Summary information and primary citation
- PDB-id
- 4iqv; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transferase-transferase inhibitor-DNA
- Method
- X-ray (2.9 Å)
- Summary
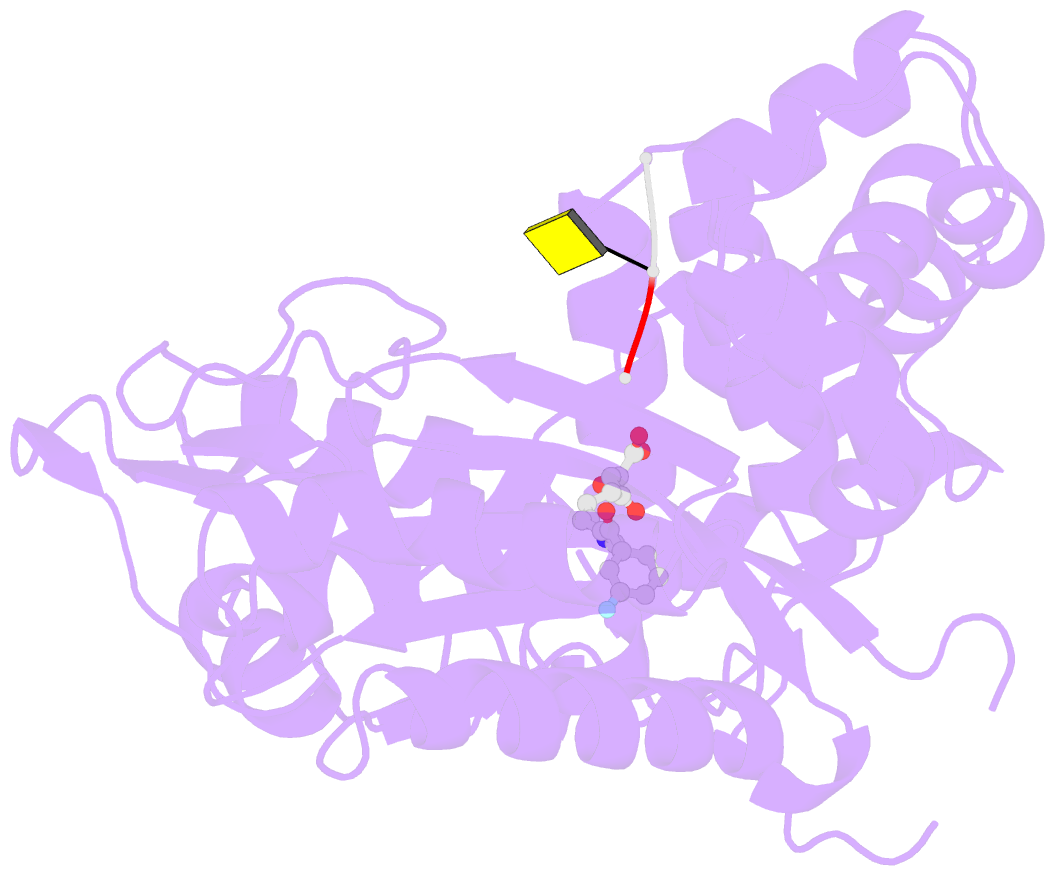
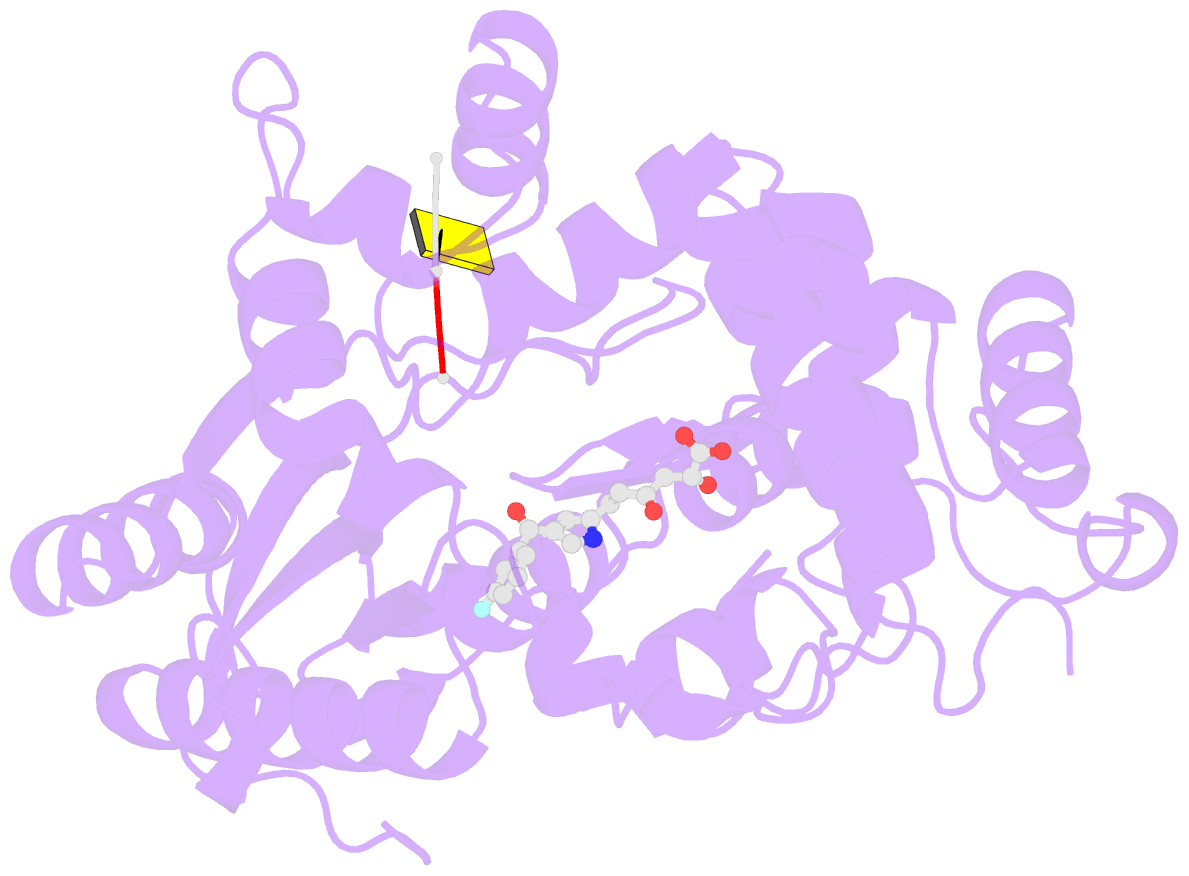
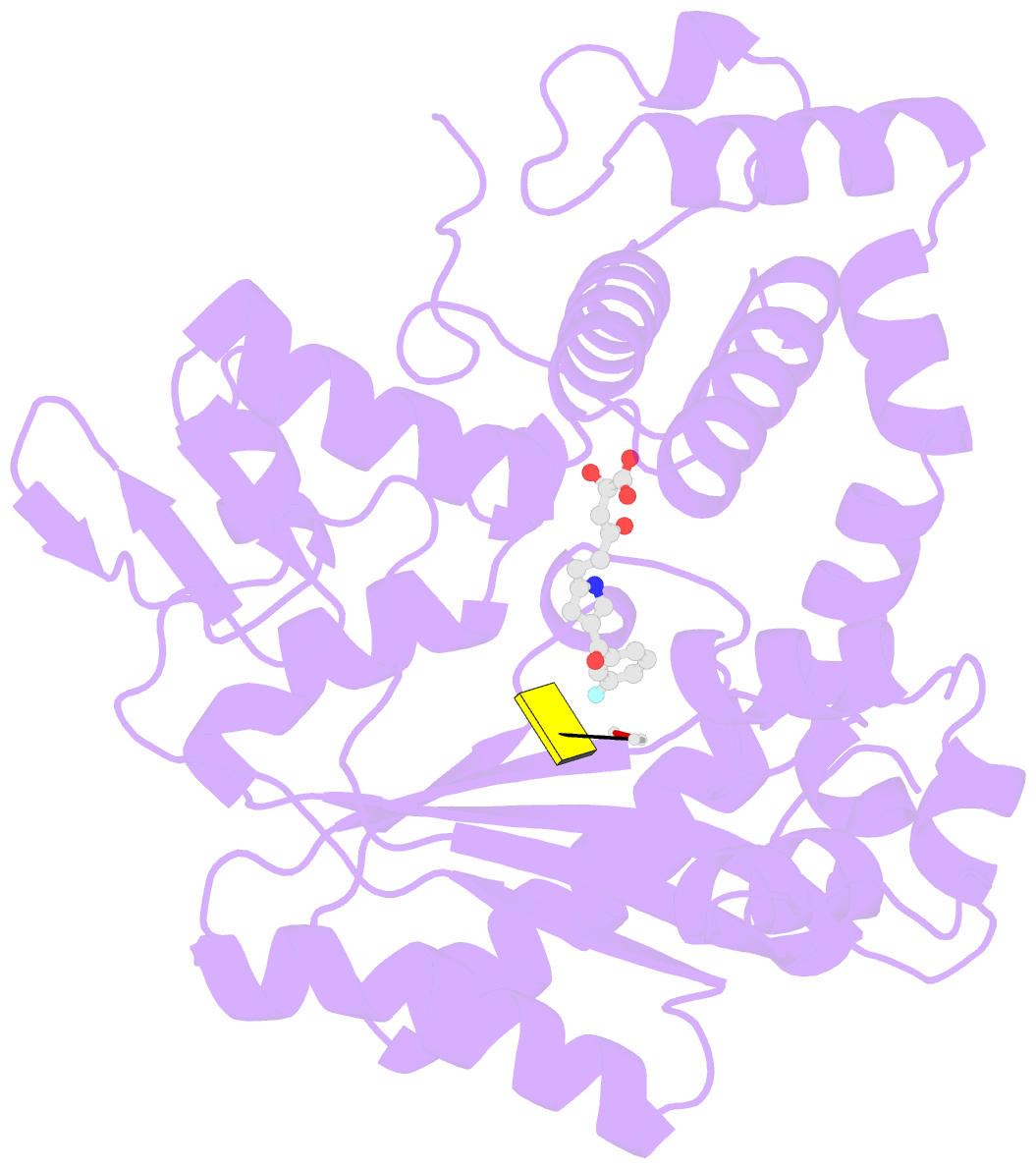
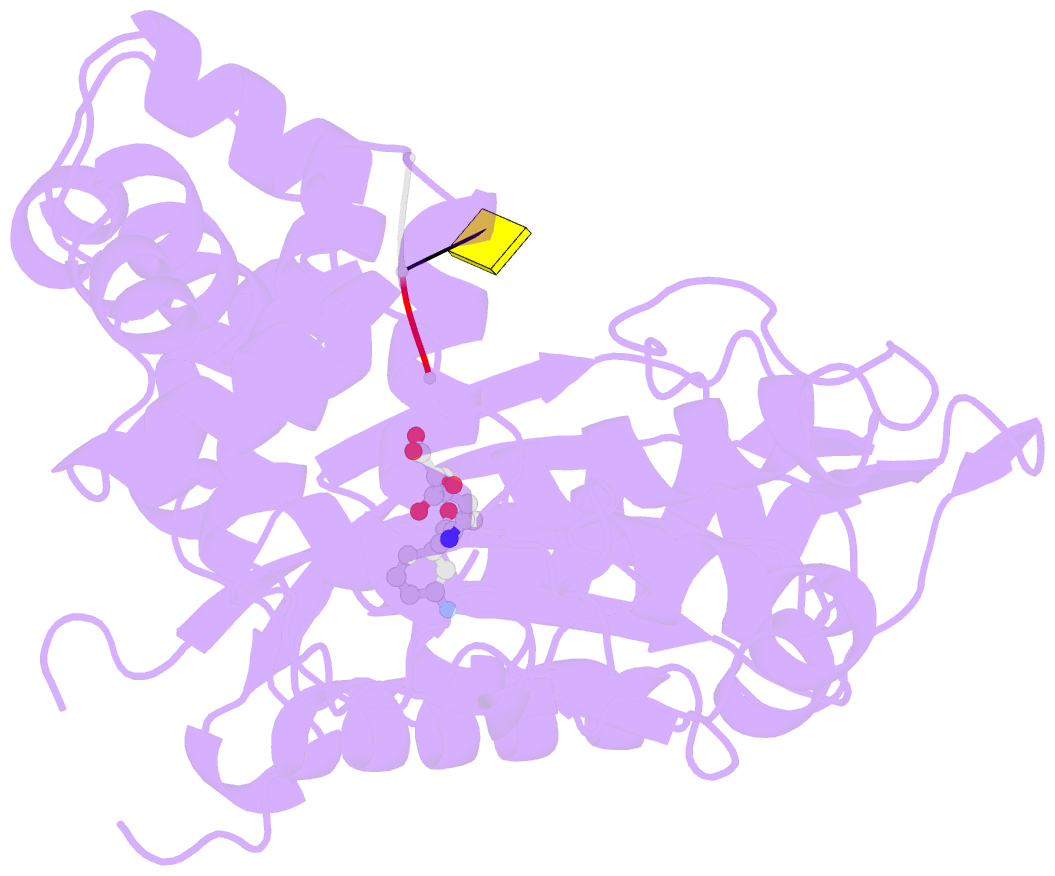
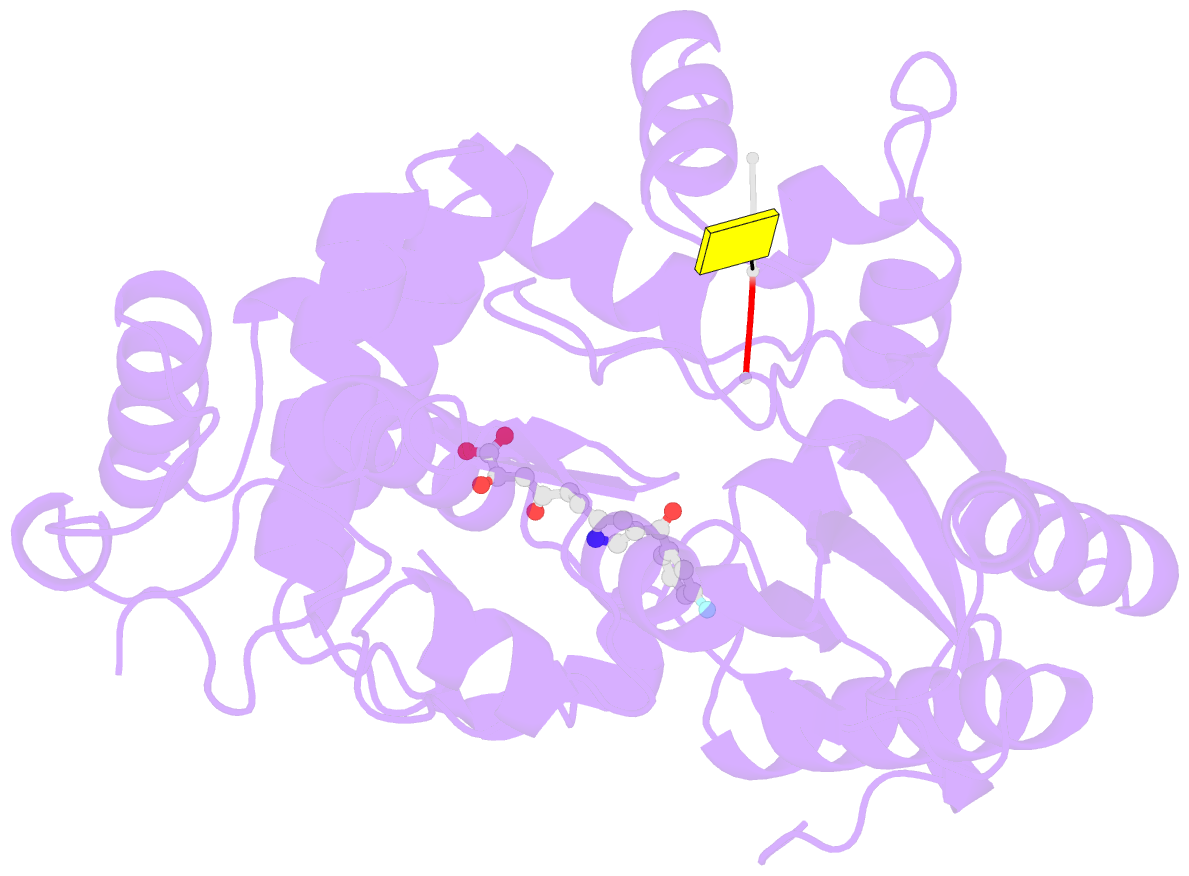
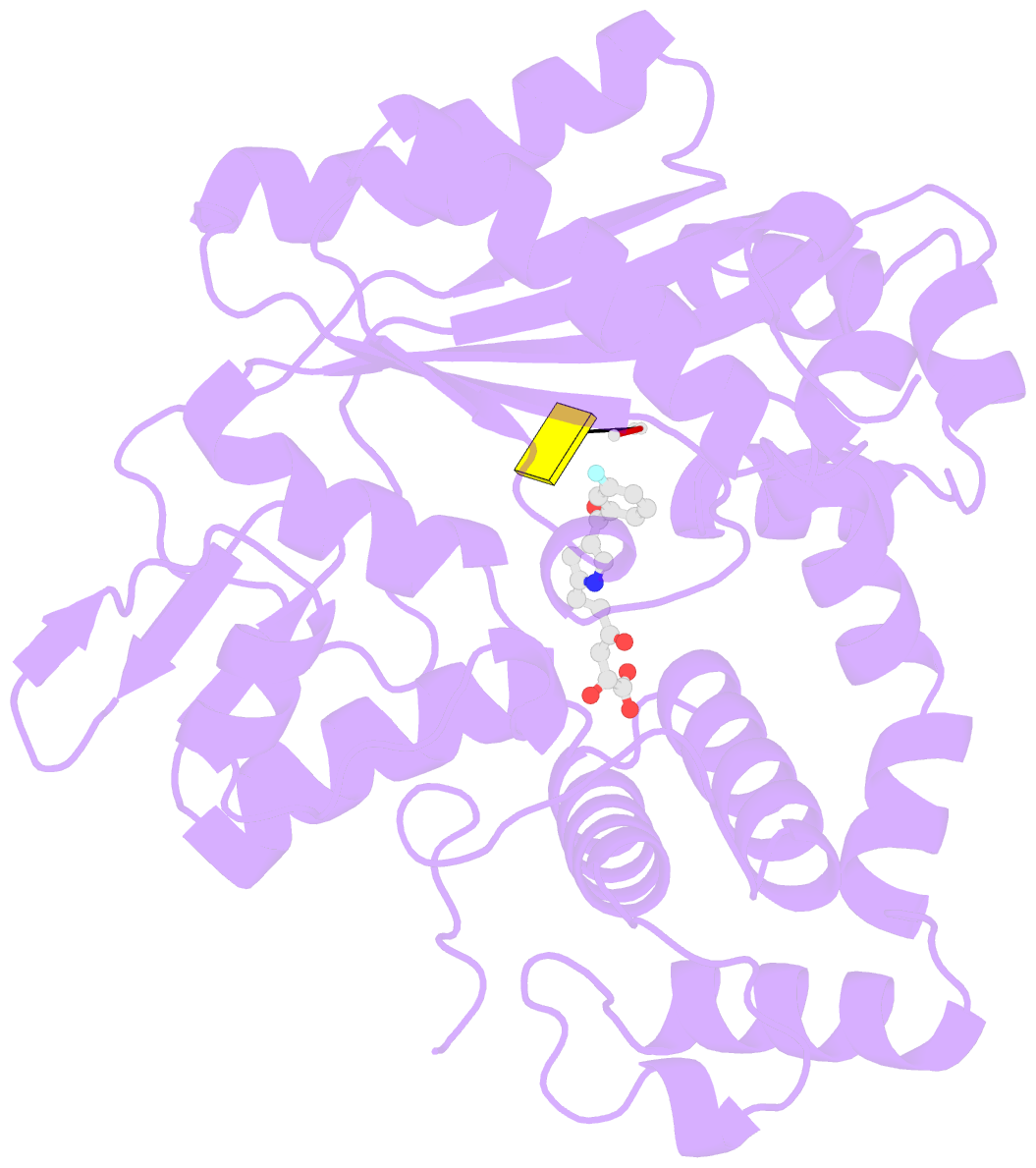
- Tdt core in complex with inhibitor 6-[4-(3-fluorobenzoyl)-1h-pyrrol-2-yl]-2-hydroxy-4-oxohexa-2,5-dienoic acid and ssDNA
- Reference
- Costi R, Cuzzucoli Crucitti G, Pescatori L, Messore A, Scipione L, Tortorella S, Amoroso A, Crespan E, Campiglia P, Maresca B, Porta A, Granata I, Novellino E, Gouge J, Delarue M, Maga G, Di Santo R (2013): "New nucleotide-competitive non-nucleoside inhibitors of terminal deoxynucleotidyl transferase: discovery, characterization, and crystal structure in complex with the target." J.Med.Chem., 56, 7431-7441. doi: 10.1021/jm4010187.
- Abstract
- Terminal deoxynucletidyl transferase (TdT) is overexpressed in some cancer types, where it might compete with pol μ during the mutagenic repair of double strand breaks (DSBs) through the nonhomologous end joining (NHEJ) pathway. Here we report the discovery and characterization of pyrrolyl and indolyl diketo acids that specifically target TdT and behave as nucleotide-competitive inhibitors. These compounds show a selective toxicity toward MOLT-4 compared to HeLa cells that correlate well with in vitro selectivity for TdT. The binding site of two of these inhibitors was determined by cocrystallization with TdT, explaining why these compounds are competitive inhibitors of the deoxynucleotide triphosphate (dNTP). In addition, because of the observed dual localization of the phenyl substituent, these studies open the possibility of rationally designing more potent compounds.