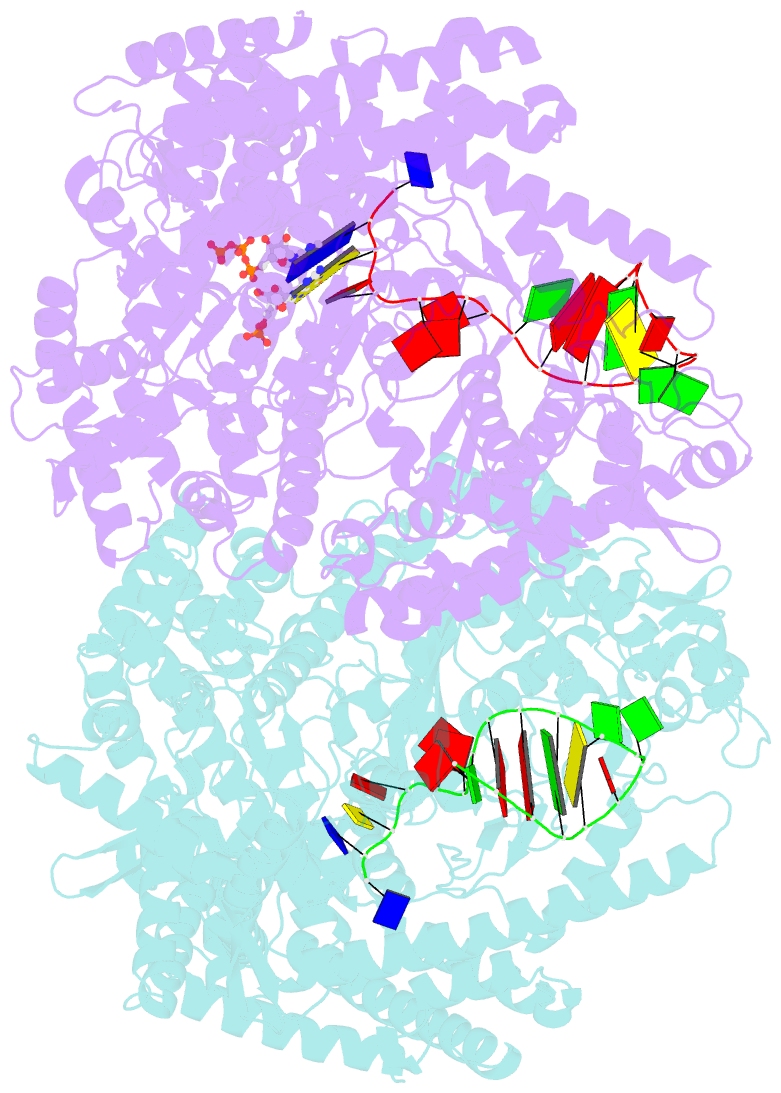
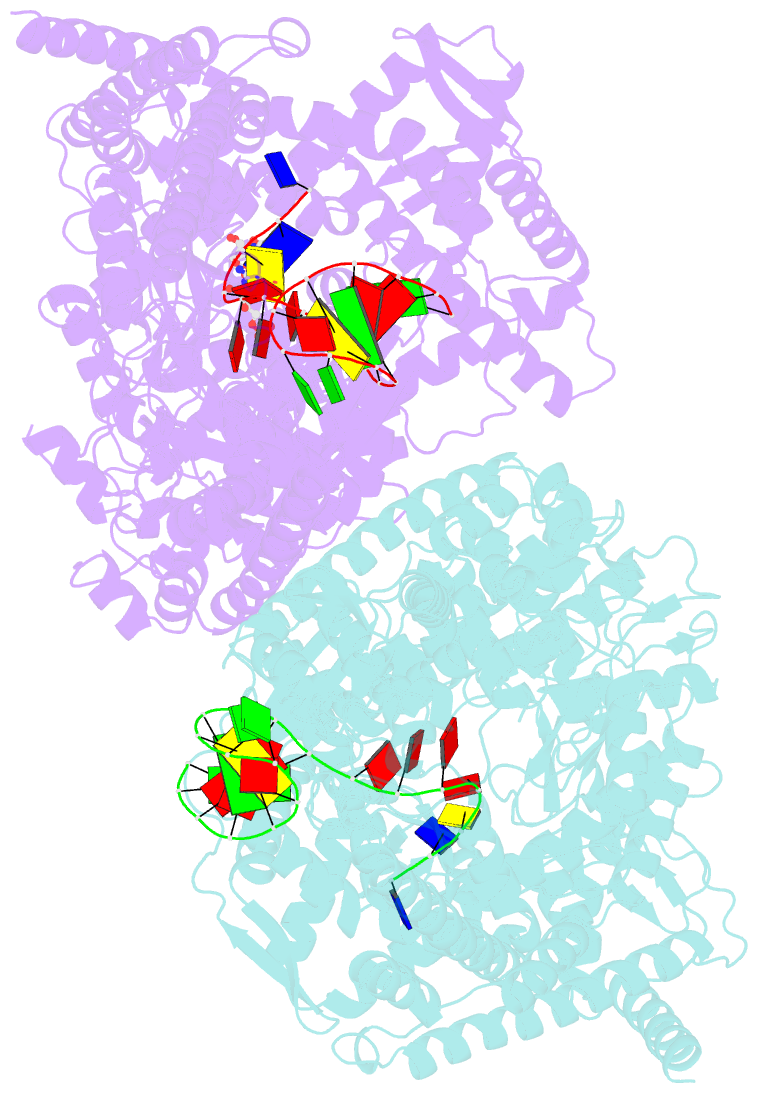
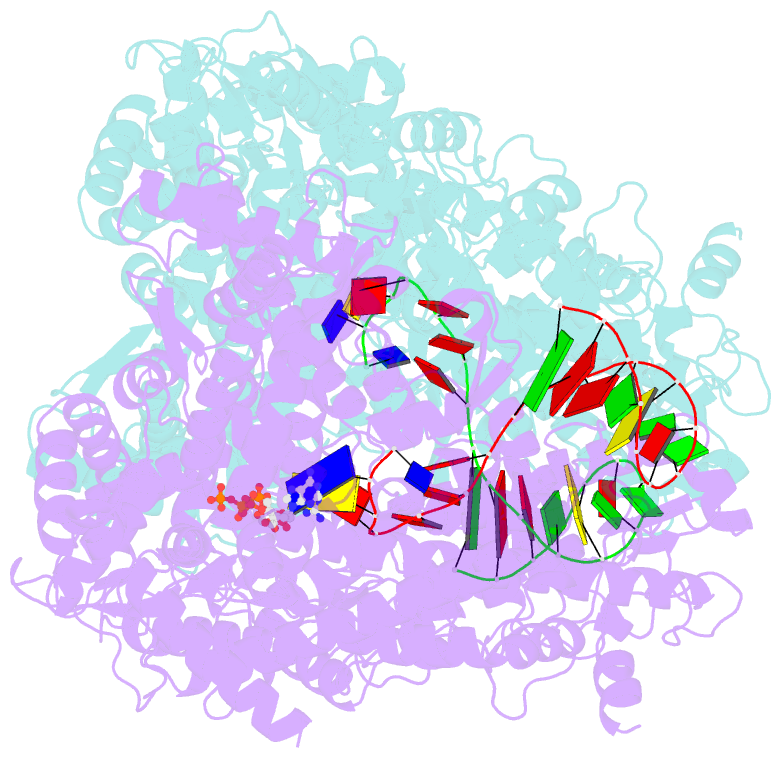
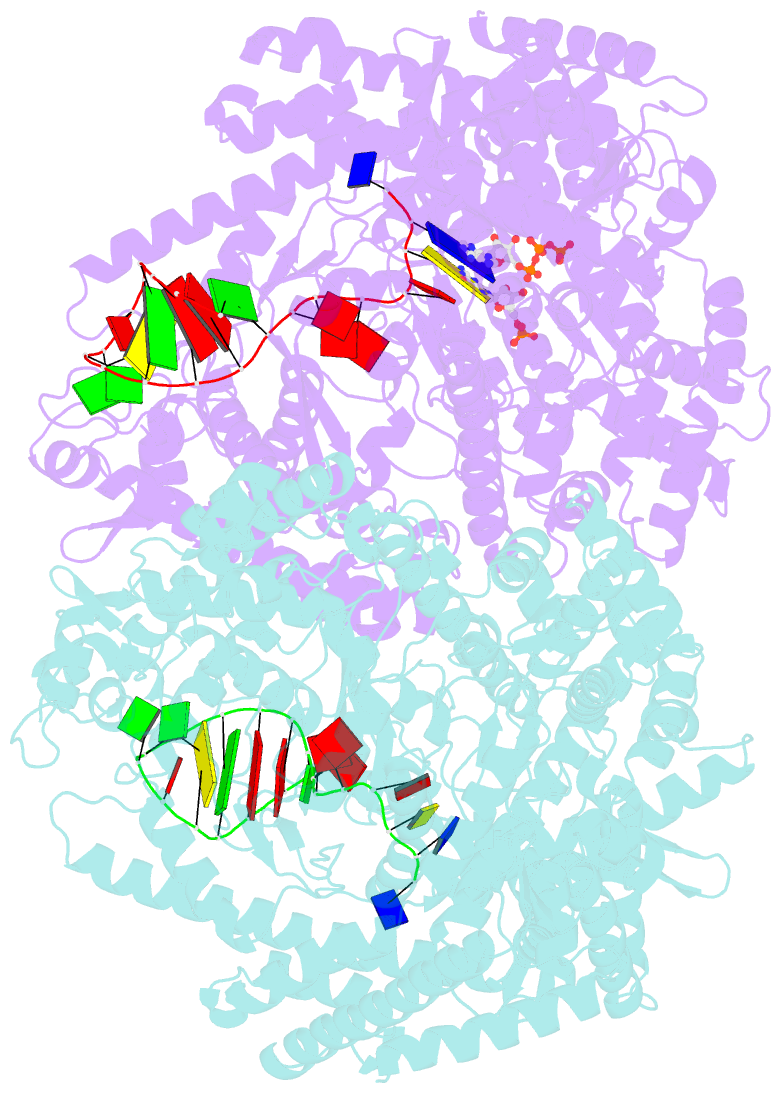
Summary information and primary citation
- PDB-id
-
4ff2;
DSSR-derived features in text and
JSON formats; DNAproDB
- Class
- transferase-DNA
- Method
- X-ray (2.0 Å)
- Summary
- N4 mini-vrnap transcription initiation complex, 2 min
after soaking gtp, atp and mn
- Reference
-
Basu RS, Murakami KS (2013): "Watching
the Bacteriophage N4 RNA Polymerase Transcription by
Time-dependent Soak-trigger-freeze X-ray
Crystallography." J.Biol.Chem.,
288, 3305-3311. doi: 10.1074/jbc.M112.387712.
- Abstract
- The challenge for structural biology is to understand
atomic-level macromolecular motions during enzymatic
reaction. X-ray crystallography can reveal high resolution
structures; however, one perceived limitation is that it
reveals only static views. Here we use time-dependent
soak-trigger-freeze X-ray crystallography, namely, soaking
nucleotide and divalent metal into the bacteriophage RNA
polymerase (RNAP)-promoter DNA complex crystals to trigger
the nucleotidyl transfer reaction and freezing crystals at
different time points, to capture real-time intermediates
in the pathway of transcription. In each crystal structure,
different intensities and shapes of electron density maps
corresponding to the nucleotide and metal were revealed at
the RNAP active site which allow watching the nucleotide
and metal bindings and the phosphodiester bond formation in
a time perspective. Our study provides the temporal order
of substrate assembly and metal co-factor binding at the
active site of enzyme which completes our understanding of
the two-metal-ion mechanism and fidelity mechanism in
single-subunit RNAPs. The nucleotide-binding metal (Me(B))
is coordinated at the active site prior to the catalytic
metal (Me(A)). Me(A) coordination is only temporal,
established just before and dissociated immediately after
phosphodiester bond formation. We captured these elusive
intermediates exploiting the slow enzymatic reaction in
crystallo. These results demonstrate that the simple
time-dependent soak-trigger-freeze X-ray crystallography
offers a direct means for monitoring enzymatic
reactions.