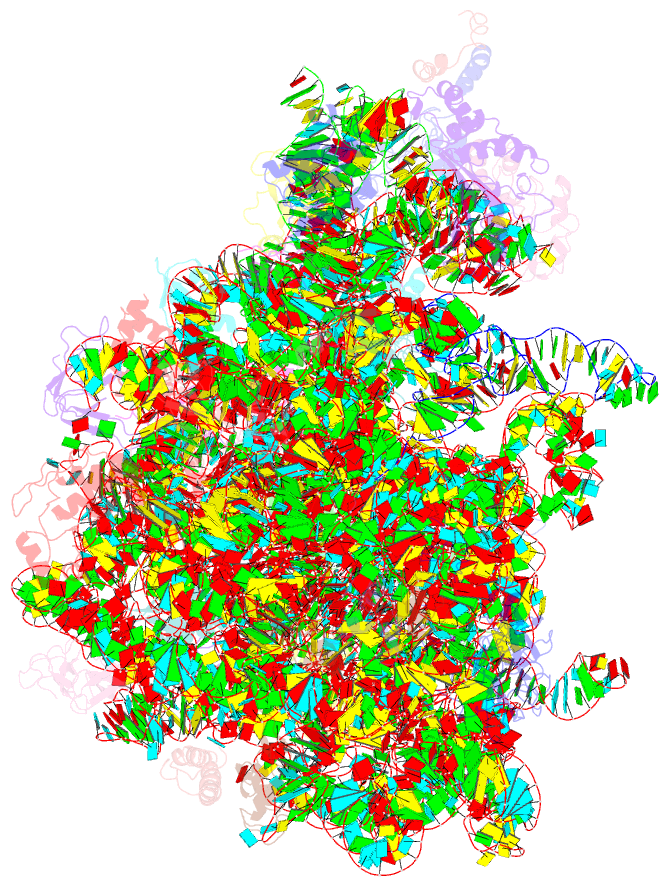
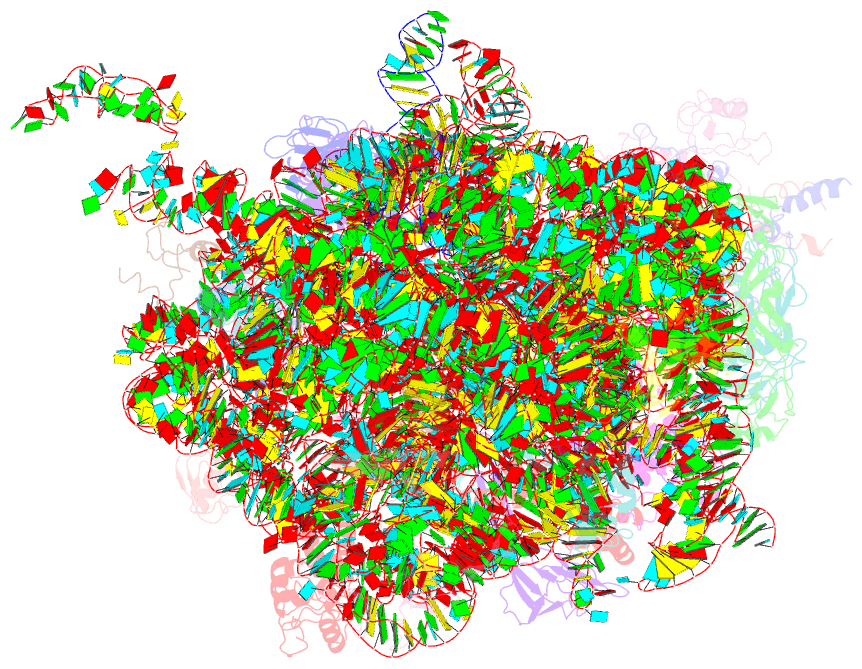
Summary information and primary citation
- PDB-id
- 4uy8; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- ribosome
- Method
- cryo-EM (3.8 Å)
- Summary
- Molecular basis for the ribosome functioning as a l-tryptophan sensor - cryo-EM structure of a tnac stalled e.coli ribosome
- Reference
- Bischoff L, Berninghausen O, Beckmann R (2014): "Molecular Basis for the Ribosome Functioning as an L-Tryptophan Sensor." Cell Rep., 9, 469. doi: 10.1016/J.CELREP.2014.09.011.
- Abstract
- Elevated levels of the free amino acid L-tryptophan (L-Trp) trigger expression of the tryptophanase tnaCAB operon in E. coli. Activation depends on tryptophan-dependent ribosomal stalling during translation of the upstream TnaC peptide. Here, we present a cryoelectron microscopy (cryo-EM) reconstruction at 3.8 Å resolution of a ribosome stalled by the TnaC peptide. Unexpectedly, we observe two L-Trp molecules in the ribosomal exit tunnel coordinated within composite hydrophobic pockets formed by the nascent TnaC peptide and the tunnel wall. As a result, the peptidyl transferase center (PTC) adopts a distinct conformation that precludes productive accommodation of release factor 2 (RF2), thereby inducing translational stalling. Collectively, our results demonstrate how the translating ribosome can act as a small molecule sensor for gene regulation.