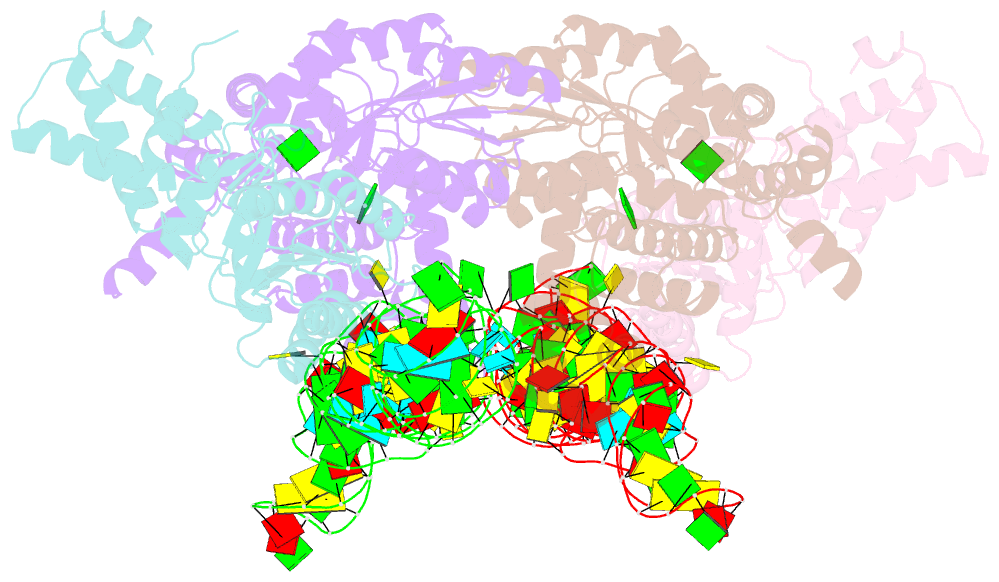
- The crystal structure of the signal recognition particle (srp) in complex with its receptor(sr)
- Reference
- Ataide SF, Schmitz N, Shen K, Ke A, Shan S, Doudna JA, Ban N (2011): "The Crystal Structure of the Signal Recognition Particle in Complex with its Receptor." Science, 331, 881. doi: 10.1126/SCIENCE.1196473.
- Abstract
- Cotranslational targeting of membrane and secretory proteins is mediated by the universally conserved signal recognition particle (SRP). Together with its receptor (SR), SRP mediates the guanine triphosphate (GTP)-dependent delivery of translating ribosomes bearing signal sequences to translocons on the target membrane. Here, we present the crystal structure of the SRP:SR complex at 3.9 angstrom resolution and biochemical data revealing that the activated SRP:SR guanine triphosphatase (GTPase) complex binds the distal end of the SRP hairpin RNA where GTP hydrolysis is stimulated. Combined with previous findings, these results suggest that the SRP:SR GTPase complex initially assembles at the tetraloop end of the SRP RNA and then relocalizes to the opposite end of the RNA. This rearrangement provides a mechanism for coupling GTP hydrolysis to the handover of cargo to the translocon.