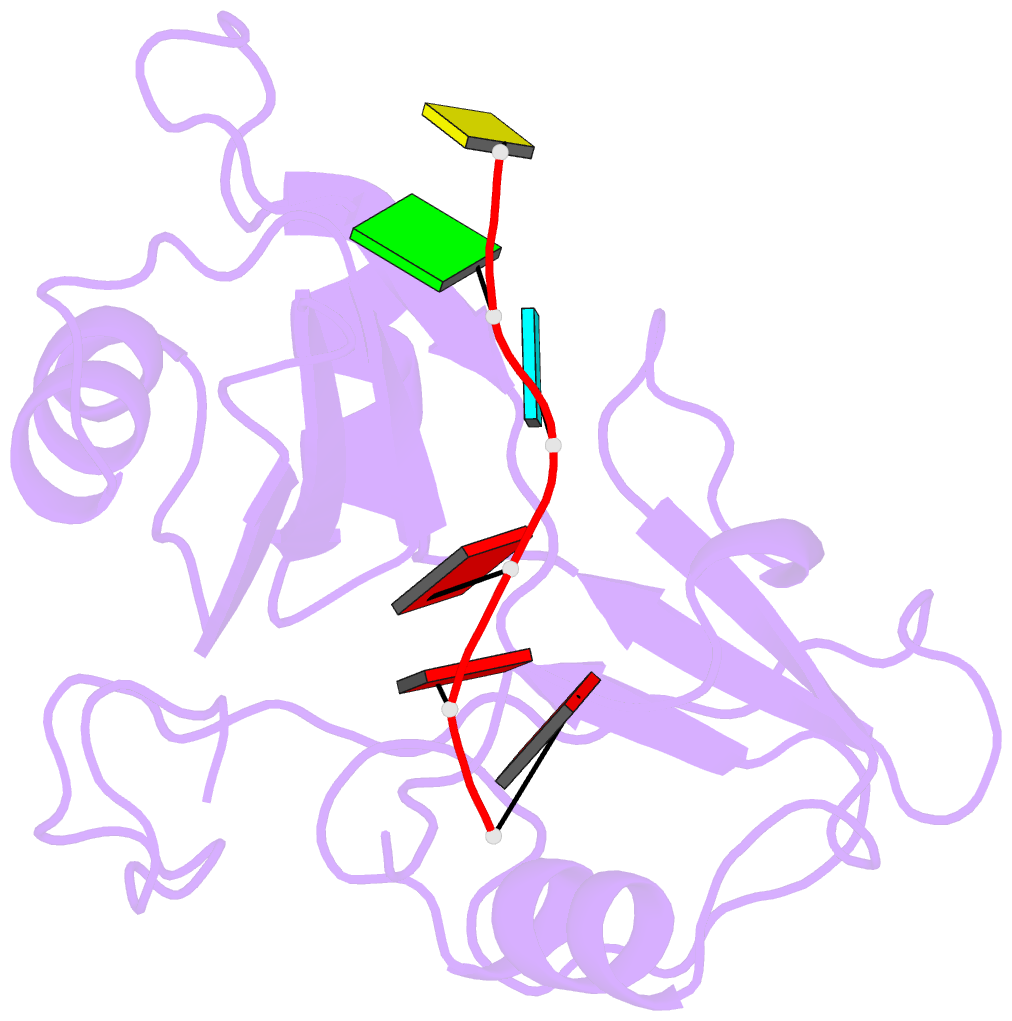
Summary information and primary citation
- PDB-id
- 5o1z; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transcription
- Method
- X-ray (3.4 Å)
- Summary
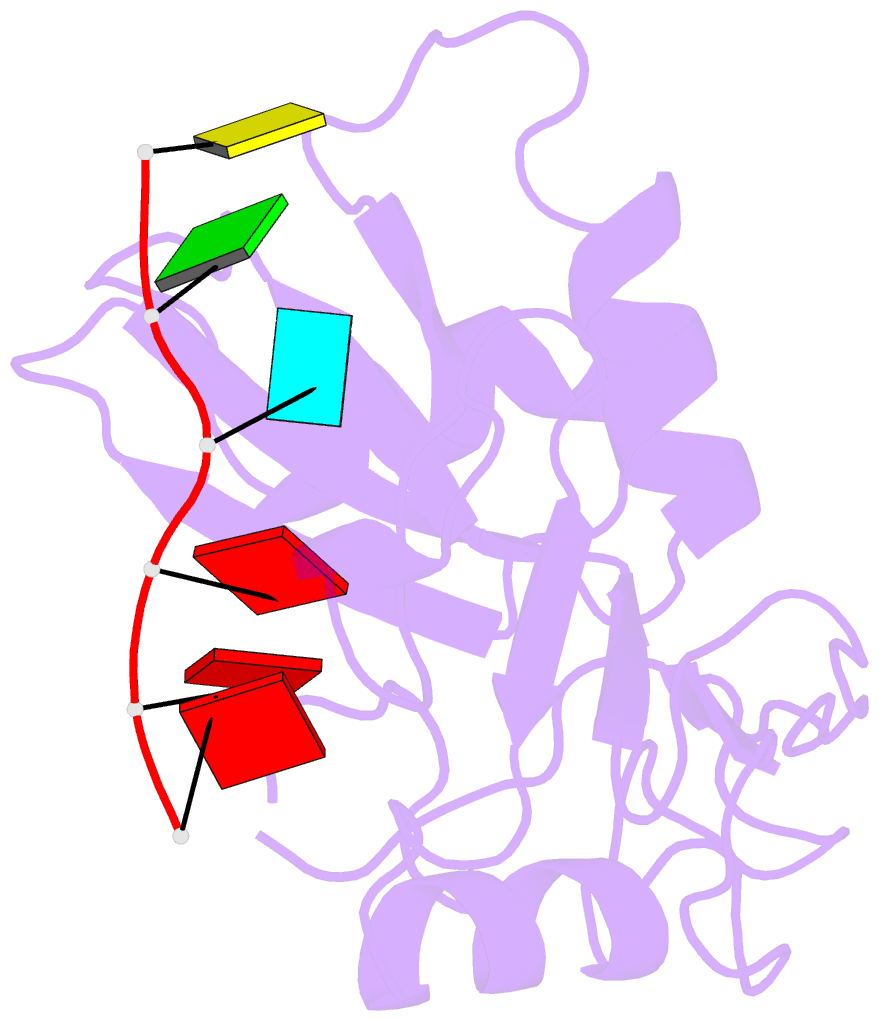
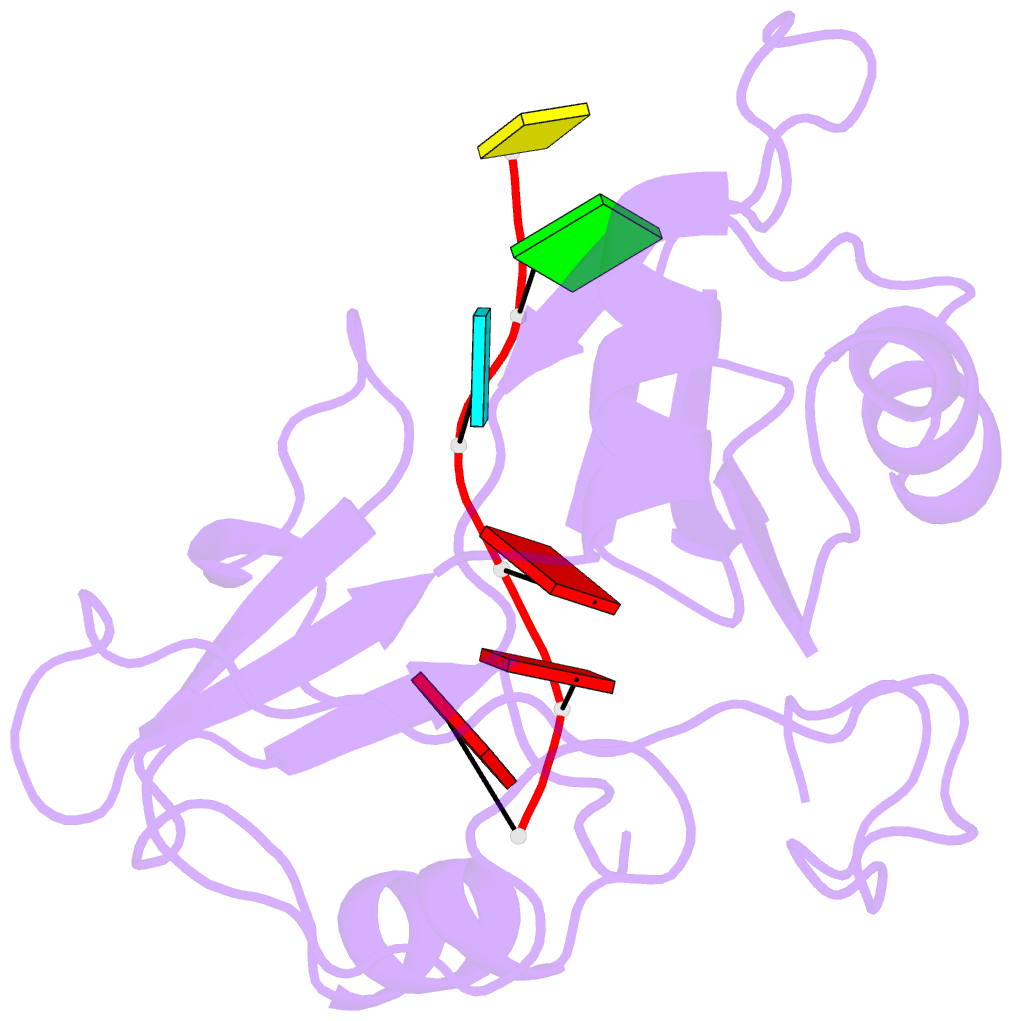
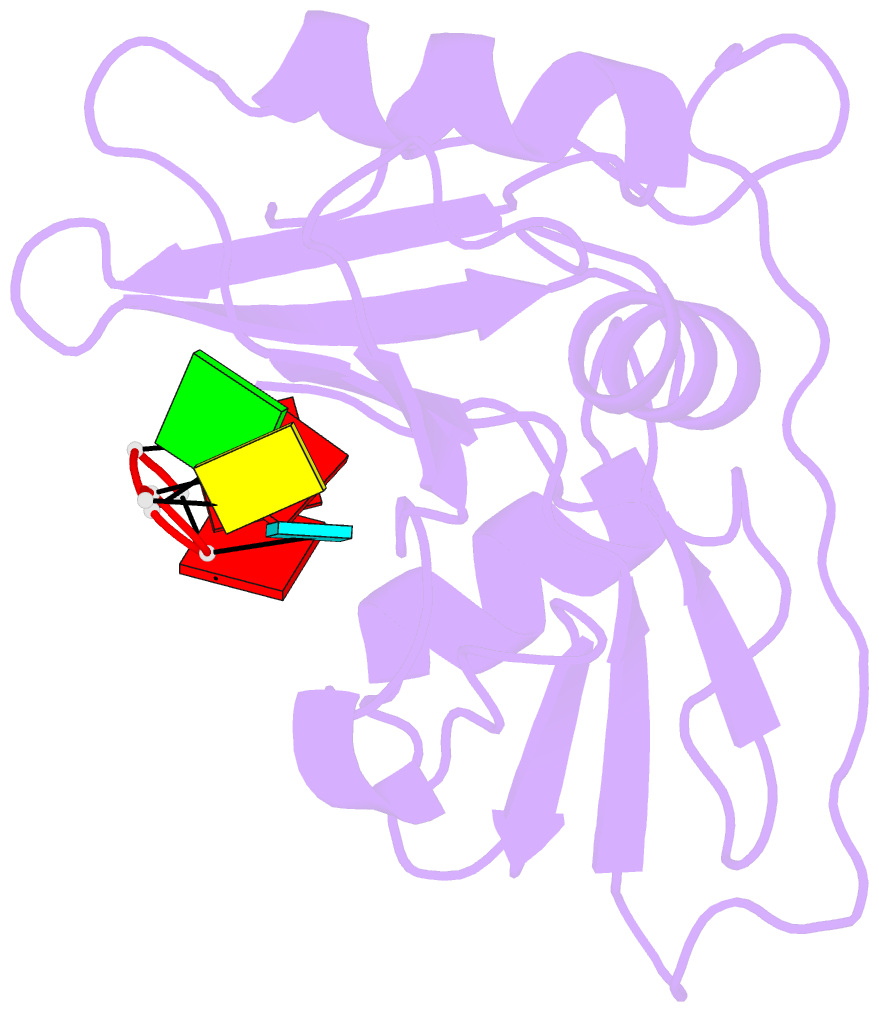
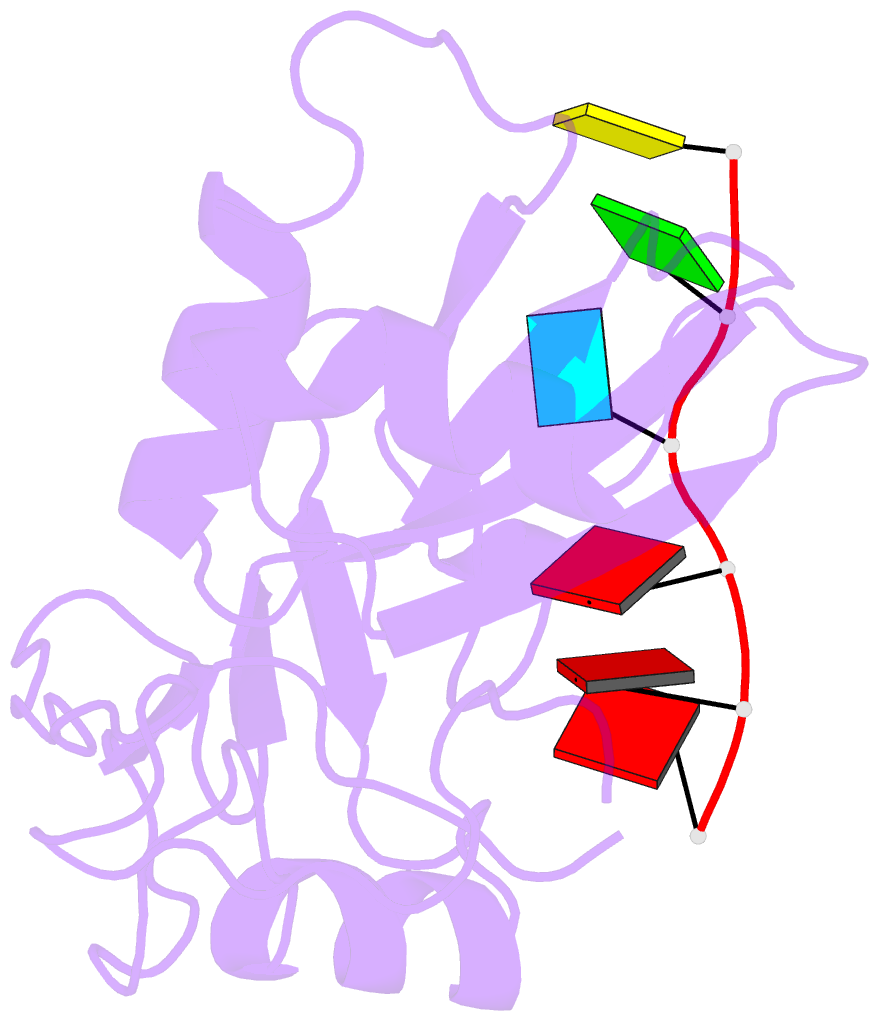
- Structure of nrd1 RNA binding domain in complex with RNA (cguaaa)
- Reference
- Franco-Echevarria E, Gonzalez-Polo N, Zorrilla S, Martinez-Lumbreras S, Santiveri CM, Campos-Olivas R, Sanchez M, Calvo O, Gonzalez B, Perez-Canadillas JM (2017): "The structure of transcription termination factor Nrd1 reveals an original mode for GUAA recognition." Nucleic Acids Res., 45, 10293-10305. doi: 10.1093/nar/gkx685.
- Abstract
- Transcription termination of non-coding RNAs is regulated in yeast by a complex of three RNA binding proteins: Nrd1, Nab3 and Sen1. Nrd1 is central in this process by interacting with Rbp1 of RNA polymerase II, Trf4 of TRAMP and GUAA/G terminator sequences. We lack structural data for the last of these binding events. We determined the structures of Nrd1 RNA binding domain and its complexes with three GUAA-containing RNAs, characterized RNA binding energetics and tested rationally designed mutants in vivo. The Nrd1 structure shows an RRM domain fused with a second α/β domain that we name split domain (SD), because it is formed by two non-consecutive segments at each side of the RRM. The GUAA interacts with both domains and with a pocket of water molecules, trapped between the two stacking adenines and the SD. Comprehensive binding studies demonstrate for the first time that Nrd1 has a slight preference for GUAA over GUAG and genetic and functional studies suggest that Nrd1 RNA binding domain might play further roles in non-coding RNAs transcription termination.