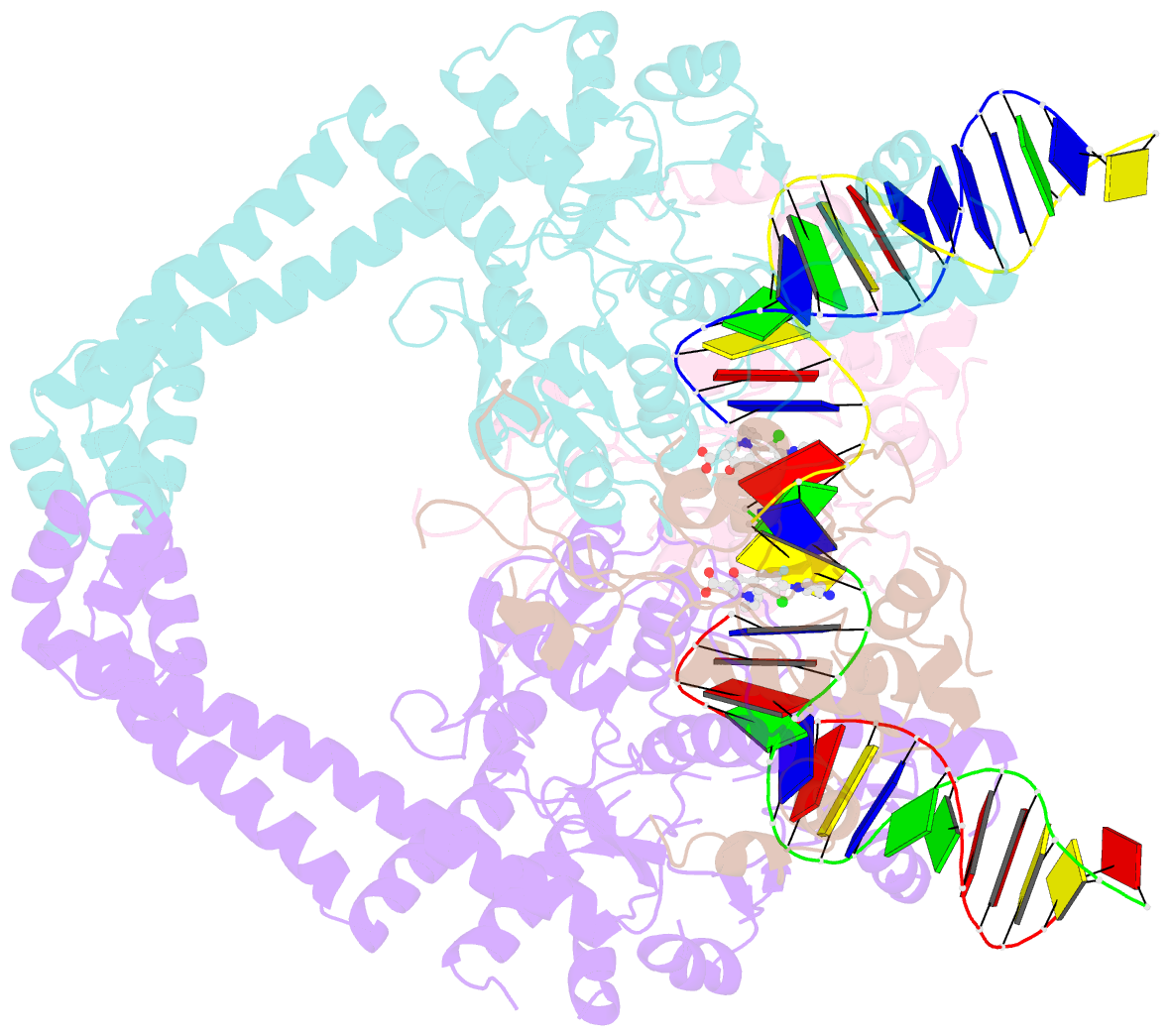
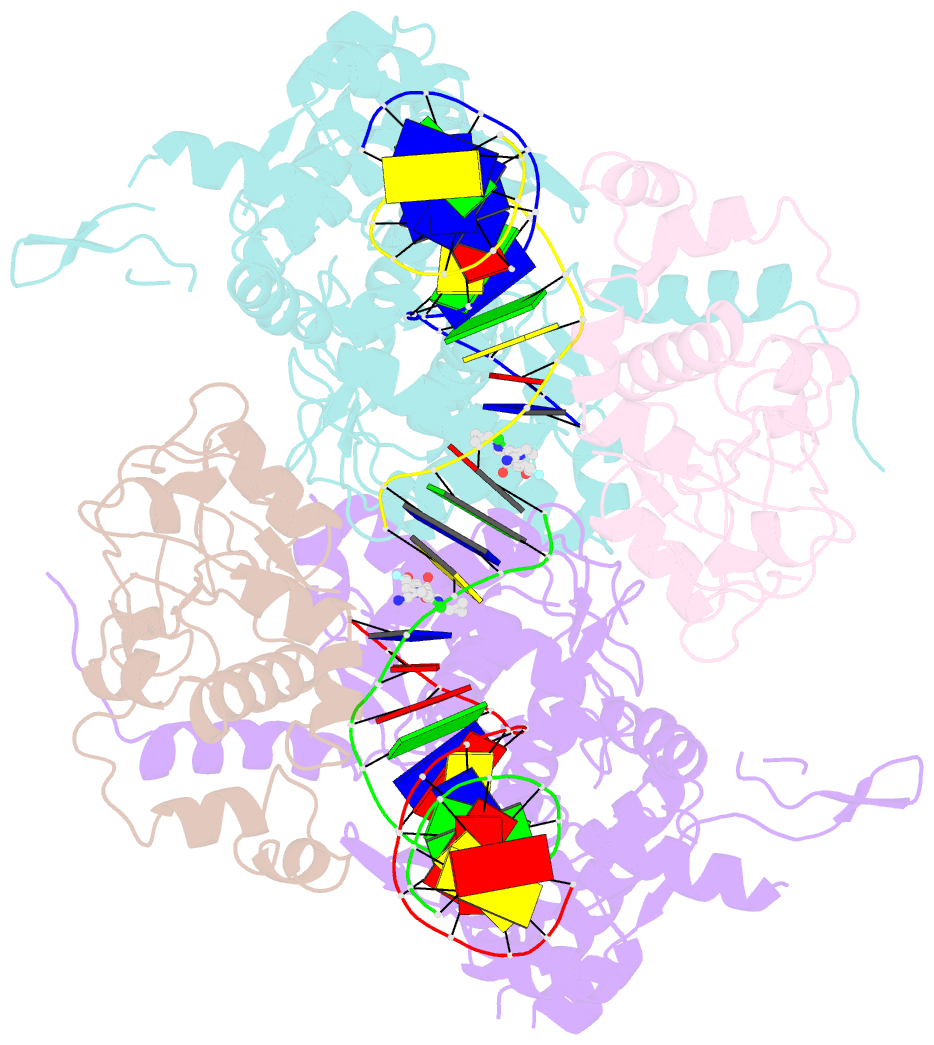
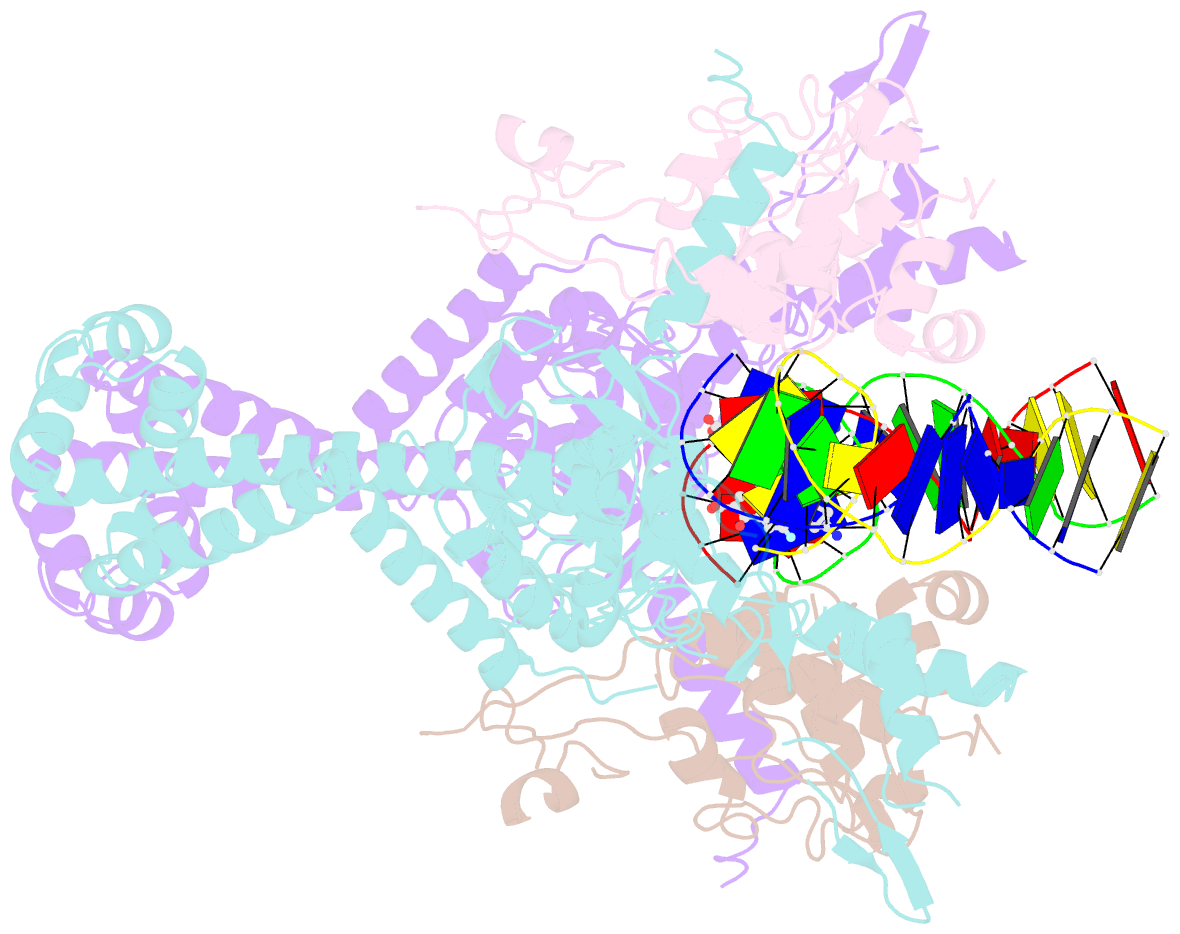
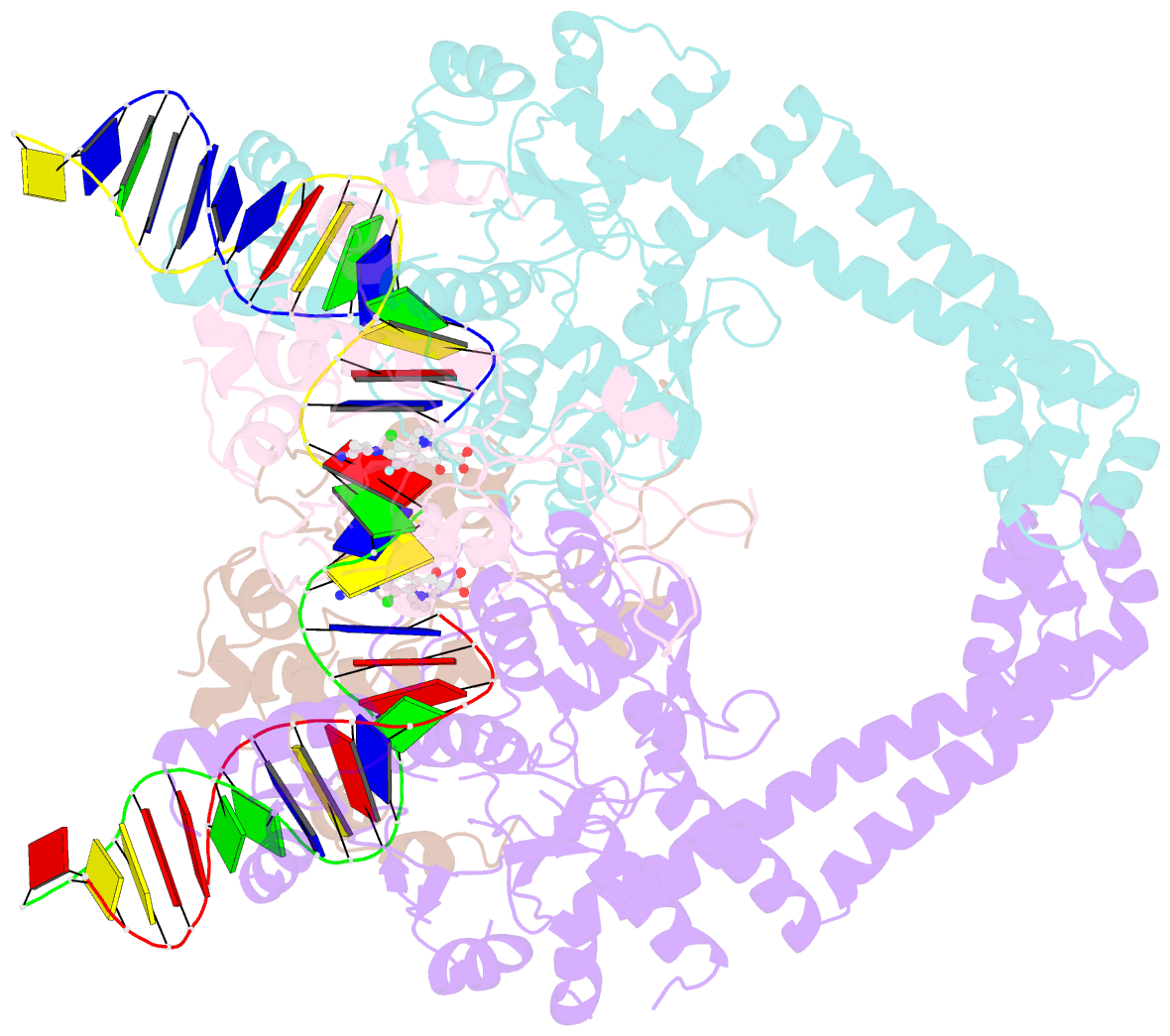
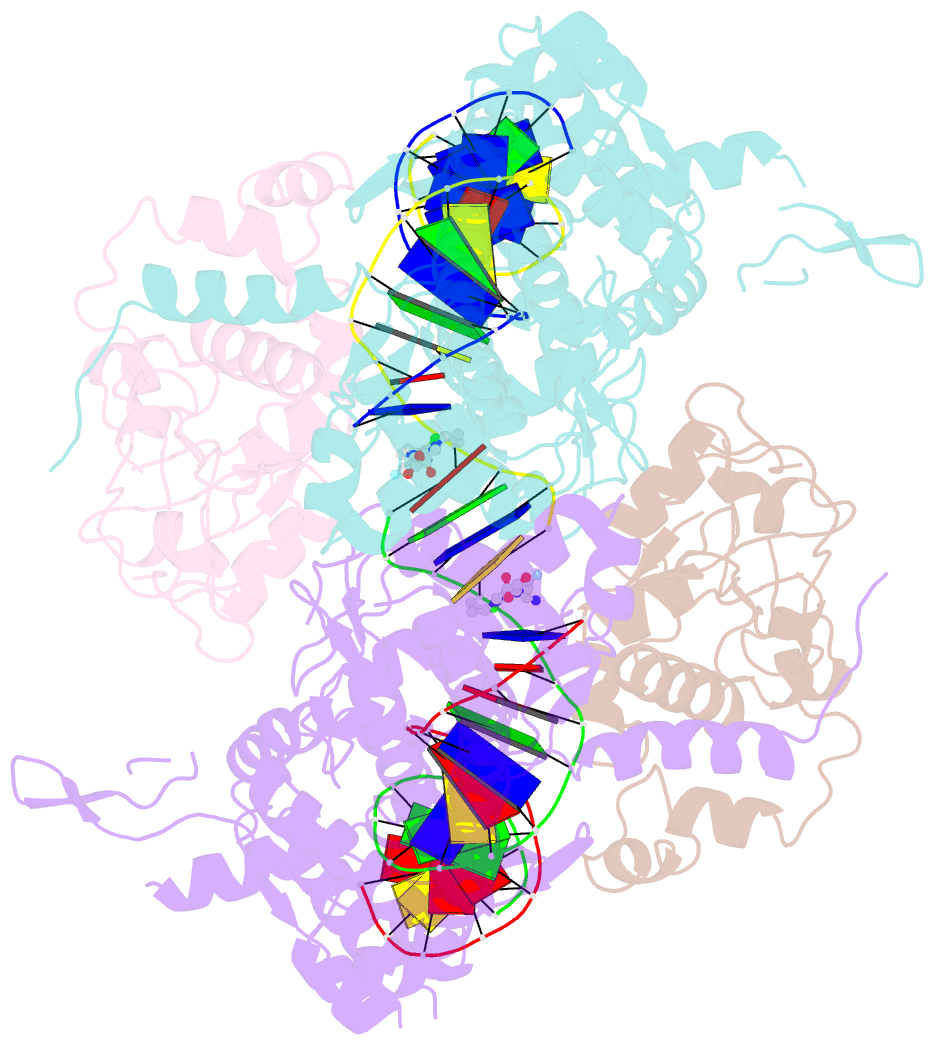
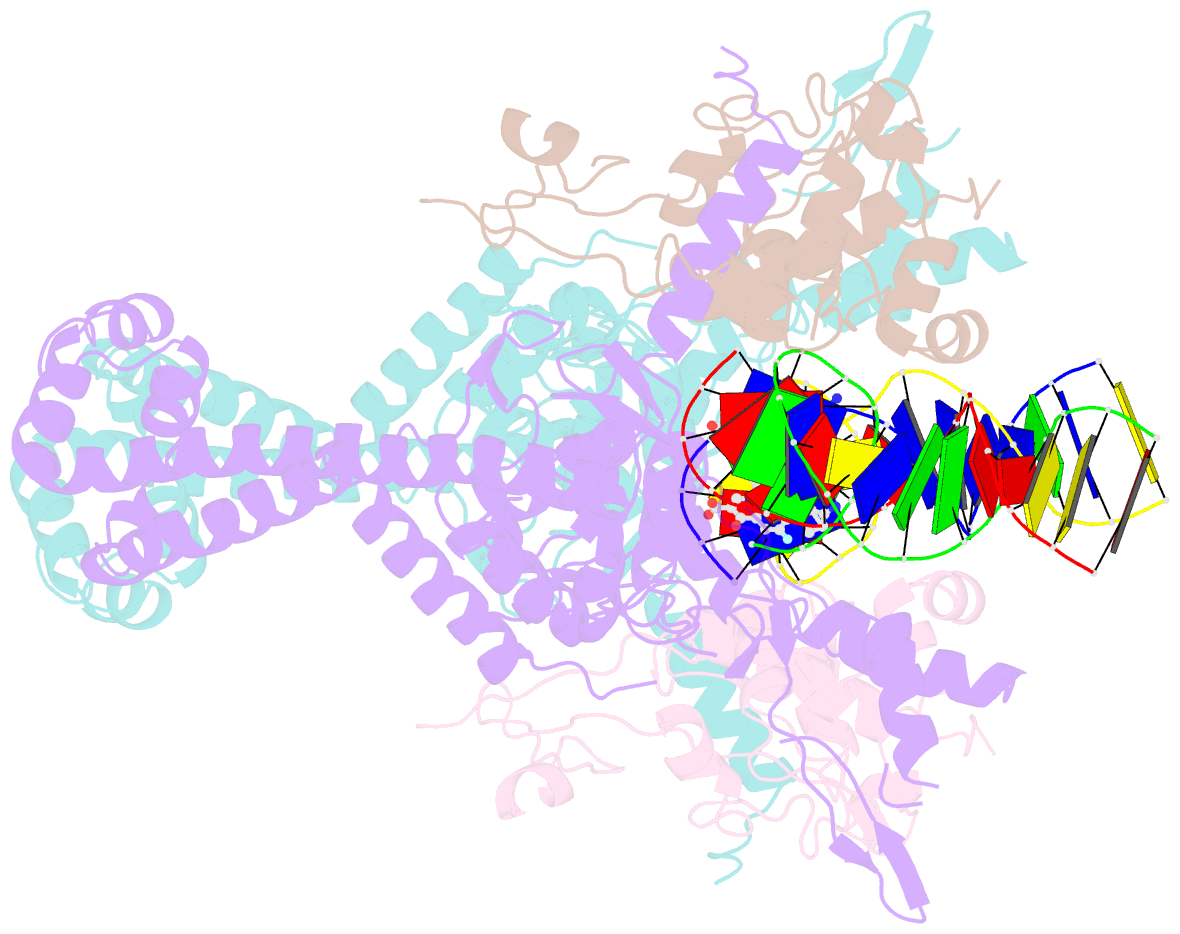
Summary information and primary citation
- PDB-id
- 3foe; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- isomerase-DNA
- Method
- X-ray (4.001 Å)
- Summary
- Structural insight into the quinolone-DNA cleavage complex of type iia topoisomerases
- Reference
- Laponogov I, Sohi MK, Veselkov DA, Pan X-S, Sawhney R, Thompson AW, McAuley KE, Fisher LM, Sanderson MR (2009): "Structural insight into the quinolone-DNA cleavage complex of type IIA topoisomerases." Nat.Struct.Mol.Biol., 16, 667-669. doi: 10.1038/nsmb.1604.
- Abstract
- Type II topoisomerases alter DNA topology by forming a covalent DNA-cleavage complex that allows DNA transport through a double-stranded DNA break. We present the structures of cleavage complexes formed by the Streptococcus pneumoniae ParC breakage-reunion and ParE TOPRIM domains of topoisomerase IV stabilized by moxifloxacin and clinafloxacin, two antipneumococcal fluoroquinolones. These structures reveal two drug molecules intercalated at the highly bent DNA gate and help explain antibacterial quinolone action and resistance.