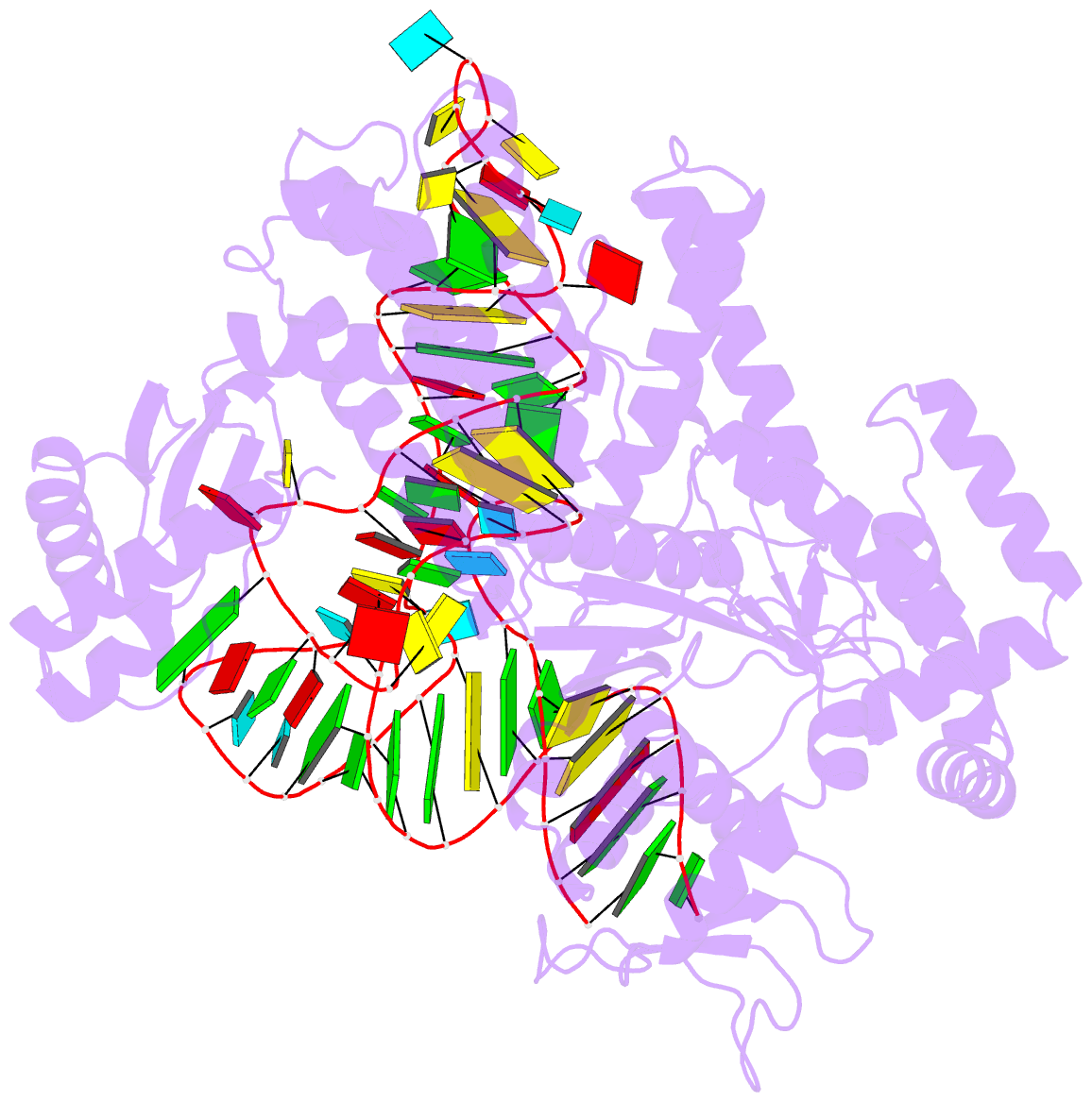
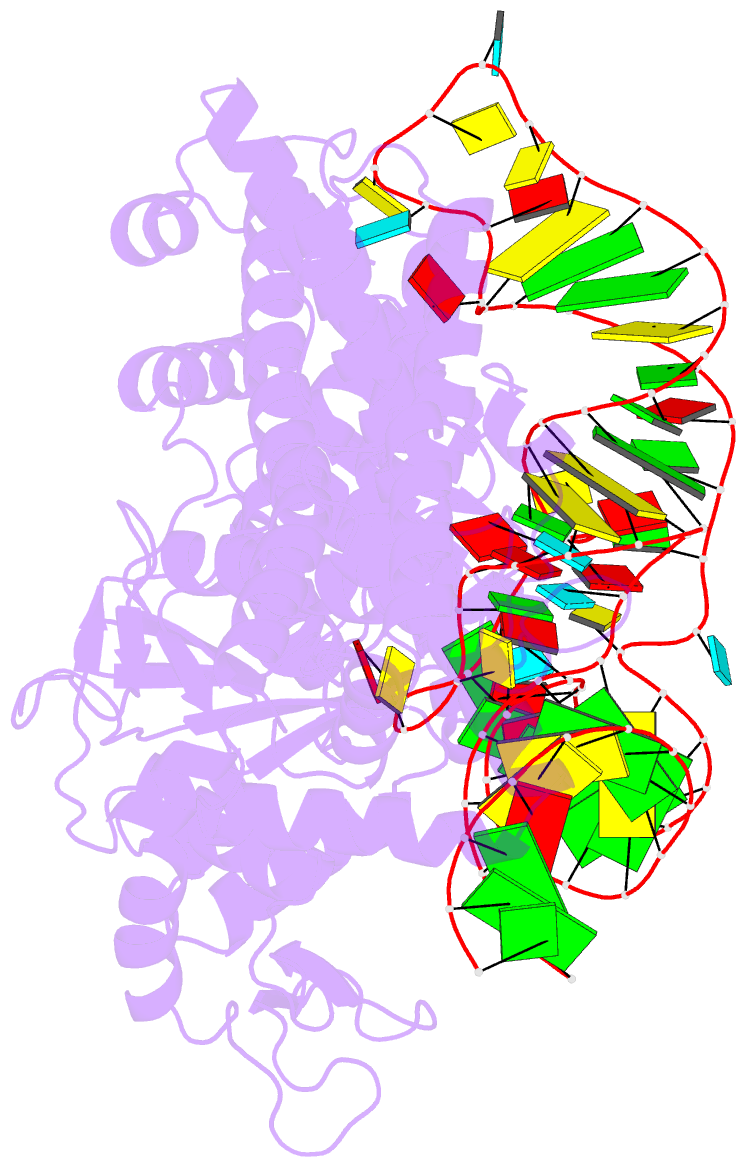
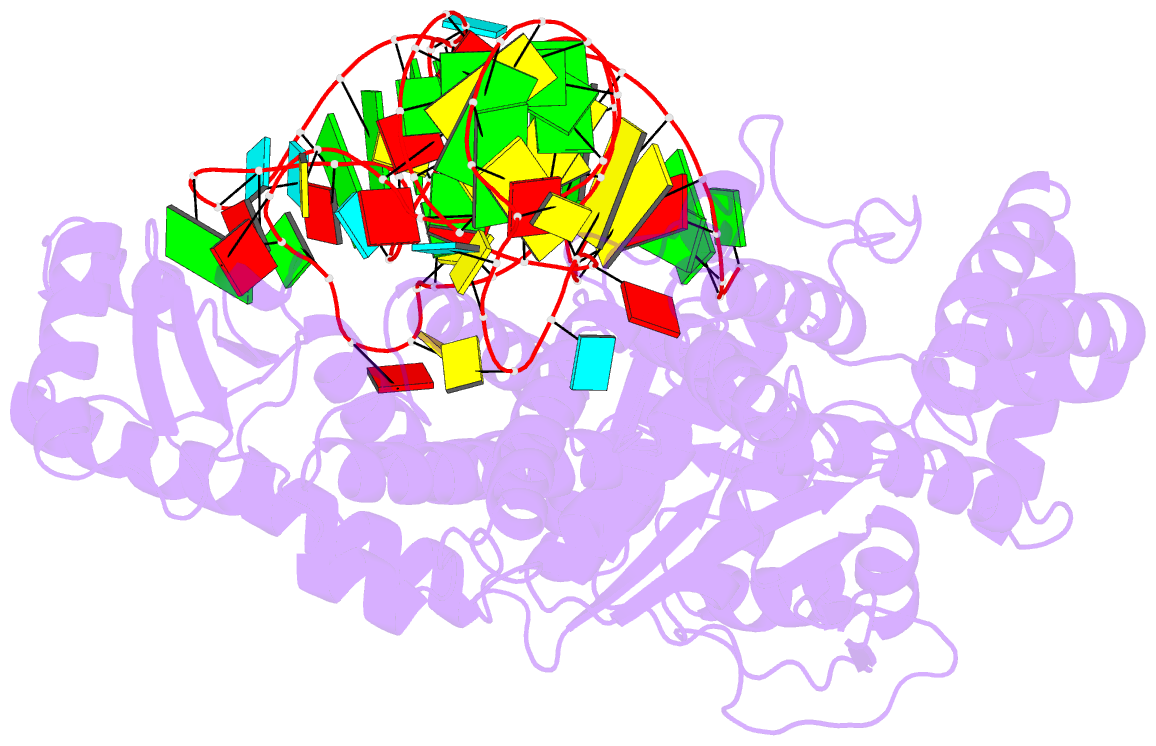
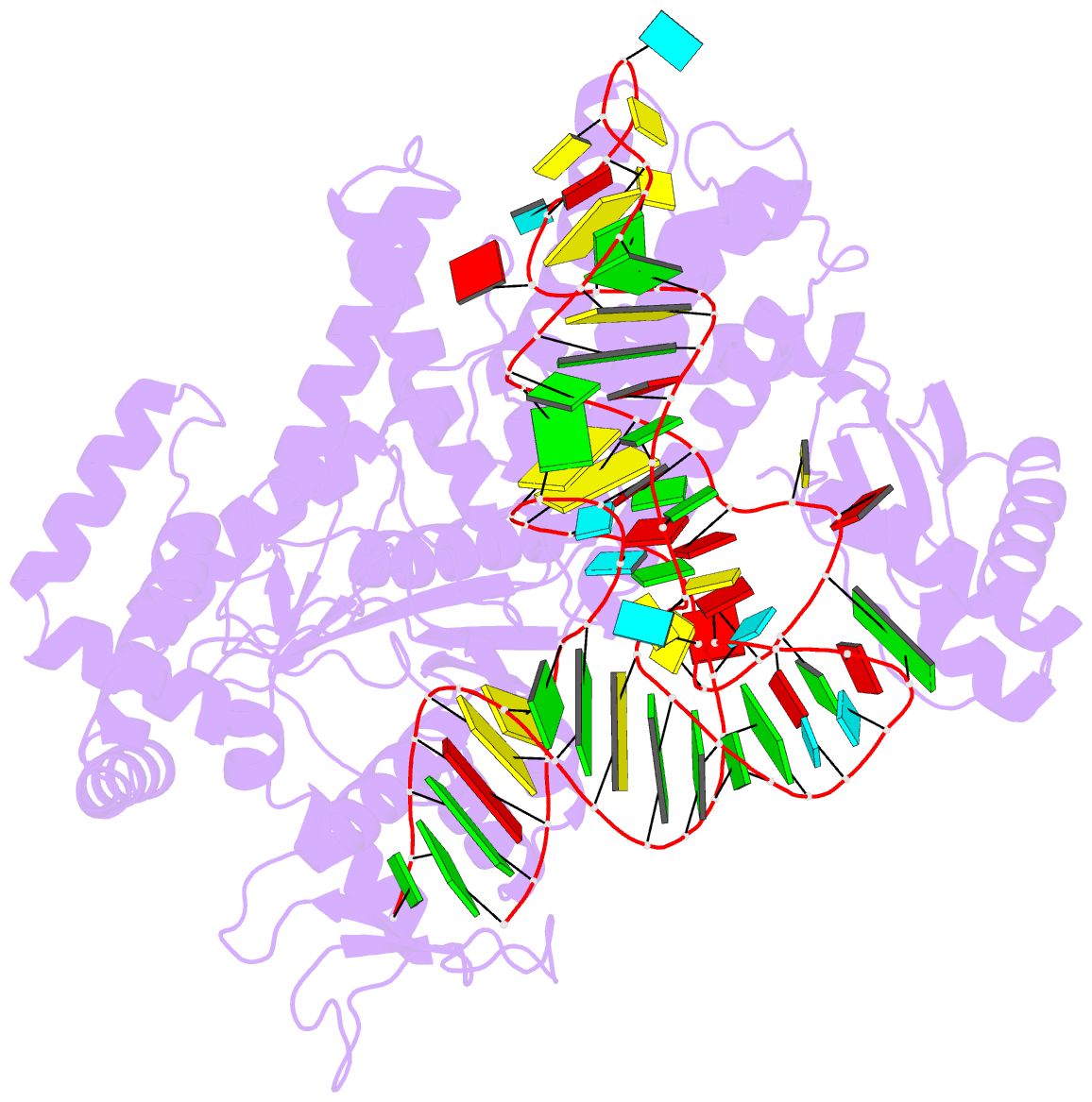
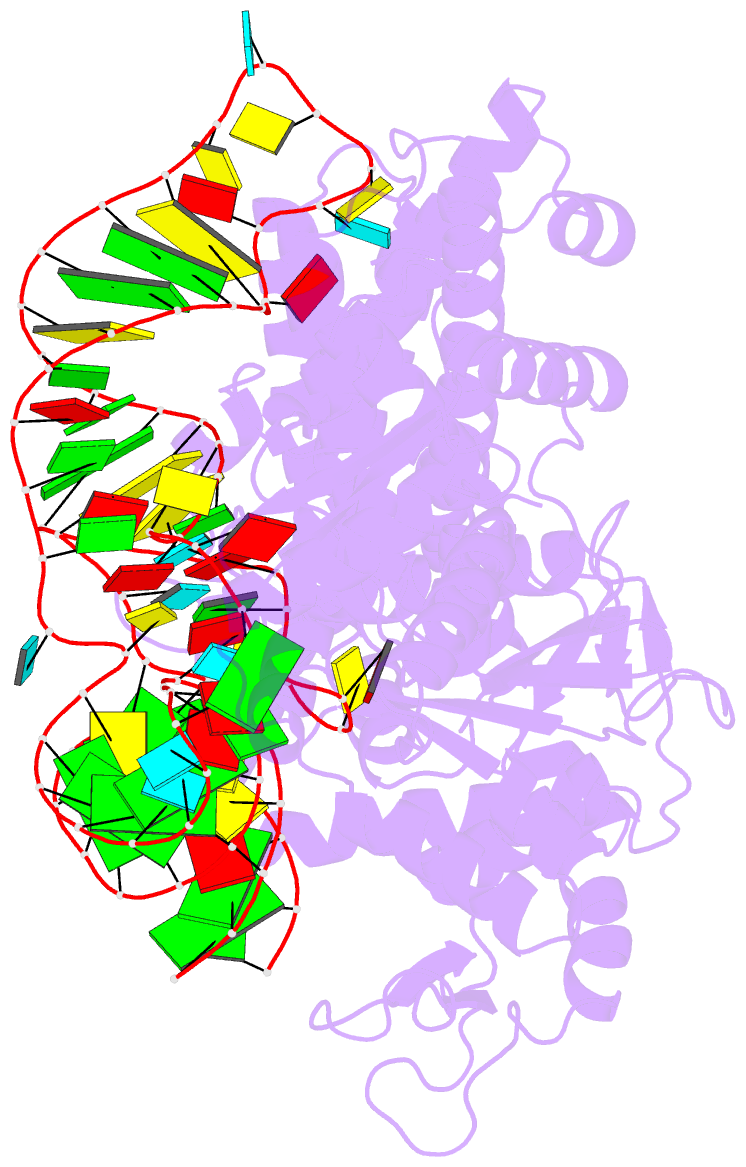
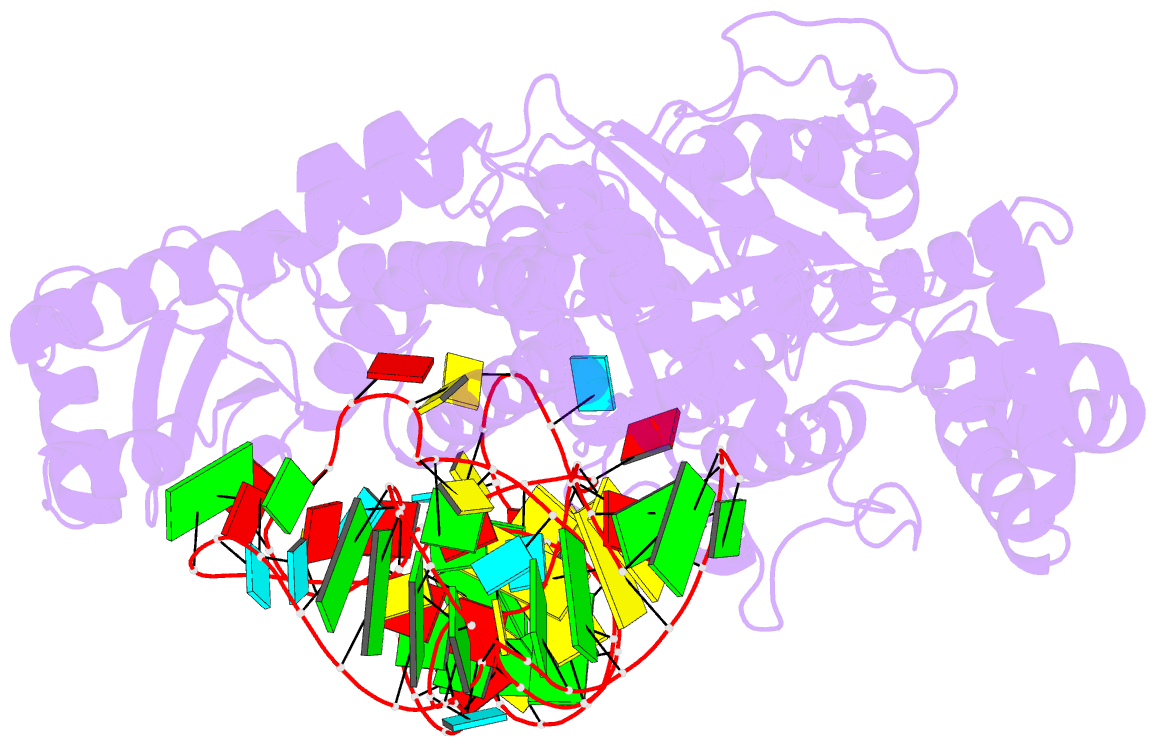
Summary information and primary citation
- PDB-id
- 2zuf; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- ligase-RNA
- Method
- X-ray (2.3 Å)
- Summary
- Crystal structure of pyrococcus horikoshii arginyl-trna synthetase complexed with trna(arg)
- Reference
- Konno M, Sumida T, Uchikawa E, Mori Y, Yanagisawa T, Sekine S, Yokoyama S (2009): "Modeling of tRNA-assisted mechanism of Arg activation based on a structure of Arg-tRNA synthetase, tRNA, and an ATP analog (ANP)." Febs J., 276, 4763-4779. doi: 10.1111/j.1742-4658.2009.07178.x.
- Abstract
- The ATP-pyrophosphate exchange reaction catalyzed by Arg-tRNA, Gln-tRNA and Glu-tRNA synthetases requires the assistance of the cognate tRNA. tRNA also assists Arg-tRNA synthetase in catalyzing the pyrophosphorolysis of synthetic Arg-AMP at low pH. The mechanism by which the 3'-end A76, and in particular its hydroxyl group, of the cognate tRNA is involved with the exchange reaction catalyzed by those enzymes has yet to be established. We determined a crystal structure of a complex of Arg-tRNA synthetase from Pyrococcus horikoshii, tRNA(Arg)(CCU) and an ATP analog with Rfactor = 0.213 (Rfree = 0.253) at 2.0 A resolution. On the basis of newly obtained structural information about the position of ATP bound on the enzyme, we constructed a structural model for a mechanism in which the formation of a hydrogen bond between the 2'-OH group of A76 of tRNA and the carboxyl group of Arg induces both formation of Arg-AMP (Arg + ATP --> Arg-AMP + pyrophosphate) and pyrophosphorolysis of Arg-AMP (Arg-AMP + pyrophosphate --> Arg + ATP) at low pH. Furthermore, we obtained a structural model of the molecular mechanism for the Arg-tRNA synthetase-catalyzed deacylation of Arg-tRNA (Arg-tRNA + AMP --> Arg-AMP + tRNA at high pH), in which the deacylation of aminoacyl-tRNA bound on Arg-tRNA synthetase and Glu-tRNA synthetase is catalyzed by a quite similar mechanism, whereby the proton-donating group (-NH-C+(NH2)2 or -COOH) of Arg and Glu assists the aminoacyl transfer from the 2'-OH group of tRNA to the phosphate group of AMP at high pH.