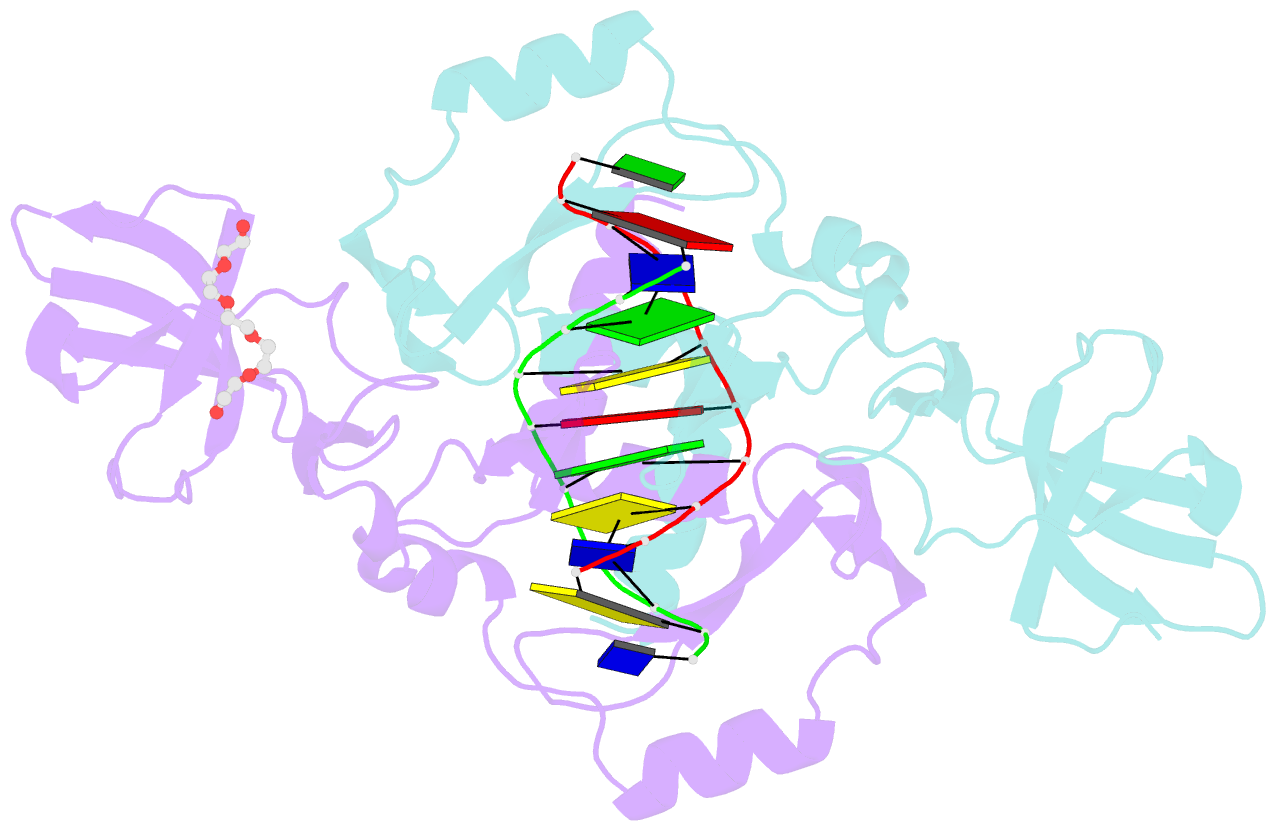
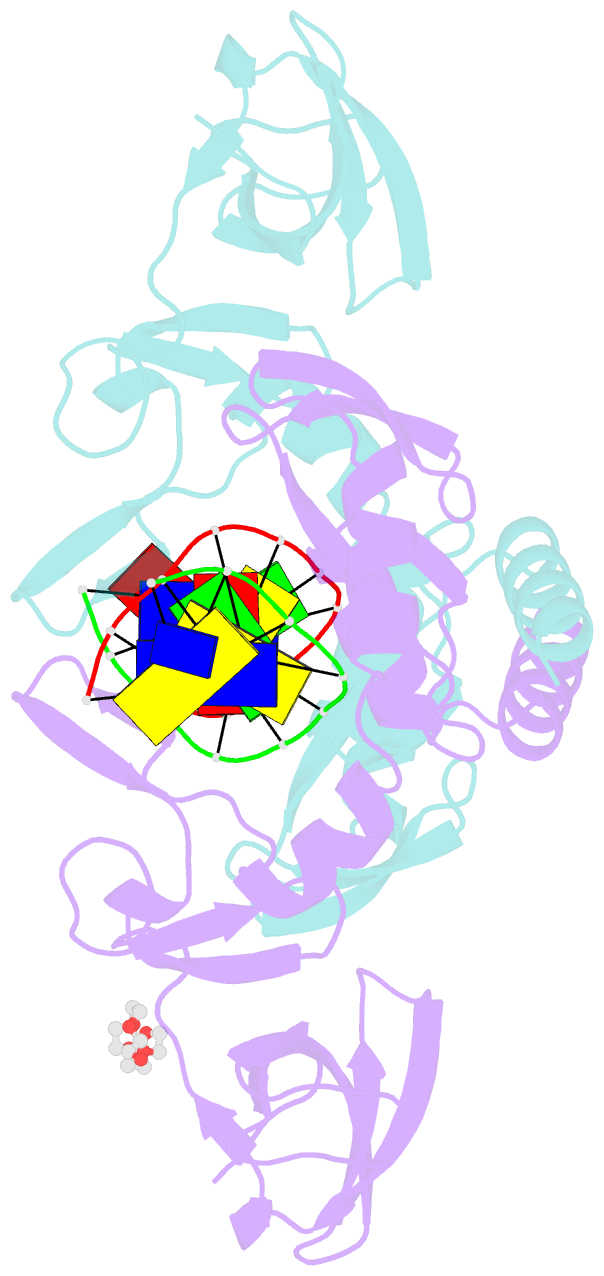
Summary information and primary citation
- PDB-id
- 3gox; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- hydrolase-DNA
- Method
- X-ray (1.5 Å)
- Summary
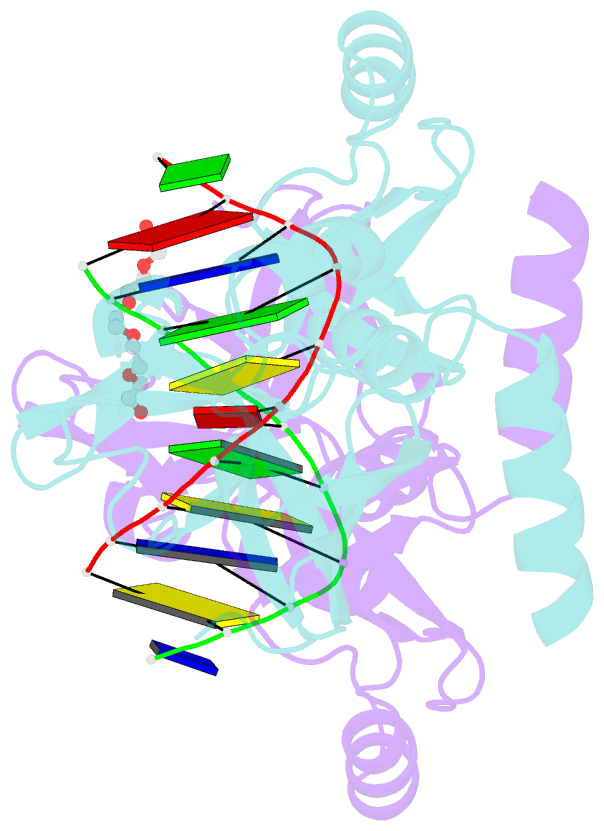
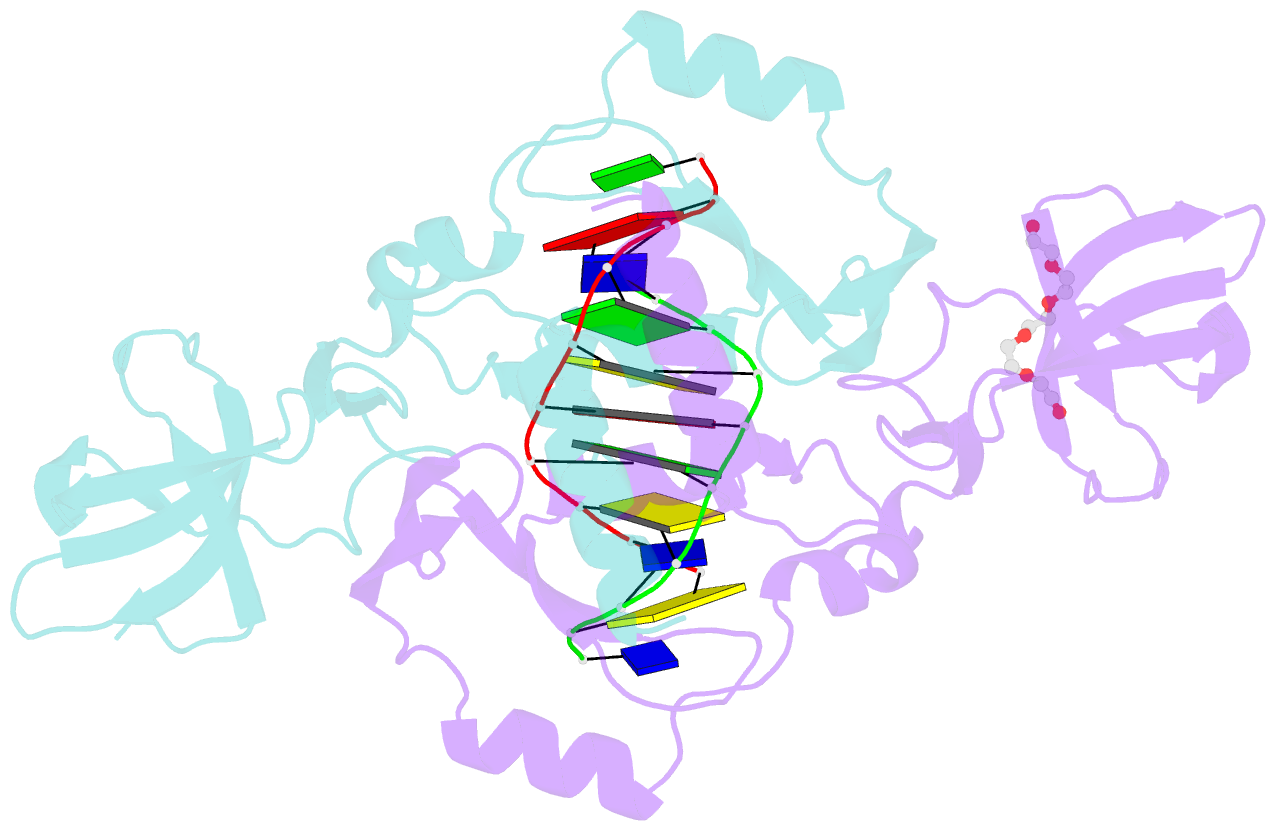
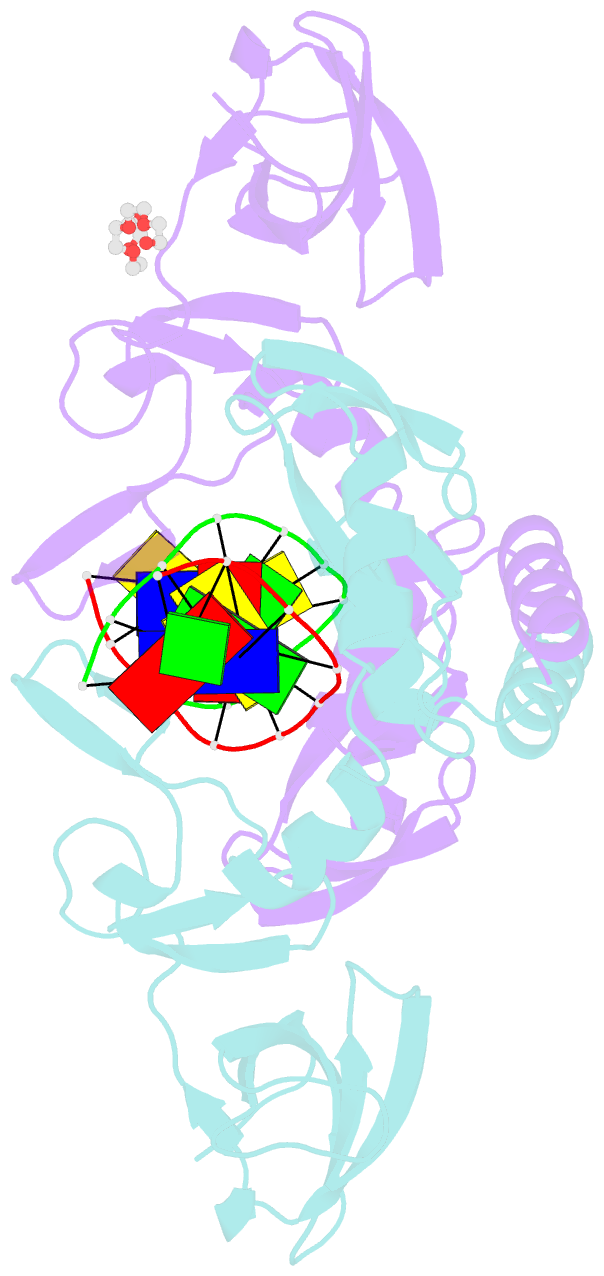
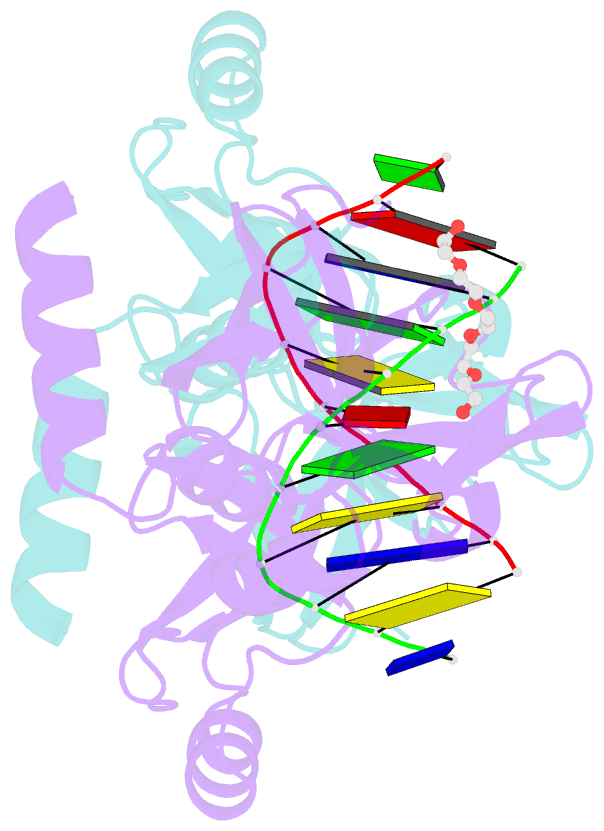
- Crystal structure of the beta-beta-alpha-me type ii restriction endonuclease hpy99i in the absence of edta
- Reference
- Sokolowska M, Czapinska H, Bochtler M (2009): "Crystal structure of the beta beta alpha-Me type II restriction endonuclease Hpy99I with target DNA." Nucleic Acids Res., 37, 3799-3810. doi: 10.1093/nar/gkp228.
- Abstract
- The beta beta alpha-Me restriction endonuclease (REase) Hpy99I recognizes the CGWCG target sequence and cleaves it with unusual stagger (five nucleotide 5'-recessed ends). Here we present the crystal structure of the specific complex of the dimeric enzyme with DNA. The Hpy99I protomer consists of an antiparallel beta-barrel and two beta 4 alpha 2 repeats. Each repeat coordinates a structural zinc ion with four cysteine thiolates in two CXXC motifs. The beta beta alpha-Me region of the second beta 4 alpha 2 repeat holds the catalytic metal ion (or its sodium surrogate) via Asp148 and Asn165 and activates a water molecule with the general base His149. In the specific complex, Hpy99I forms a ring-like structure around the DNA that contacts DNA bases on the major and minor groove sides via the first and second beta 4 alpha 2 repeats, respectively. Hpy99I interacts with the central base pair of the recognition sequence only on the minor groove side, where A:T resembles T:A and G:C is similar to C:G. The Hpy99I-DNA co-crystal structure provides the first detailed illustration of the beta beta alpha-Me site in REases and complements structural information on the use of this active site motif in other groups of endonucleases such as homing endonucleases (e.g. I-PpoI) and Holliday junction resolvases (e.g. T4 endonuclease VII).