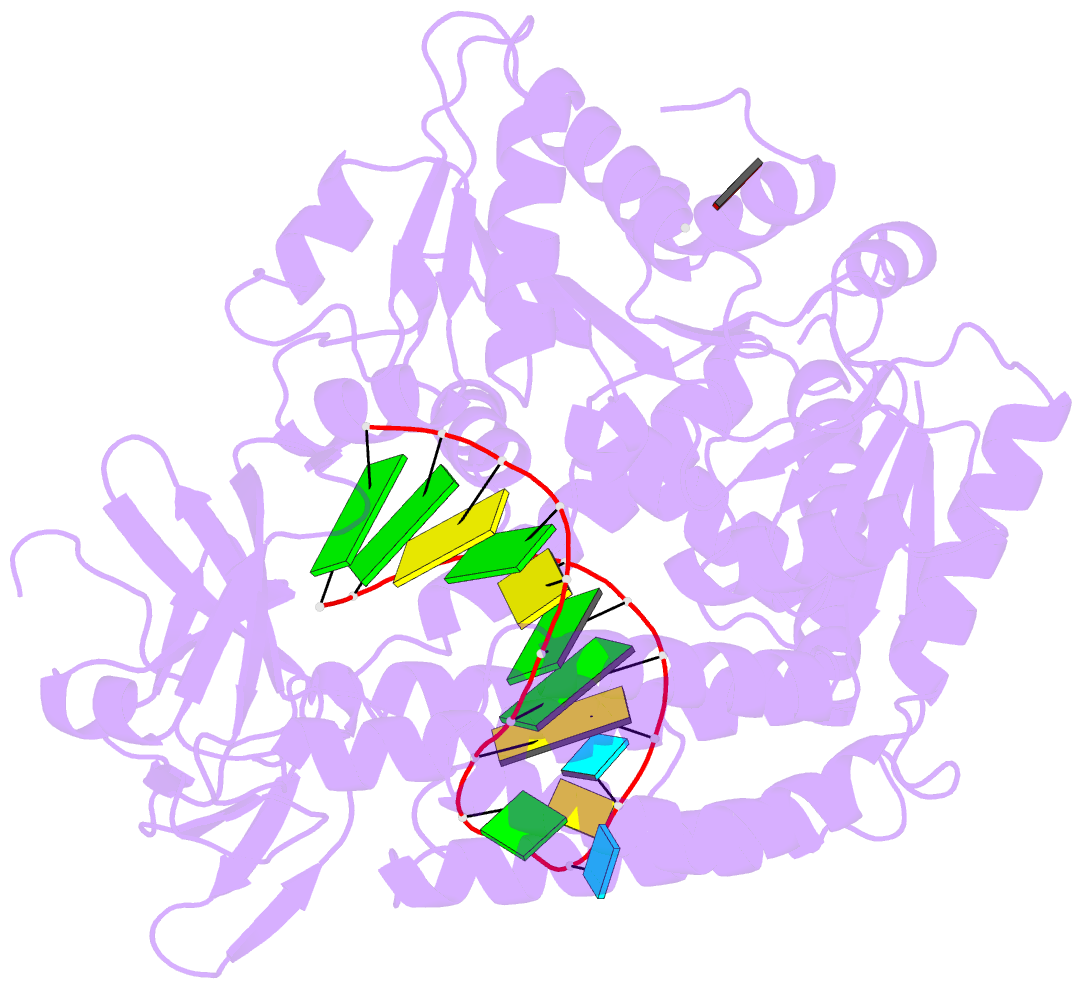
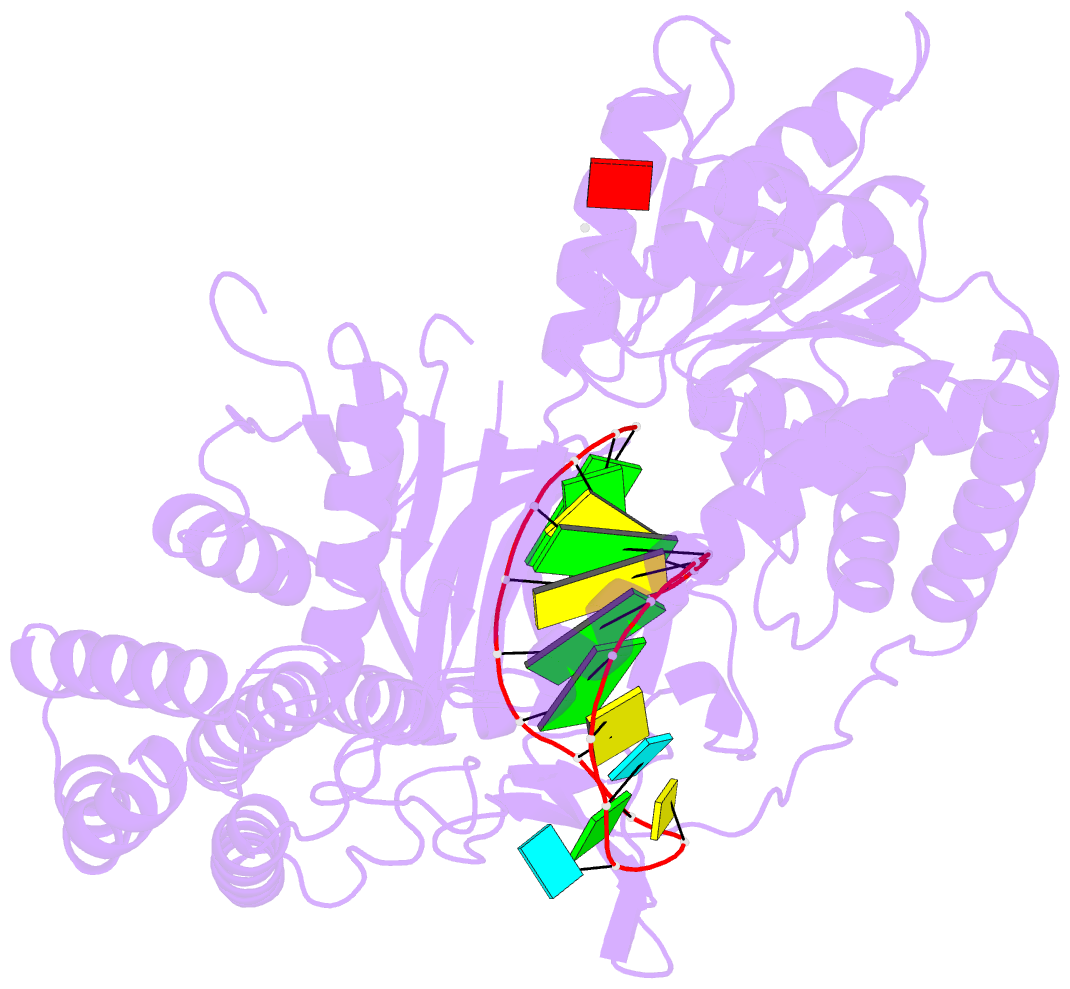
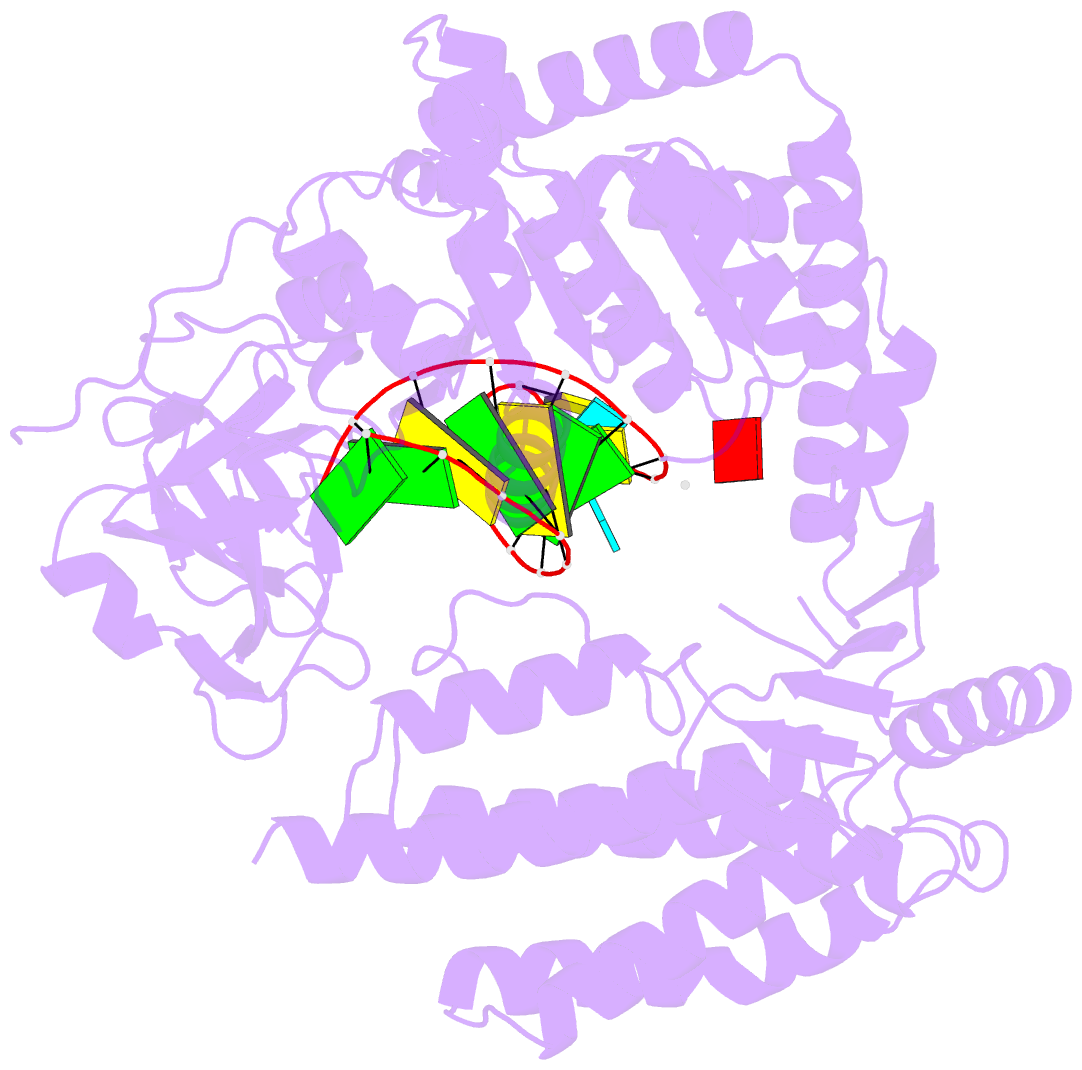
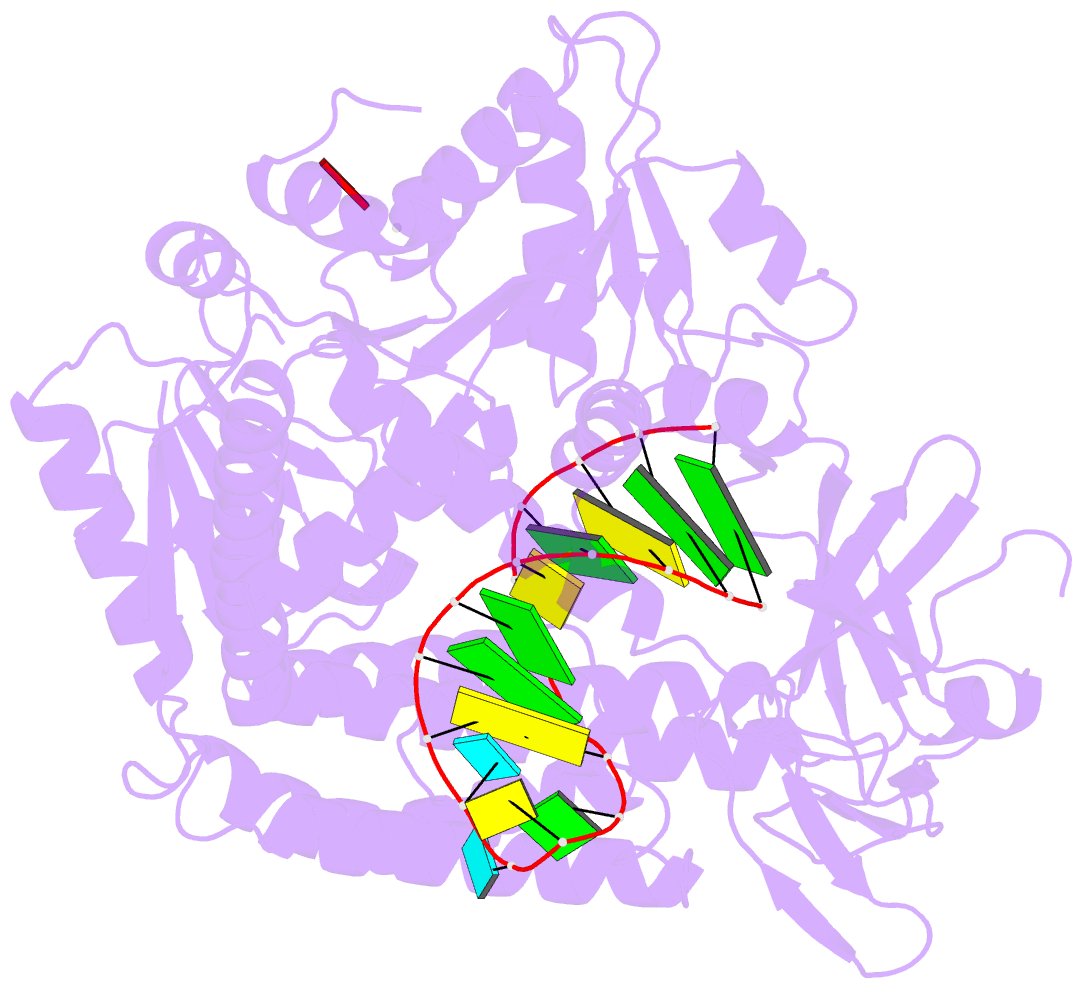
Summary information and primary citation
- PDB-id
- 4ay2; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- hydrolase-RNA
- Method
- X-ray (2.8 Å)
- Summary
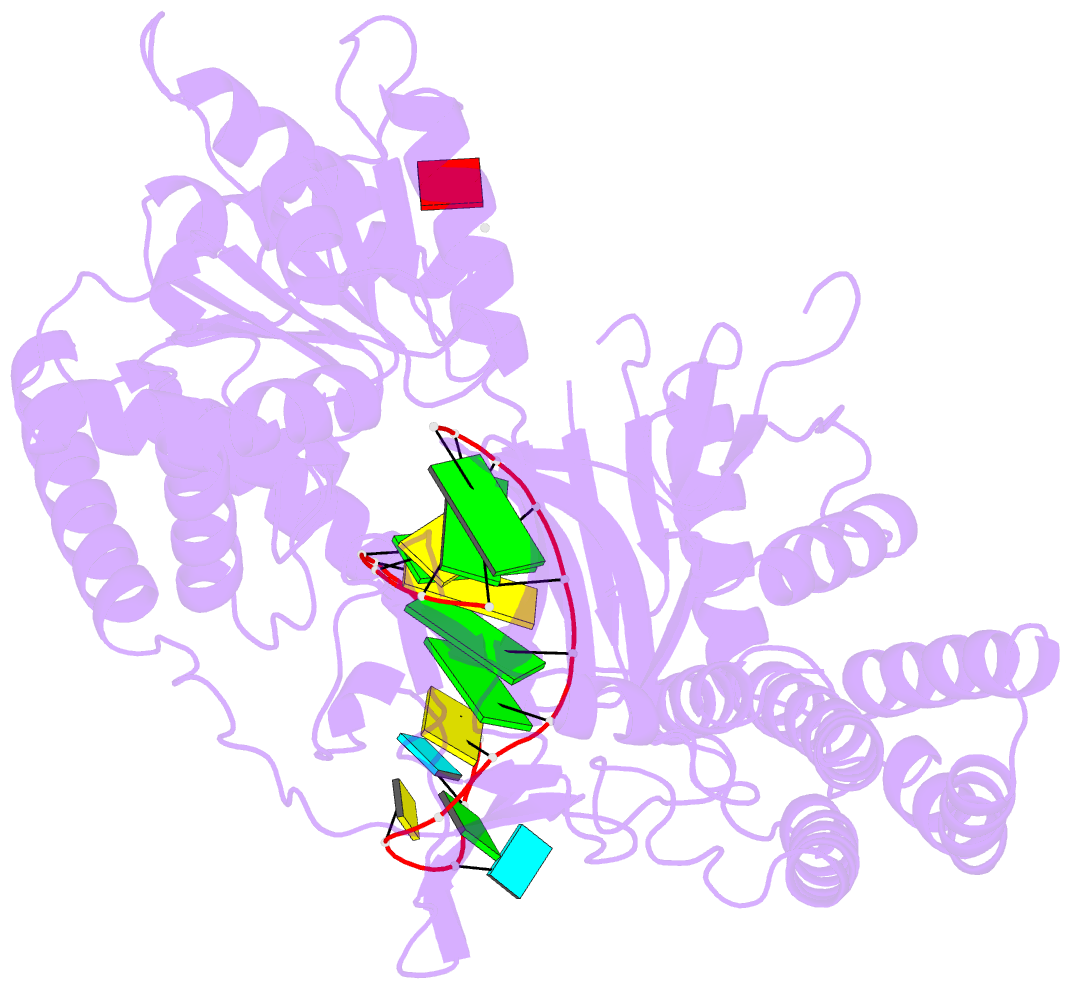
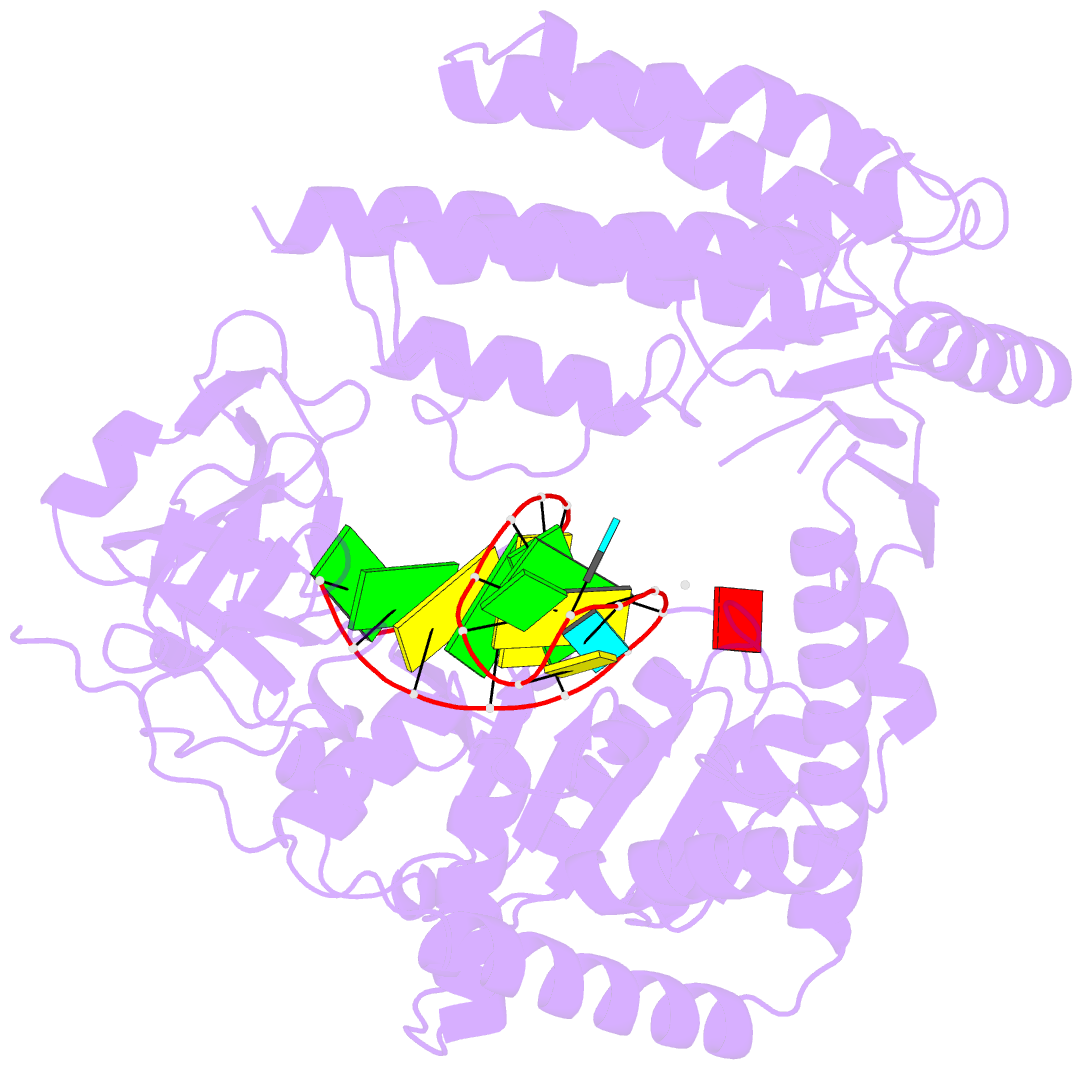
- Capturing 5' tri-phosphorylated RNA duplex by rig-i
- Reference
- Luo D, Kohlway A, Vela A, Pyle AM (2012): "Visualizing the Determinants of Viral RNA Recognition by Innate Immune Sensor Rig-I." Structure, 20, 1983. doi: 10.1016/J.STR.2012.08.029.
- Abstract
- Retinoic acid inducible gene-I (RIG-I) is a key intracellular immune receptor for pathogenic RNAs, particularly from RNA viruses. Here, we report the crystal structure of human RIG-I bound to a 5' triphosphorylated RNA hairpin and ADP nucleotide at 2.8 Å resolution. The RNA ligand contains all structural features that are essential for optimal recognition by RIG-I, as it mimics the panhandle-like signatures within the genome of negative-stranded RNA viruses. RIG-I adopts an intermediate, semiclosed conformation in this product state of ATP hydrolysis. The structure of this complex allows us to visualize the first steps in RIG-I recognition and activation upon viral infection.