Summary information and primary citation
- PDB-id
- 3qx3; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- isomerase-DNA-isomerase inhibitor
- Method
- X-ray (2.162 Å)
- Summary
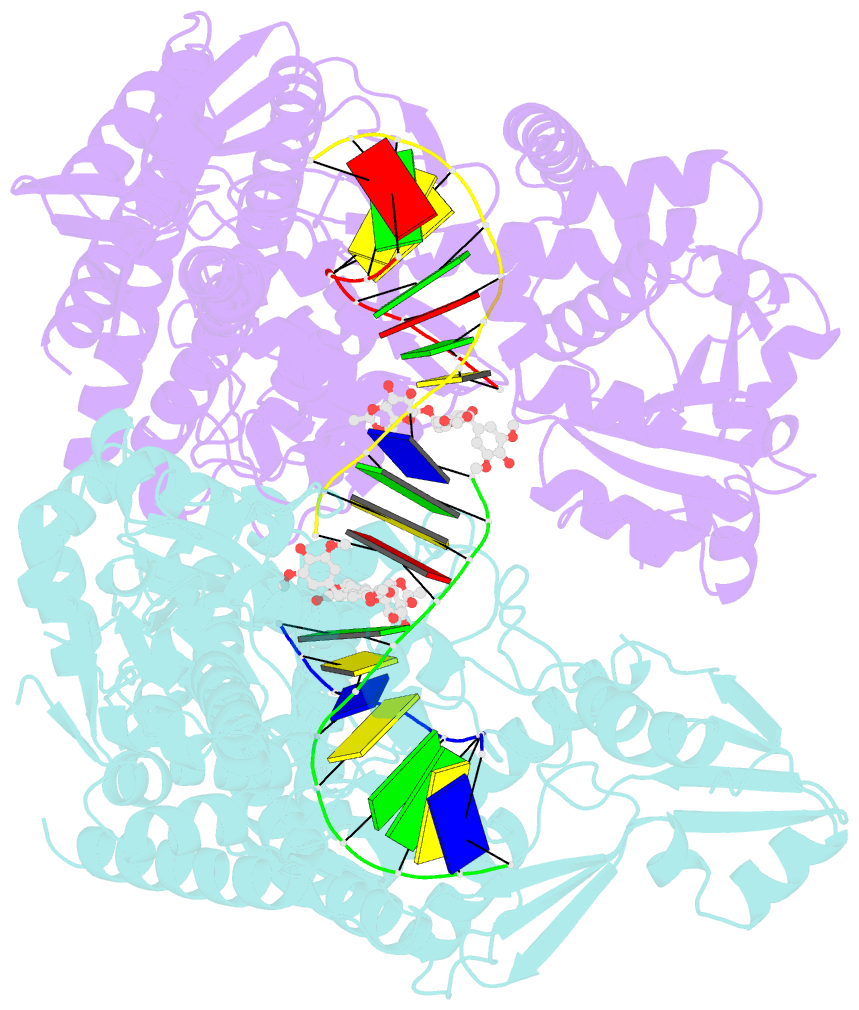
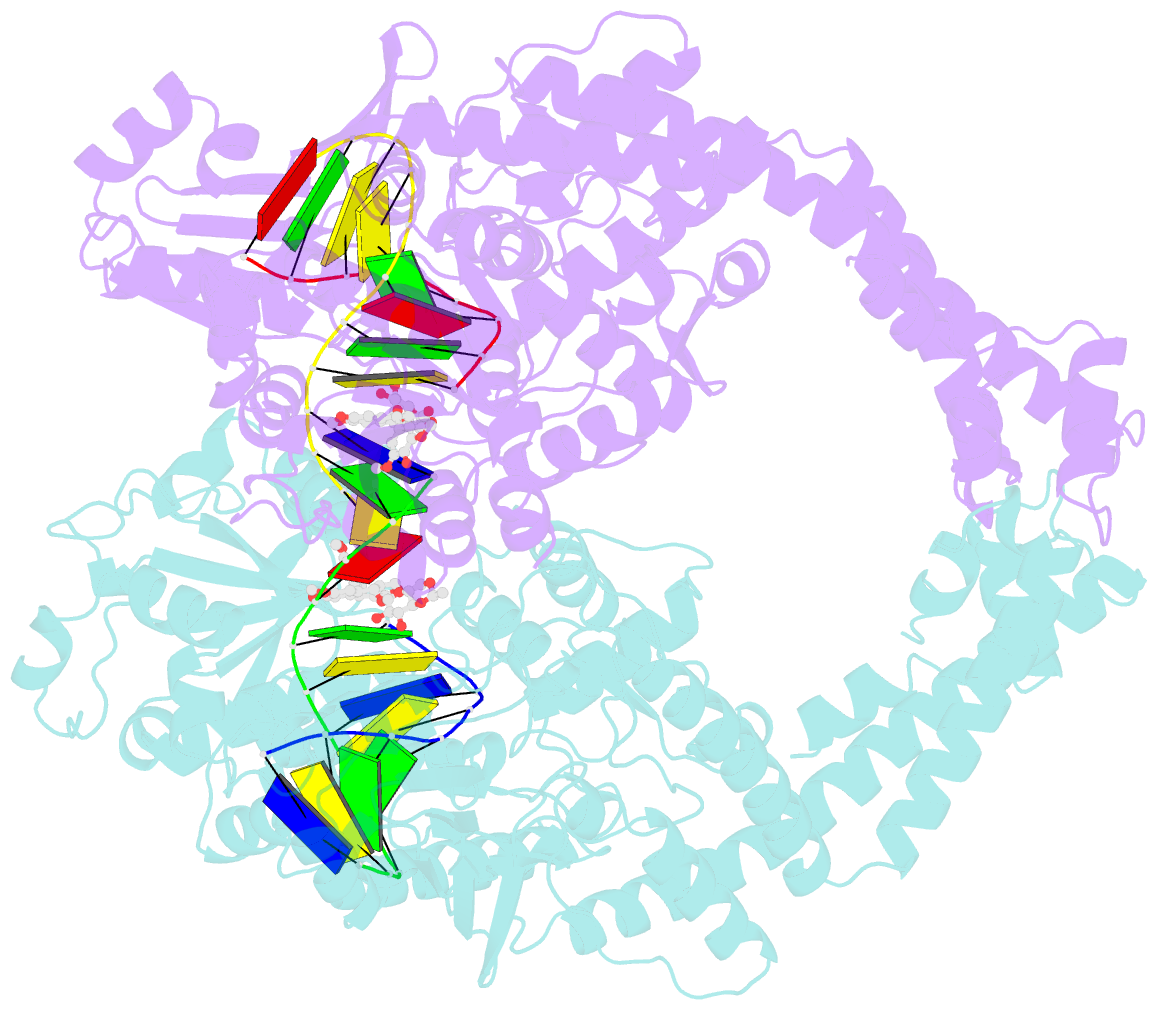
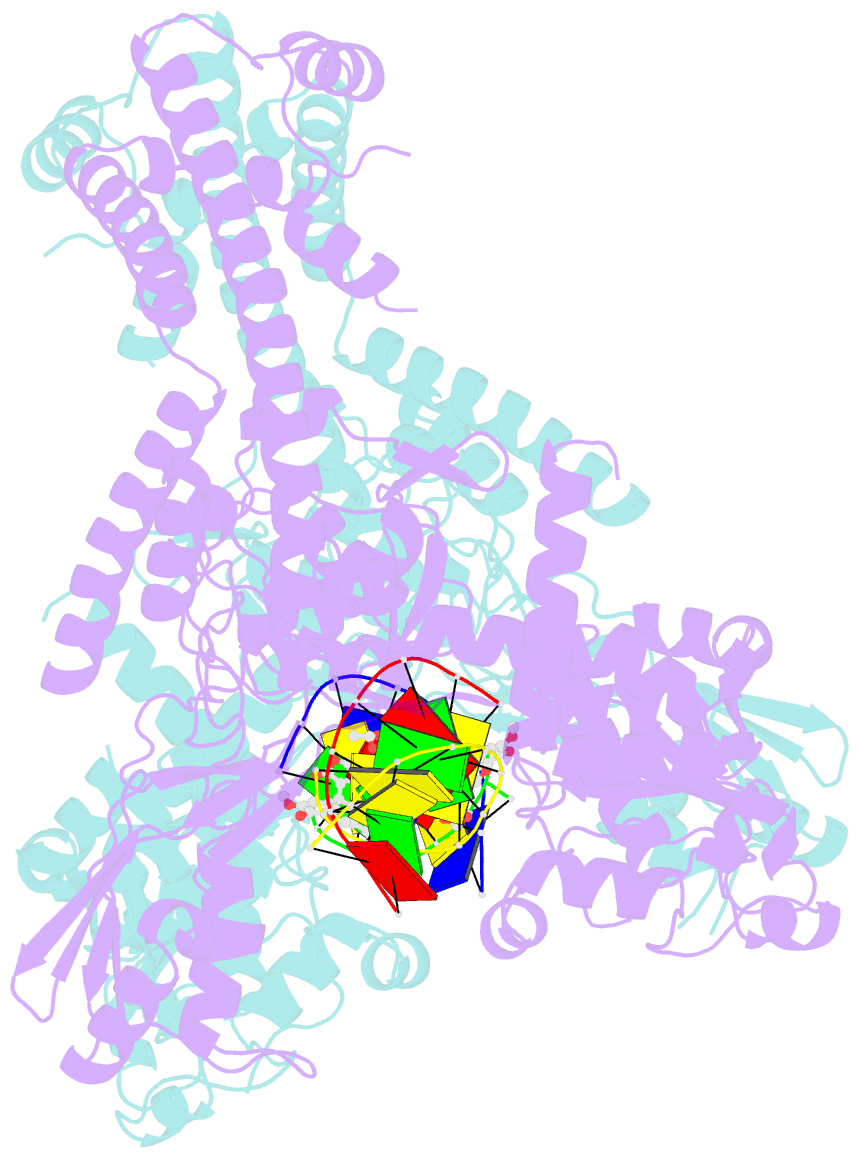
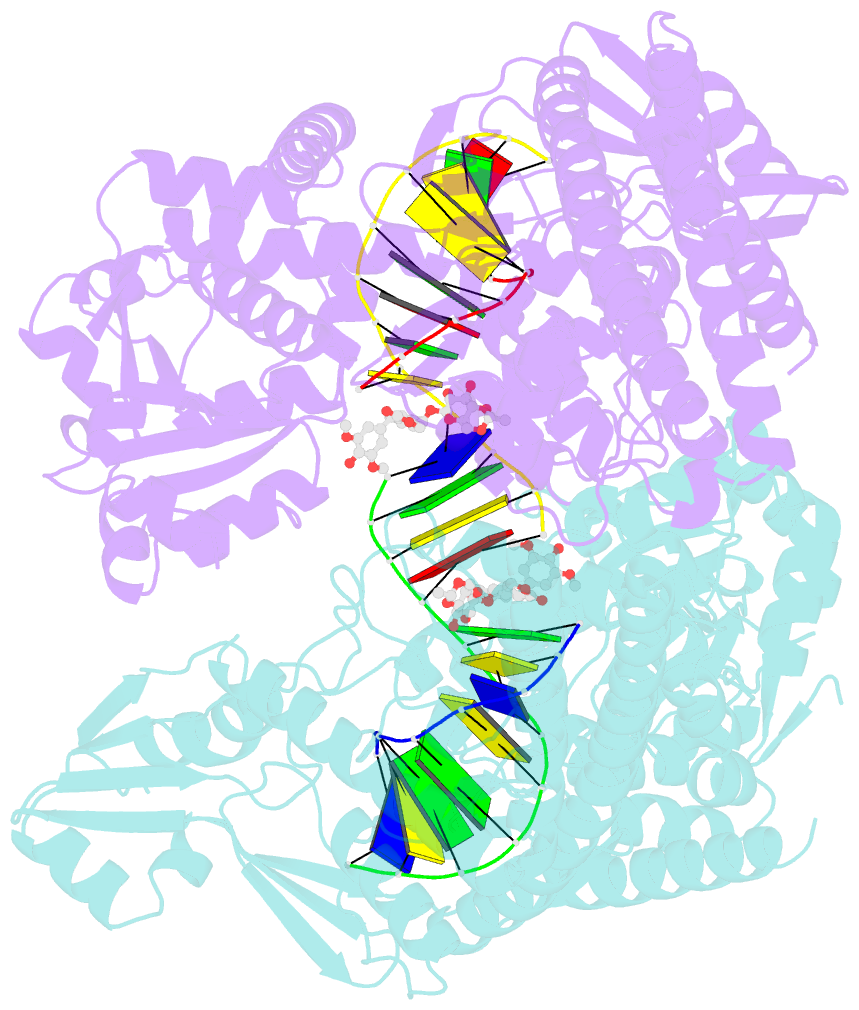
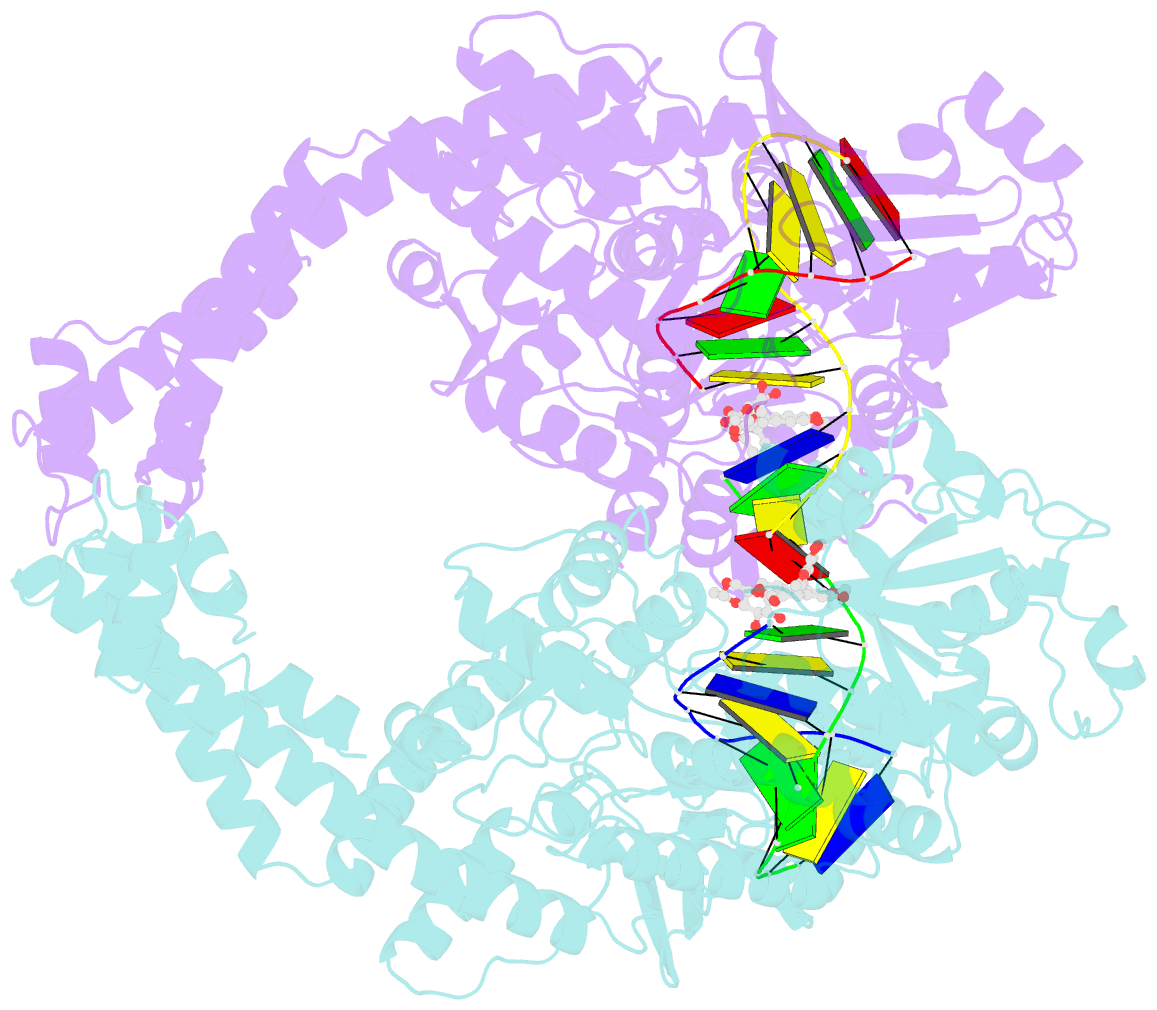
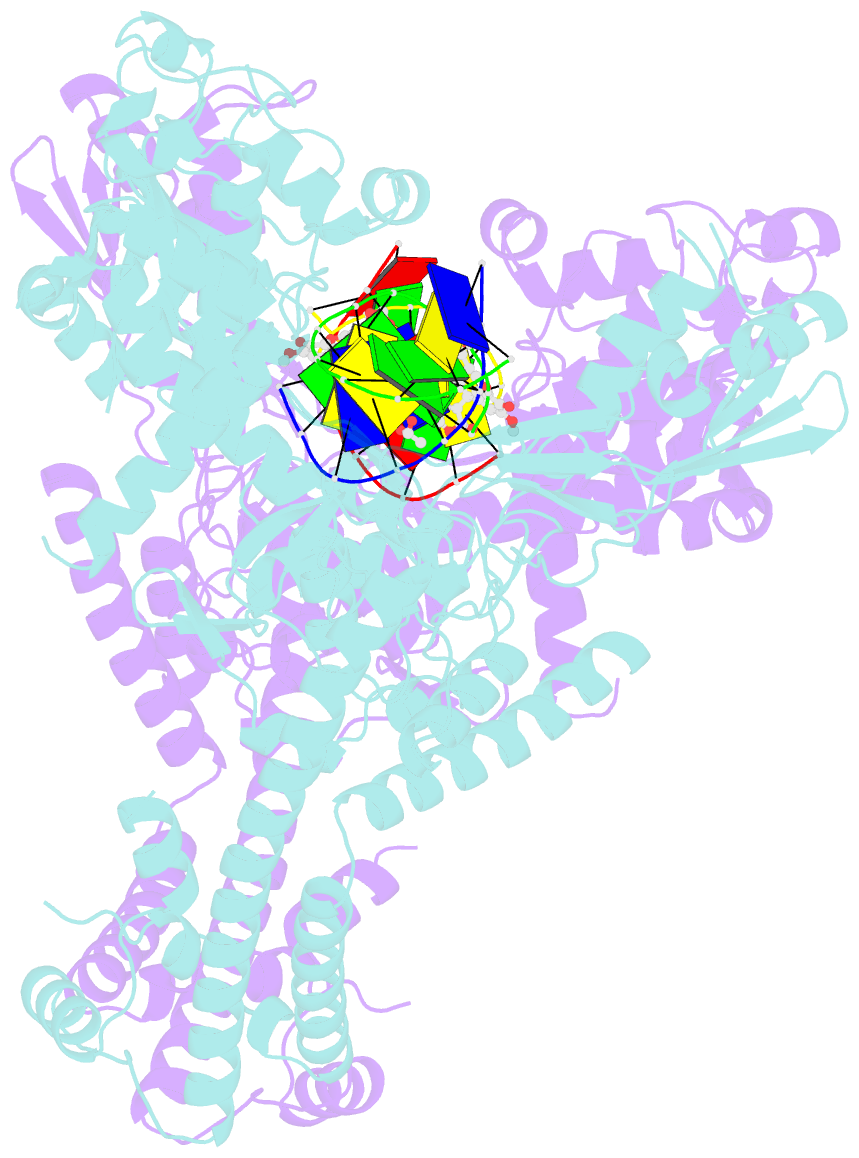
- Human topoisomerase iibeta in complex with DNA and etoposide
- Reference
- Wu CC, Li TK, Farh L, Lin LY, Lin TS, Yu YJ, Yen TJ, Chiang CW, Chan NL (2011): "Structural basis of type II topoisomerase inhibition by the anticancer drug etoposide." Science, 333, 459-462. doi: 10.1126/science.1204117.
- Abstract
- Type II topoisomerases (TOP2s) resolve the topological problems of DNA by transiently cleaving both strands of a DNA duplex to form a cleavage complex through which another DNA segment can be transported. Several widely prescribed anticancer drugs increase the population of TOP2 cleavage complex, which leads to TOP2-mediated chromosome DNA breakage and death of cancer cells. We present the crystal structure of a large fragment of human TOP2β complexed to DNA and to the anticancer drug etoposide to reveal structural details of drug-induced stabilization of a cleavage complex. The interplay between the protein, the DNA, and the drug explains the structure-activity relations of etoposide derivatives and the molecular basis of drug-resistant mutations. The analysis of protein-drug interactions provides information applicable for developing an isoform-specific TOP2-targeting strategy.