Summary information and primary citation
- PDB-id
- 4zq9; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- DNA binding protein-DNA
- Method
- X-ray (2.6 Å)
- Summary
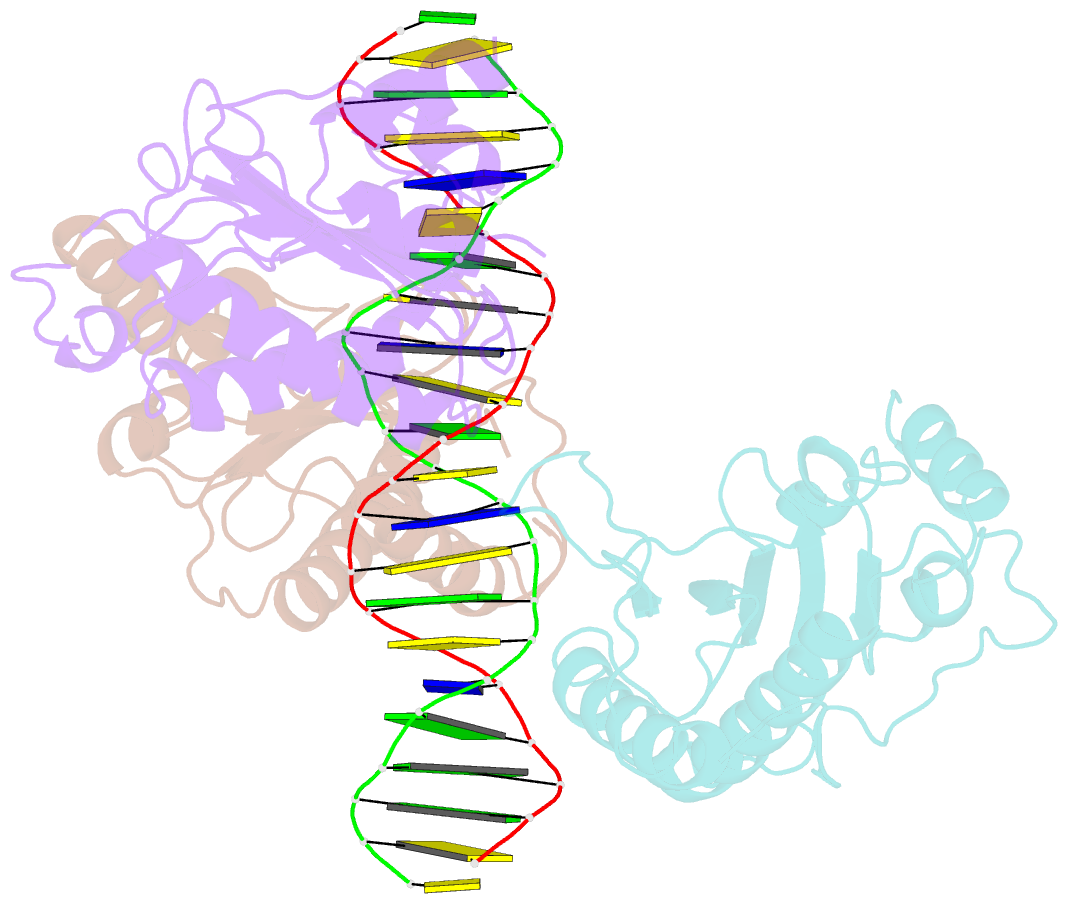
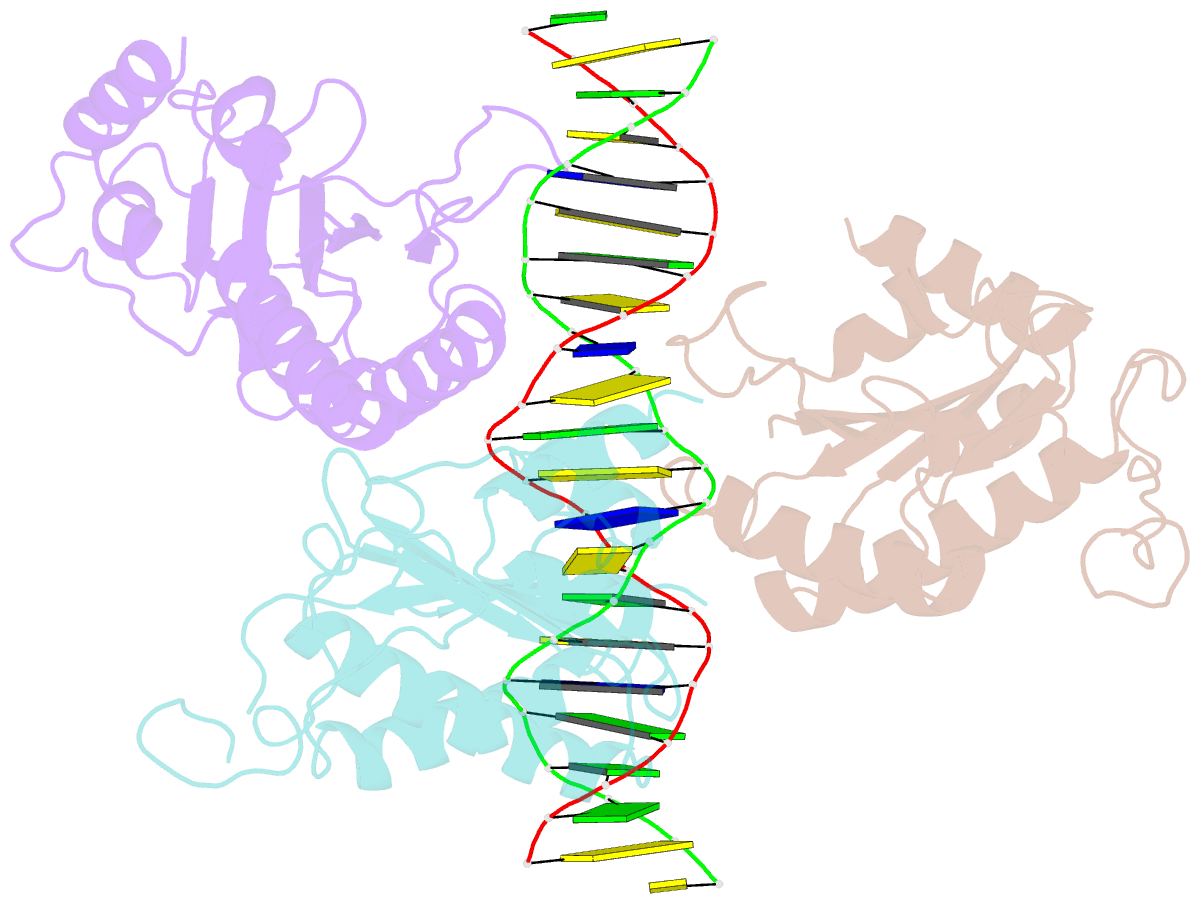
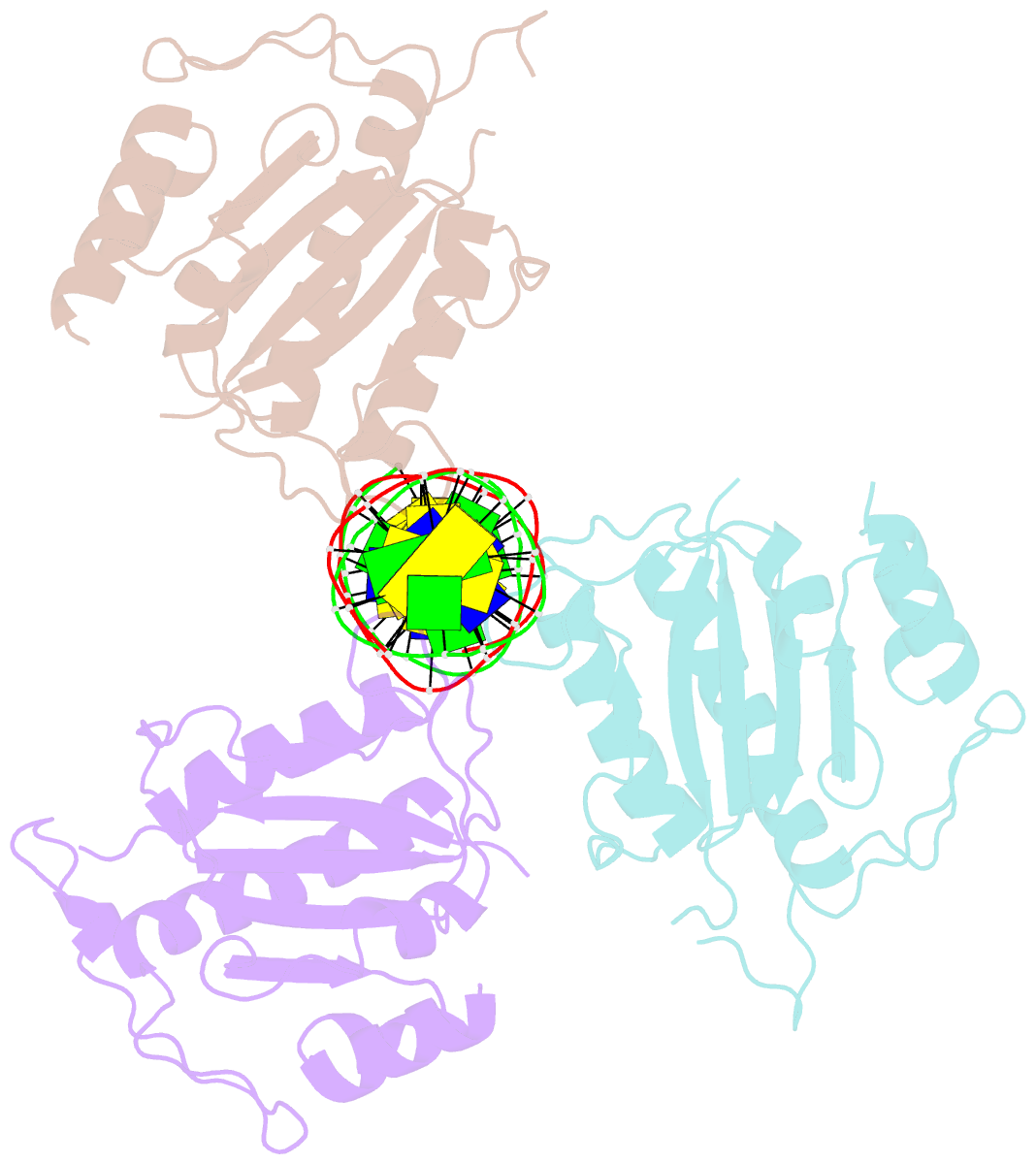
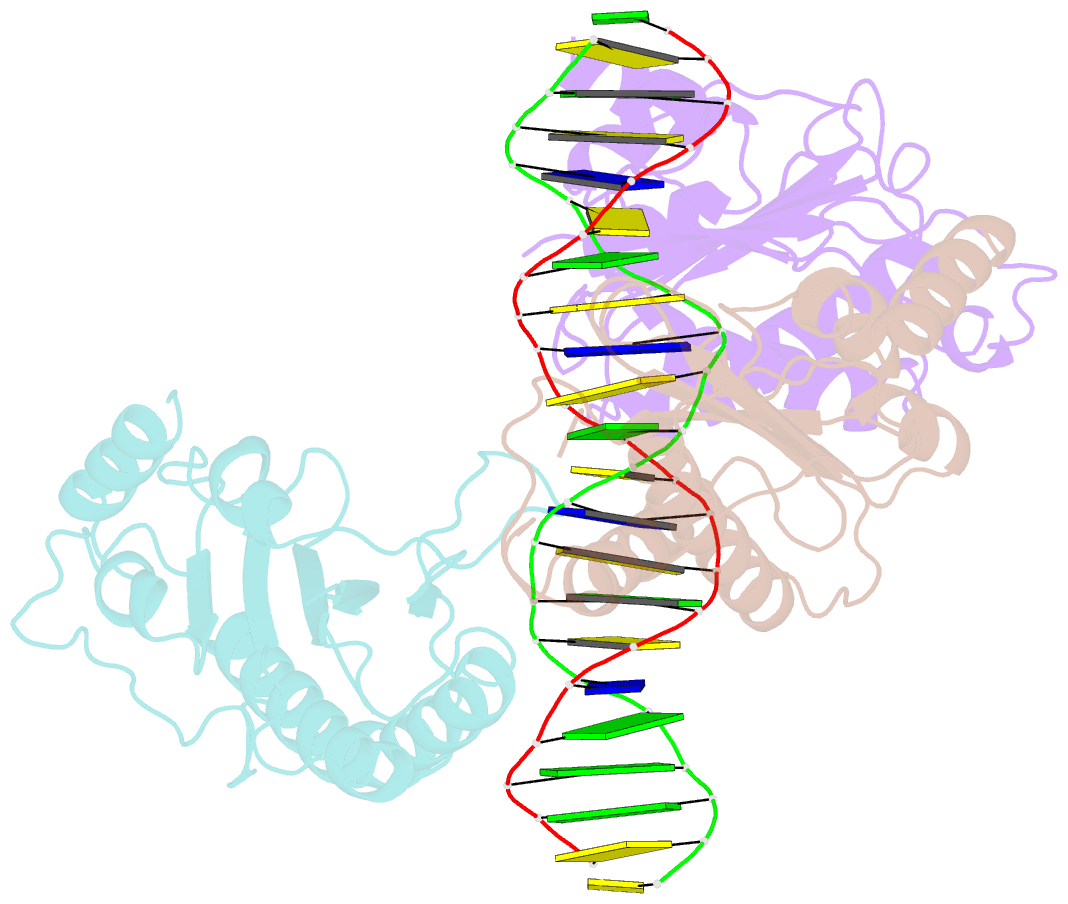
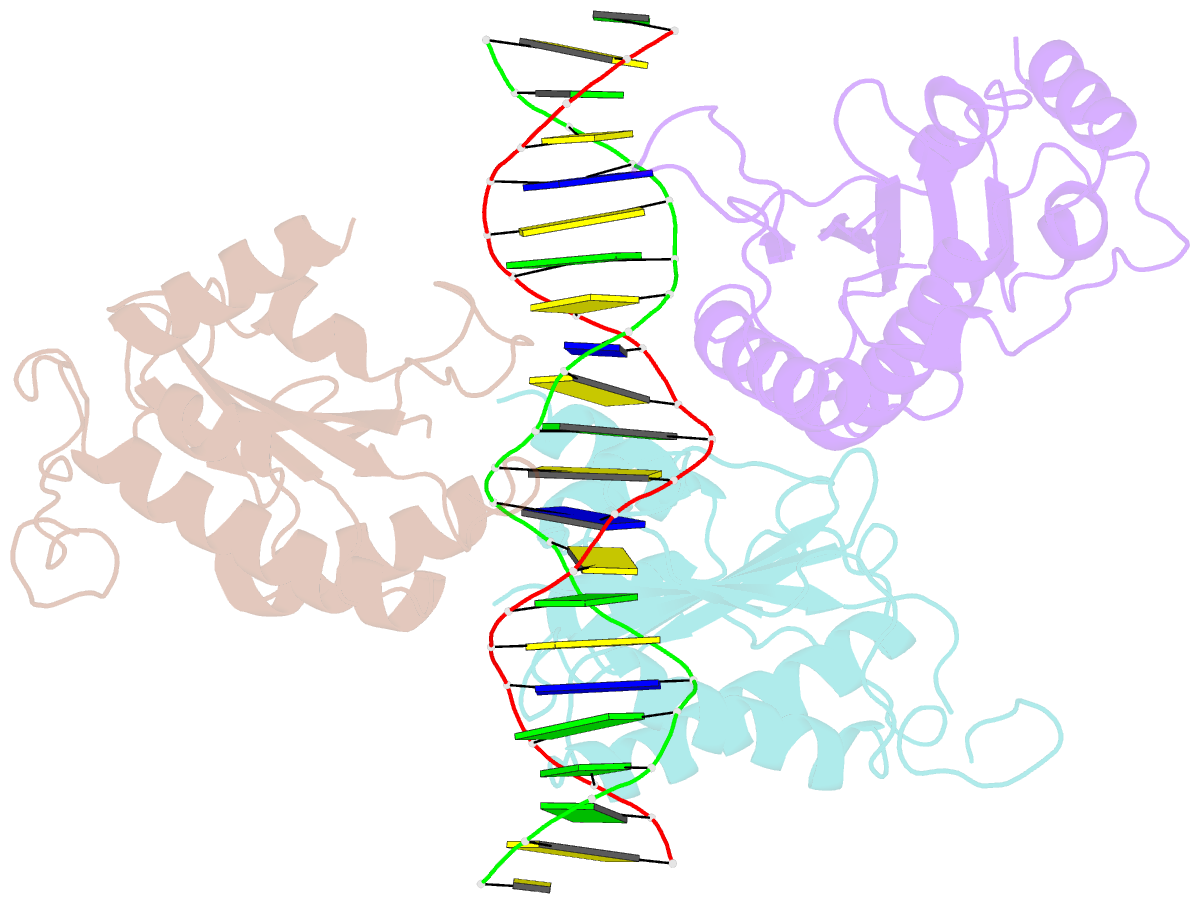
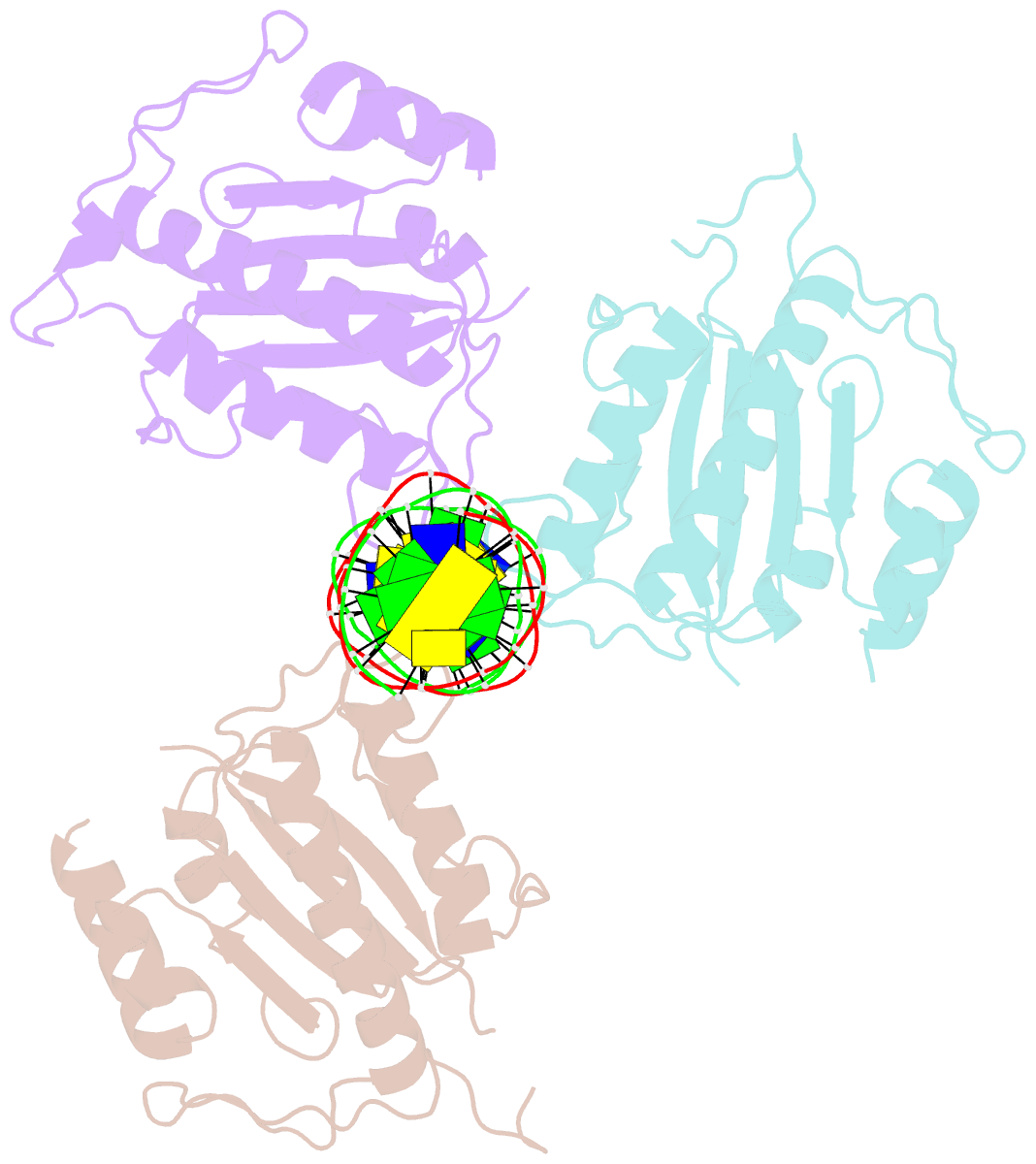
- X-ray structure of aav-2 obd bound to aavs1 site 3:1
- Reference
- Musayev FN, Zarate-Perez F, Bishop C, Burgner JW, Escalante CR (2015): "Structural Insights into the Assembly of the Adeno-associated Virus Type 2 Rep68 Protein on the Integration Site AAVS1." J.Biol.Chem., 290, 27487-27499. doi: 10.1074/jbc.M115.669960.
- Abstract
- Adeno-associated virus (AAV) is the only eukaryotic virus with the property of establishing latency by integrating site-specifically into the human genome. The integration site known as AAVS1 is located in chromosome 19 and contains multiple GCTC repeats that are recognized by the AAV non-structural Rep proteins. These proteins are multifunctional, with an N-terminal origin-binding domain (OBD) and a helicase domain joined together by a short linker. As a first step to understand the process of site-specific integration, we proceeded to characterize the recognition and assembly of Rep68 onto the AAVS1 site. We first determined the x-ray structure of AAV-2 Rep68 OBD in complex with the AAVS1 DNA site. Specificity is achieved through the interaction of a glycine-rich loop that binds the major groove and an α-helix that interacts with a downstream minor groove on the same face of the DNA. Although the structure shows a complex with three OBD molecules bound to the AAVS1 site, we show by using analytical centrifugation and electron microscopy that the full-length Rep68 forms a heptameric complex. Moreover, we determined that a minimum of two direct repeats is required to form a stable complex and to melt DNA. Finally, we show that although the individual domains bind DNA poorly, complex assembly requires oligomerization and cooperation between its OBD, helicase, and the linker domains.