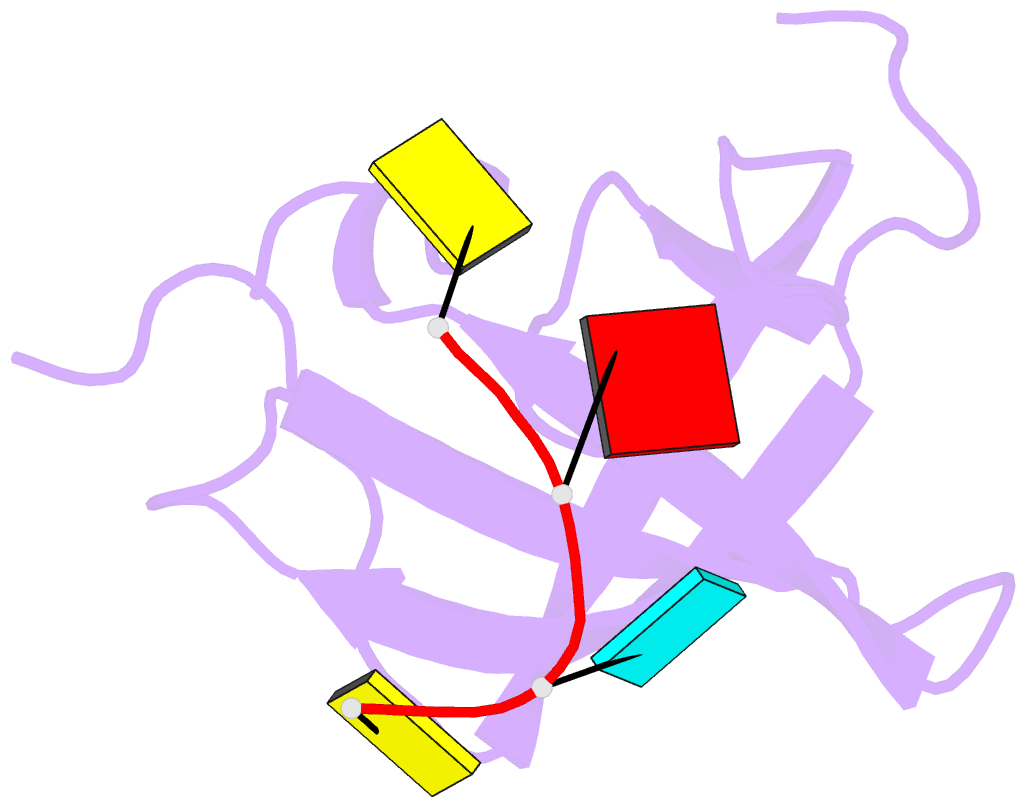
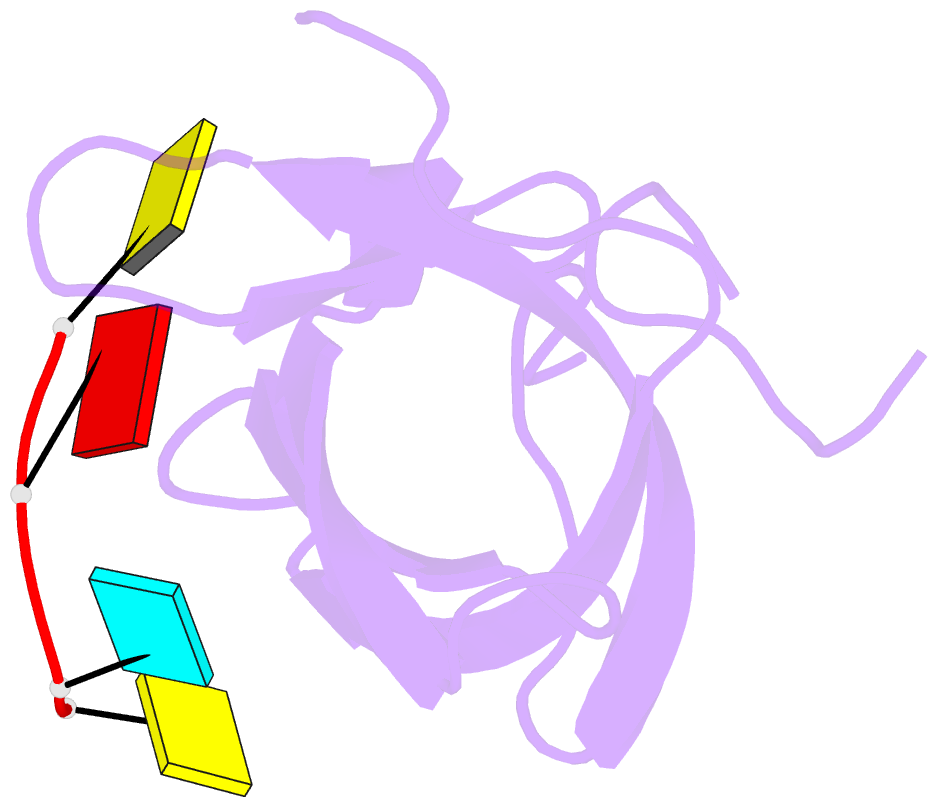
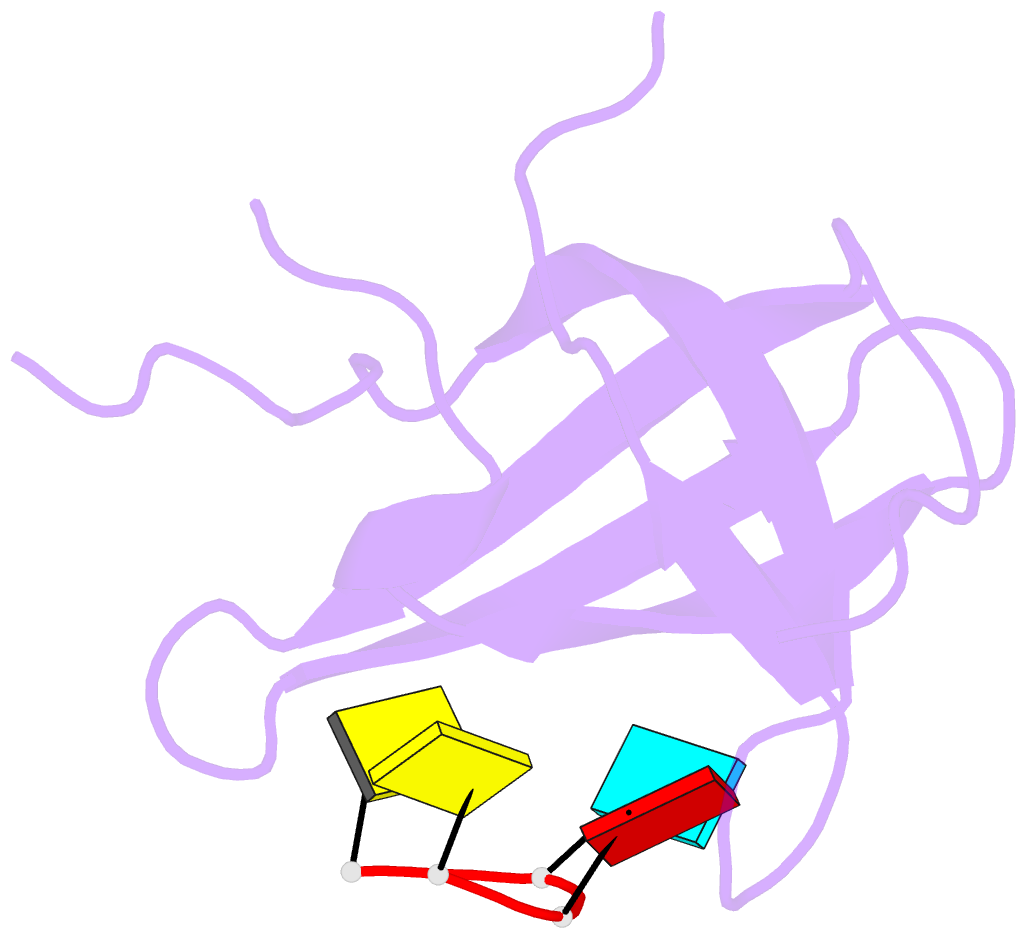
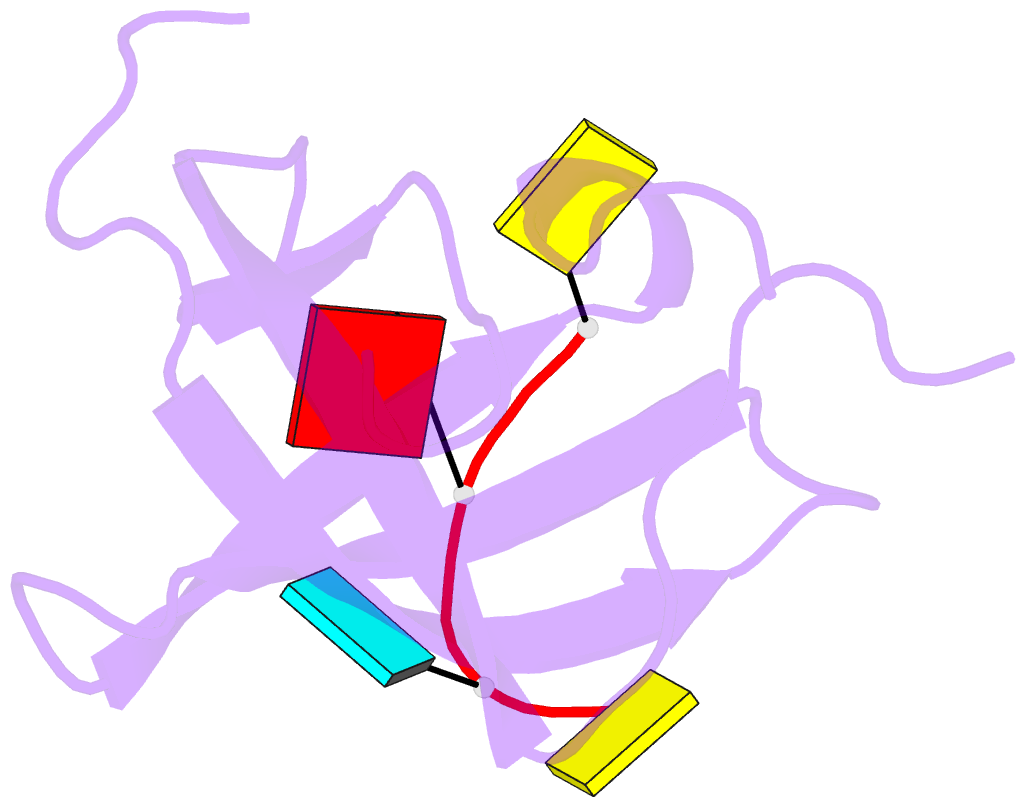
Summary information and primary citation
- PDB-id
- 5ytv; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- RNA binding protein-RNA
- Method
- X-ray (1.7 Å)
- Summary
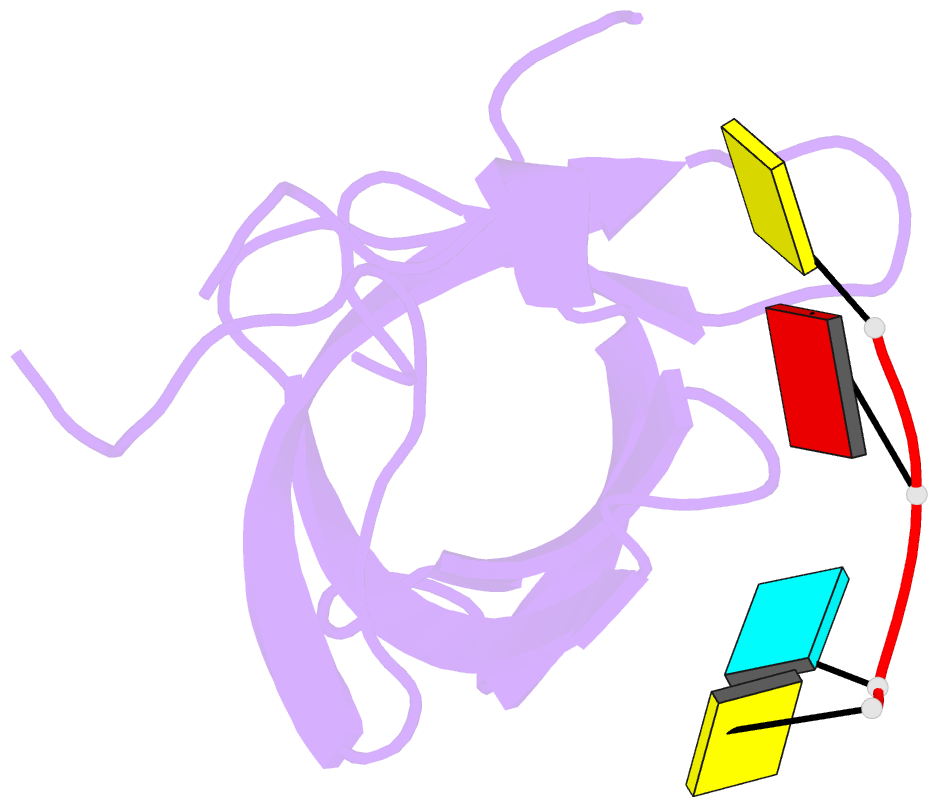
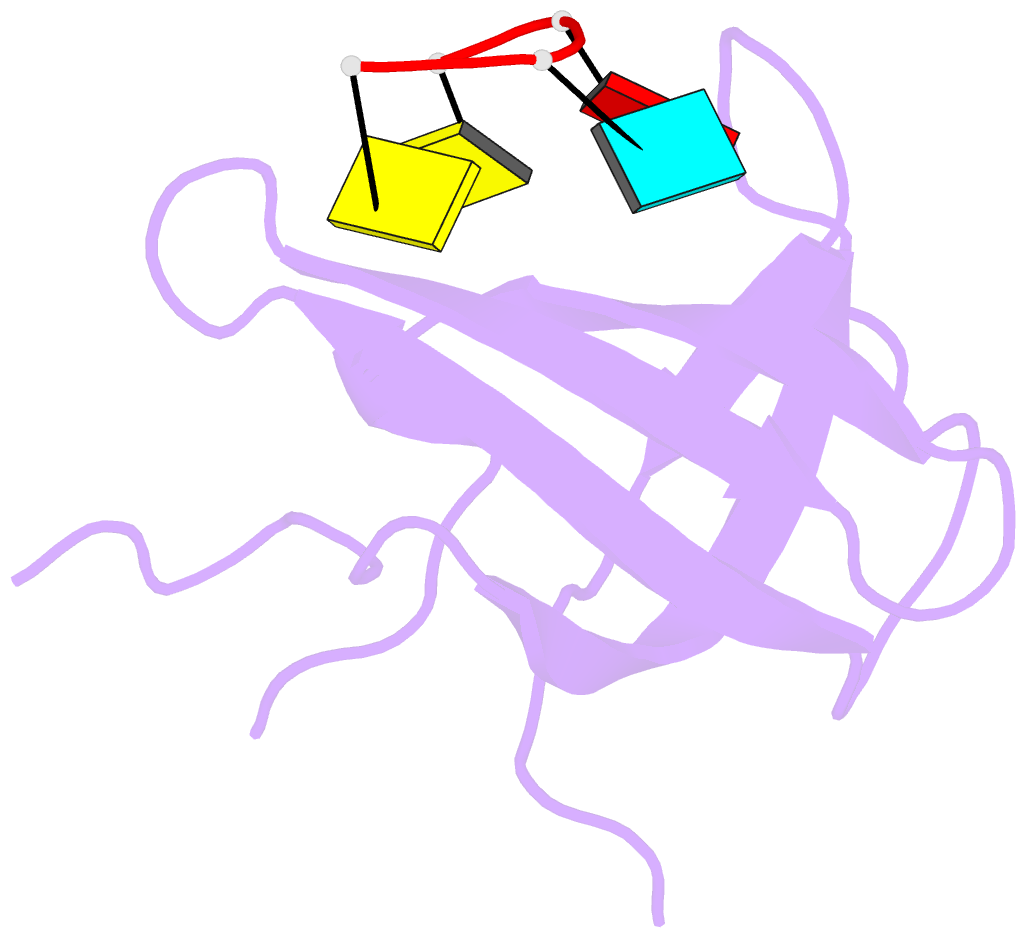
- Crystal structure of yb1 cold-shock domain in complex with ucaucu
- Reference
- Yang XJ, Zhu H, Mu SR, Wei WJ, Yuan X, Wang M, Liu Y, Hui J, Huang Y (2019): "Crystal structure of a Y-box binding protein 1 (YB-1)-RNA complex reveals key features and residues interacting with RNA." J.Biol.Chem., 294, 10998-11010. doi: 10.1074/jbc.RA119.007545.
- Abstract
- The Y-box binding protein 1 (YB-1) is a member of the cold shock domain (CSD) protein family and is recognized as an oncogenic factor in several solid tumors. By binding to RNA, YB-1 participates in several steps of posttranscriptional regulation of gene expression, including mRNA splicing, stability, and translation; microRNA processing; and stress granule assembly. However, the mechanisms in YB-1-mediated regulation of RNAs are unclear. Previously, we used both systematic evolution of ligands by exponential enrichment (SELEX) and individual-nucleotide resolution UV cross-linking and immunoprecipitation coupled RNA-Seq (iCLIP-Seq) analyses, which defined the RNA-binding consensus sequence of YB-1 as CA(U/C)C. We also reported that through binding to its core motif CAUC in primary transcripts, YB-1 regulates the alternative splicing of a CD44 variable exon and the biogenesis of miR-29b-2 during both Drosha and Dicer steps. To elucidate the molecular basis of the YB-1-RNA interactions, we report high-resolution crystal structures of the YB-1 CSD in complex with different RNA oligos at 1.7 Å resolution. The structure revealed that CSD interacts with RNA mainly through π-π stacking interactions assembled by four highly conserved aromatic residues. Interestingly, YB-1 CSD forms a homodimer in solution, and we observed that two residues, Tyr-99 and Asp-105, at the dimer interface are important for YB-1 CSD dimerization. Substituting these two residues with Ala reduced CSD's RNA-binding activity and abrogated the splicing activation of YB-1 targets. The YB-1 CSD-RNA structures presented here at atomic resolution provide mechanistic insights into gene expression regulated by CSD-containing proteins.