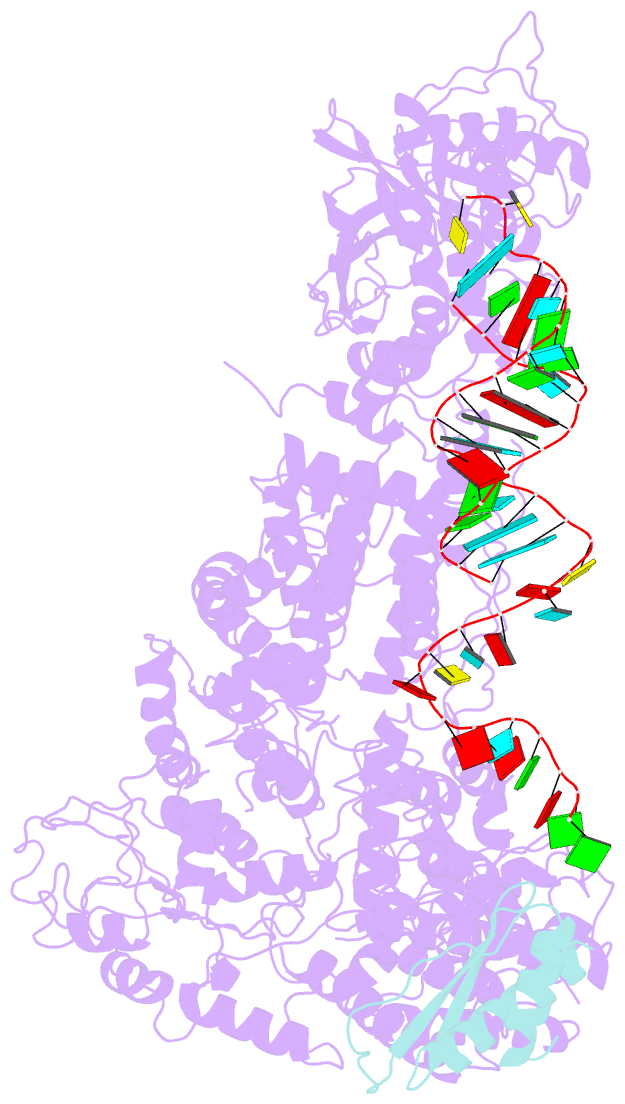
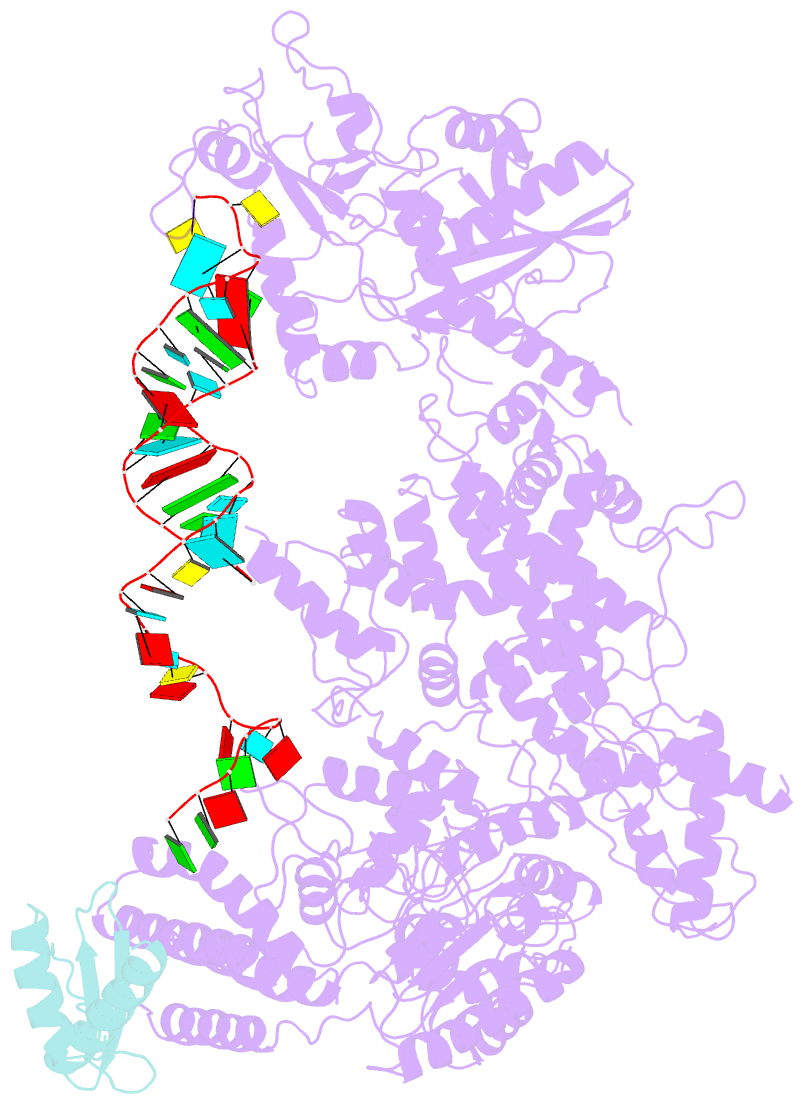
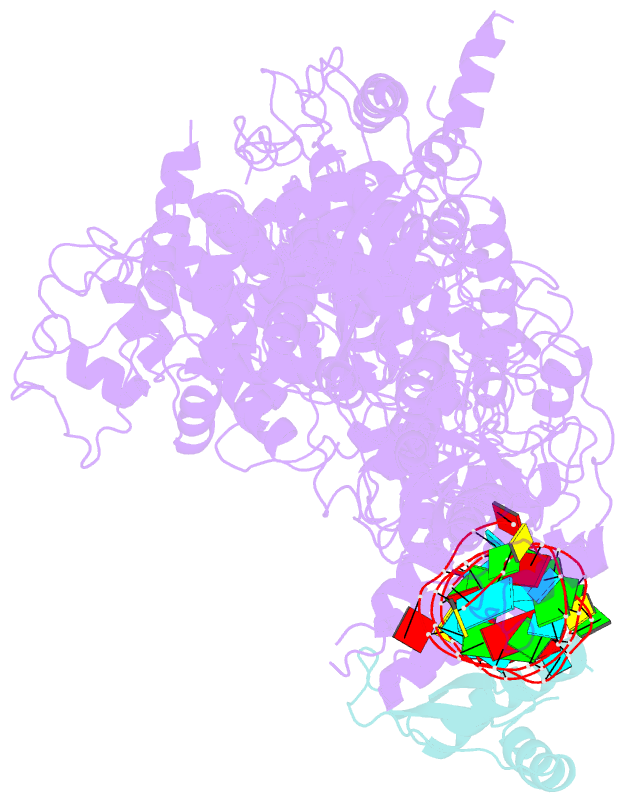
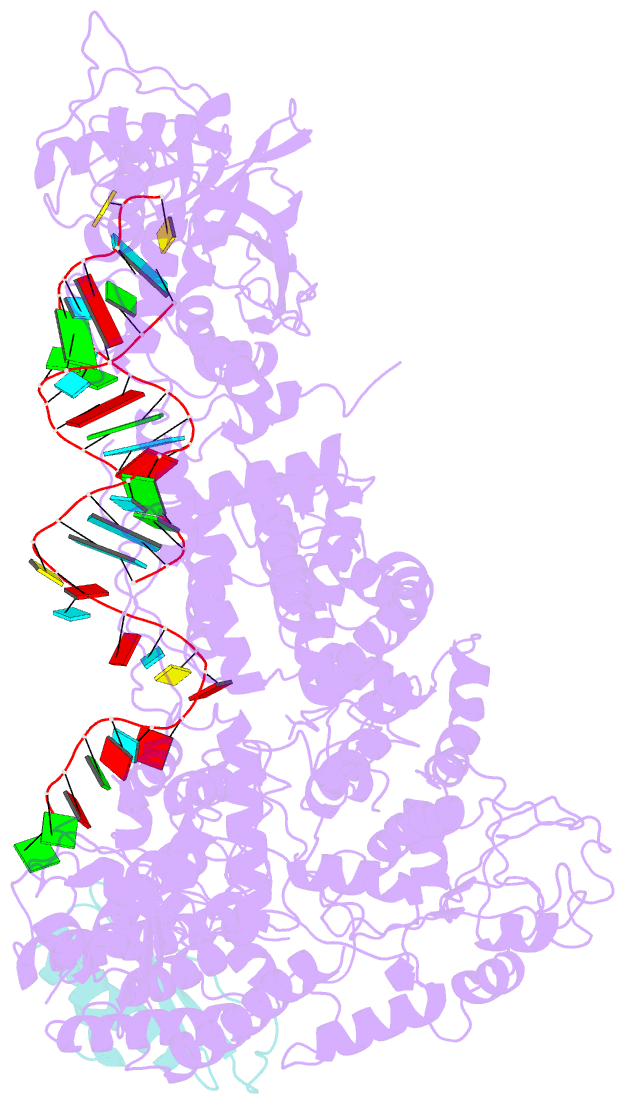
Summary information and primary citation
- PDB-id
-
5zam;
DSSR-derived features in text and
JSON formats; DNAproDB
- Class
- hydrolase-protein binding-RNA
- Method
- cryo-EM (5.7 Å)
- Summary
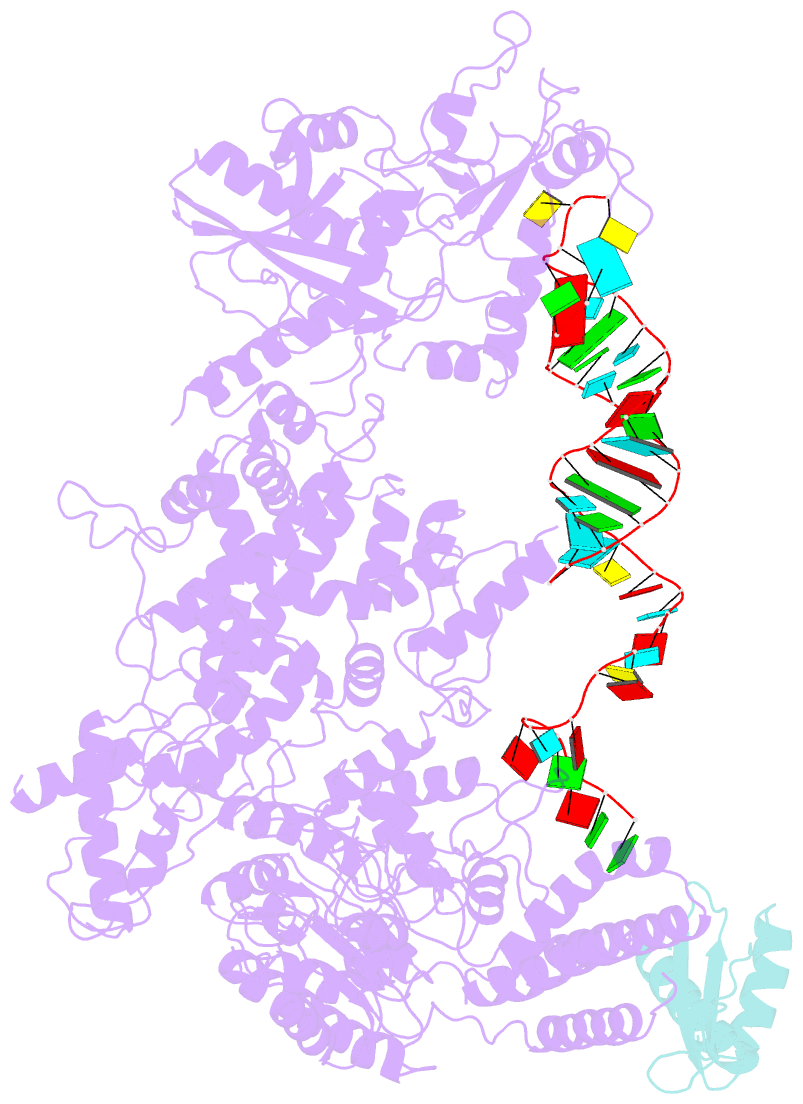
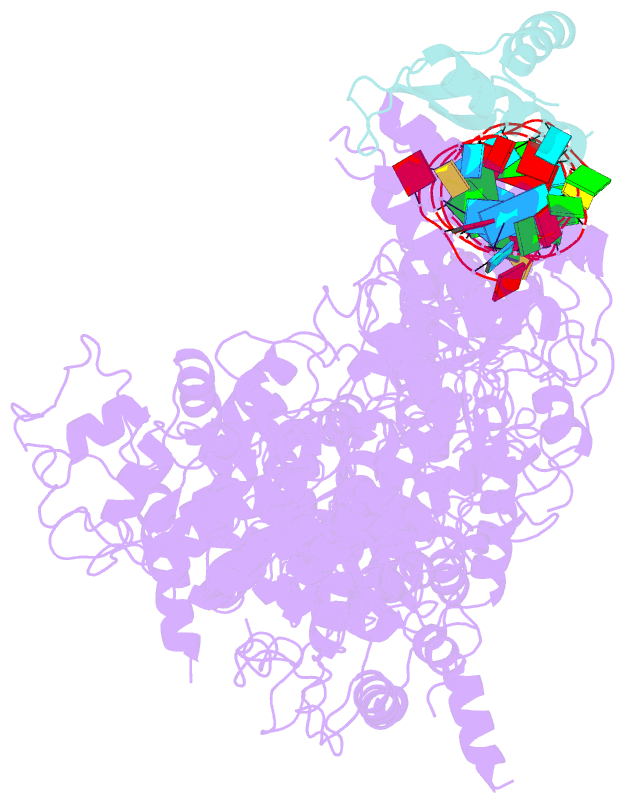
- cryo-EM structure of human dicer and its complexes with
a pre-mirna substrate
- Reference
-
Liu Z, Wang J, Cheng H, Ke X, Sun L, Zhang QC, Wang HW
(2018): "Cryo-EM
Structure of Human Dicer and Its Complexes with a
Pre-miRNA Substrate." Cell,
173, 1191-1203.e12. doi: 10.1016/j.cell.2018.03.080.
- Abstract
- Human Dicer (hDicer) is a multi-domain protein
belonging to the RNase III family. It plays pivotal
roles in small RNA biogenesis during the RNA
interference (RNAi) pathway by processing a diverse range
of double-stranded RNA (dsRNA) precursors to generate ∼22
nt microRNA (miRNA) or small interfering RNA (siRNA)
products for sequence-directed gene silencing. In this
work, we solved the cryoelectron microscopy (cryo-EM)
structure of hDicer in complex with its cofactor protein
TRBP and revealed the precise spatial arrangement of
hDicer's multiple domains. We further solved
structures of the hDicer-TRBP complex bound with pre-let-7
RNA in two distinct conformations. In combination with
biochemical analysis, these structures reveal a property of
the hDicer-TRBP complex to promote the stability of
pre-miRNA's stem duplex in a pre-dicing state. These
results provide insights into the mechanism of RNA
processing by hDicer and illustrate the regulatory role of
hDicer's N-terminal helicase domain.