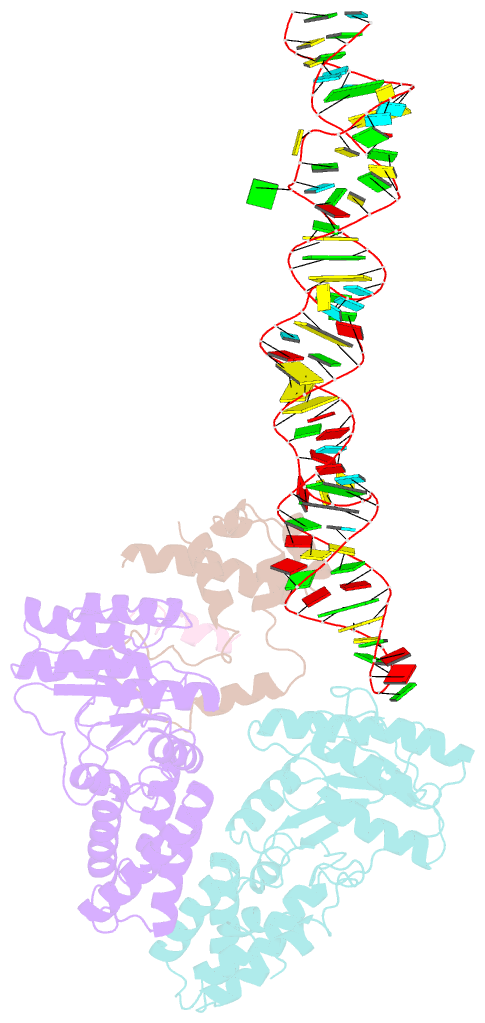
Summary information and primary citation
- PDB-id
- 3zn8; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- protein transport
- Method
- cryo-EM (12.0 Å)
- Summary
- Structural basis of signal sequence surveillance and selection by the srp-sr complex
- Reference
- Von Loeffelholz O, Knoops K, Ariosa A, Zhang X, Karuppasamy M, Huard K, Schoehn G, Berger I, Shan SO, Schaffitzel C (2013): "Structural Basis of Signal Sequence Surveillance and Selection by the Srp-Sr Complex." Nat.Struct.Mol.Biol., 20, 604. doi: 10.1038/NSMB.2546.
- Abstract
- Signal-recognition particle (SRP)-dependent targeting of translating ribosomes to membranes is a multistep quality-control process. Ribosomes that are translating weakly hydrophobic signal sequences can be rejected from the targeting reaction even after they are bound to the SRP. Here we show that the early complex, formed by Escherichia coli SRP and its receptor FtsY with ribosomes translating the incorrect cargo EspP, is unstable and rearranges inefficiently into subsequent conformational states, such that FtsY dissociation is favored over successful targeting. The N-terminal extension of EspP is responsible for these defects in the early targeting complex. The cryo-electron microscopy structure of this 'false' early complex with EspP revealed an ordered M domain of SRP protein Ffh making two ribosomal contacts, and the NG domains of Ffh and FtsY forming a distorted, flexible heterodimer. Our results provide a structural basis for SRP-mediated signal-sequence selection during recruitment of the SRP receptor.