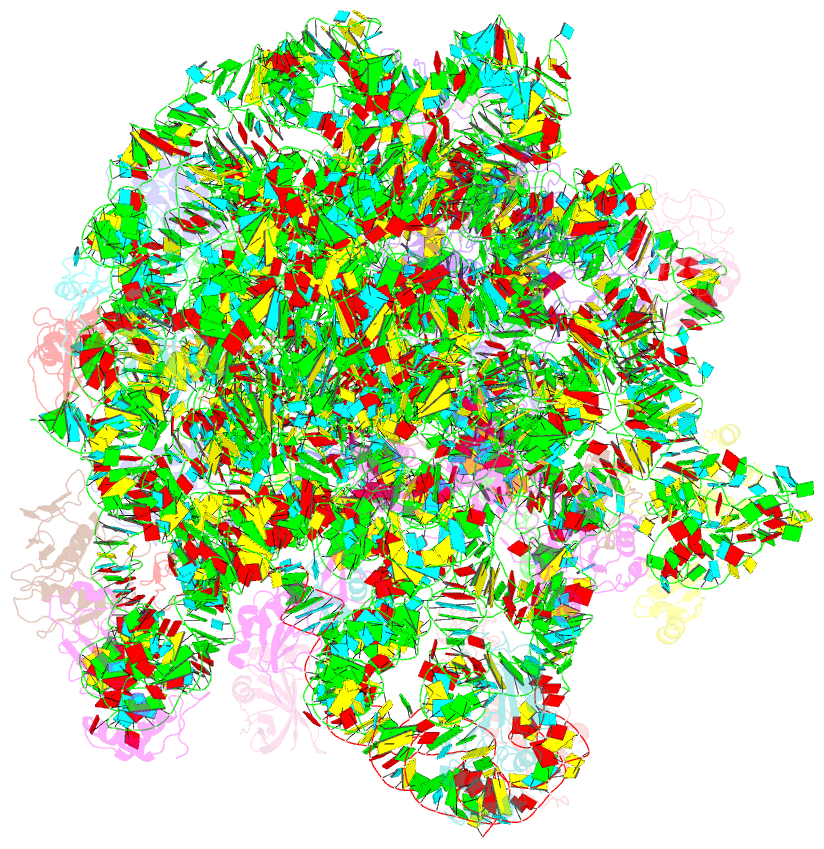
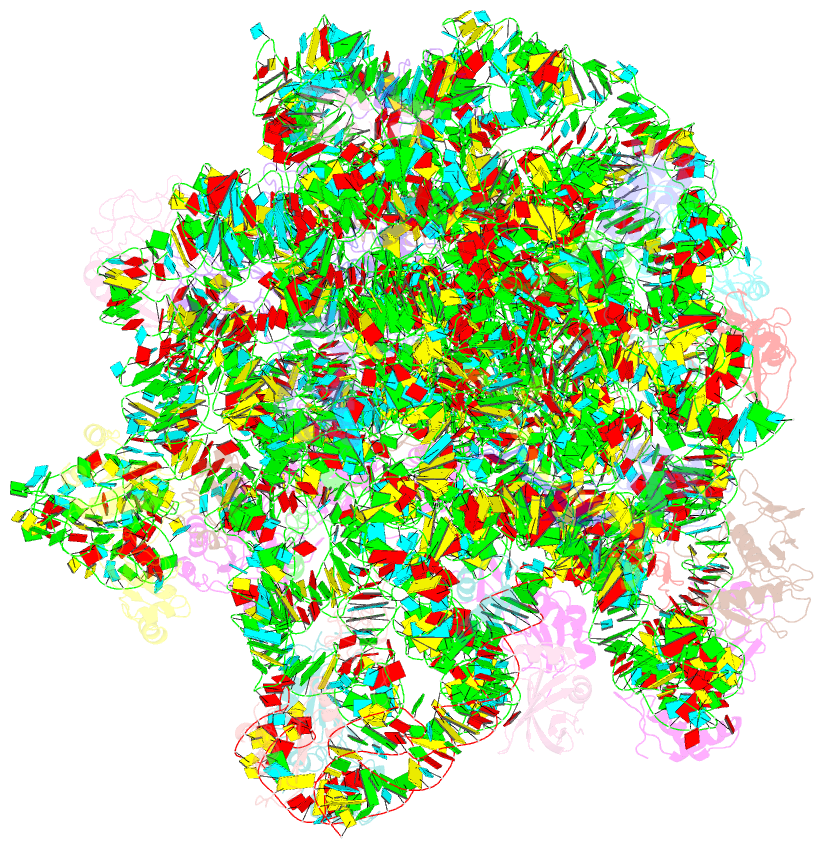
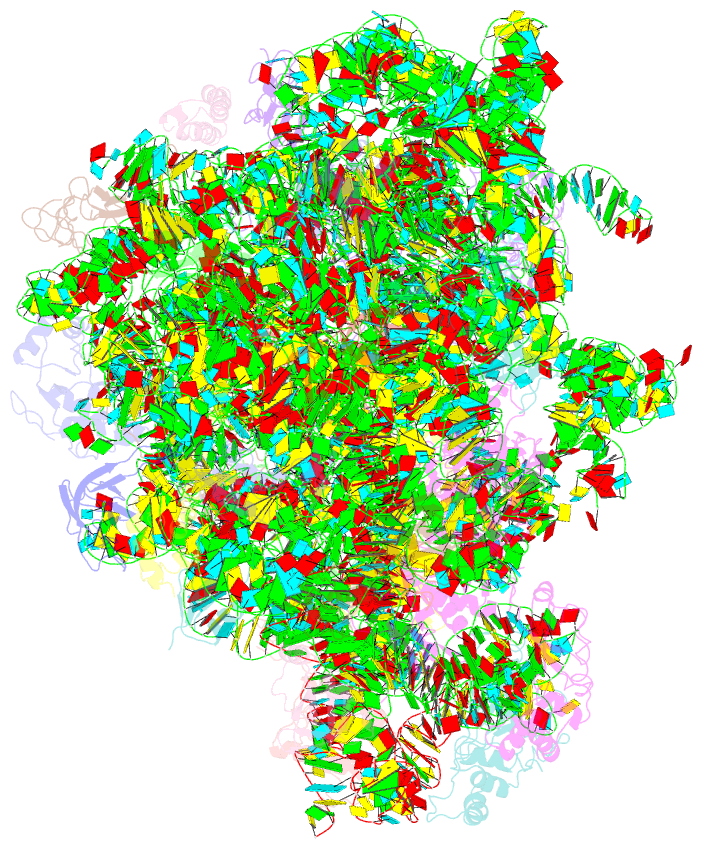
Summary information and primary citation
- PDB-id
- 3j8g; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- ribosome
- Method
- cryo-EM (5.0 Å)
- Summary
- Electron cryo-microscopy structure of enga bound with the 50s ribosomal subunit
- Reference
- Zhang X, Yan K, Zhang Y, Li N, Ma C, Li Z, Zhang Y, Feng B, Liu J, Sun Y, Xu Y, Lei J, Gao N (2014): "Structural insights into the function of a unique tandem GTPase EngA in bacterial ribosome assembly." Nucleic Acids Res. doi: 10.1093/nar/gku1135.
- Abstract
- Many ribosome-interacting GTPases, with proposed functions in ribosome biogenesis, are also implicated in the cellular regulatory coupling between ribosome assembly process and various growth control pathways. EngA is an essential GTPase in bacteria, and intriguingly, it contains two consecutive GTPase domains (GD), being one-of-a-kind among all known GTPases. EngA is required for the 50S subunit maturation. However, its molecular role remains elusive. Here, we present the structure of EngA bound to the 50S subunit. Our data show that EngA binds to the peptidyl transferase center (PTC) and induces dramatic conformational changes on the 50S subunit, which virtually returns the 50S subunit to a state similar to that of the late-stage 50S assembly intermediates. Very interestingly, our data show that the two GDs exhibit a pseudo-two-fold symmetry in the 50S-bound conformation. Our results indicate that EngA recognizes certain forms of the 50S assembly intermediates, and likely facilitates the conformational maturation of the PTC of the 23S rRNA in a direct manner. Furthermore, in a broad context, our data also suggest that EngA might be a sensor of the cellular GTP/GDP ratio, endowed with multiple conformational states, in response to fluctuations in cellular nucleotide pool, to facilitate and regulate ribosome assembly.