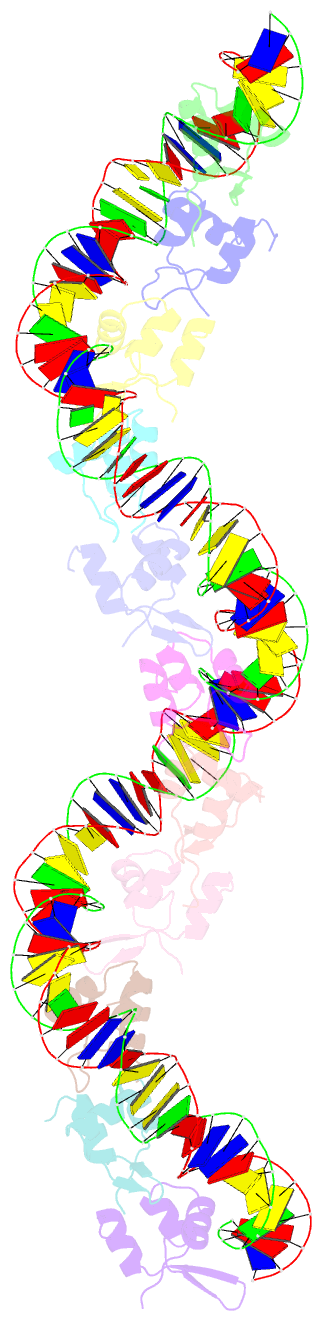
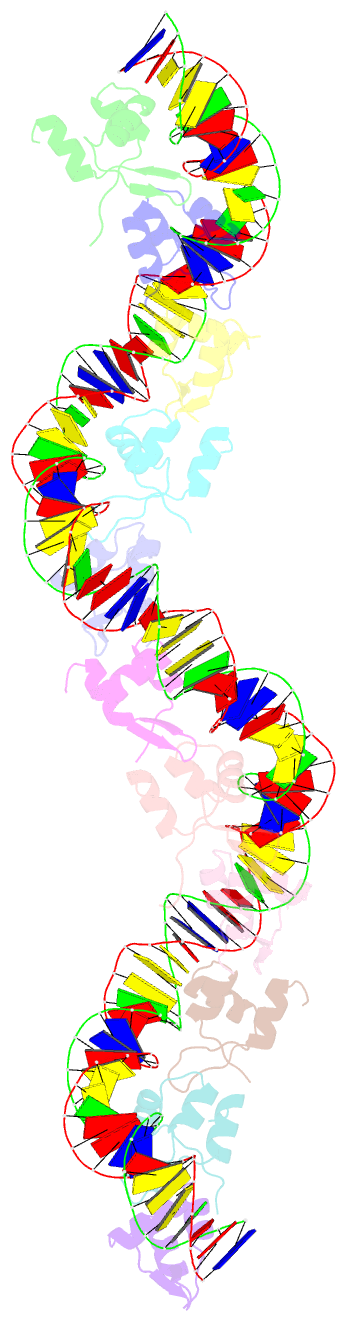
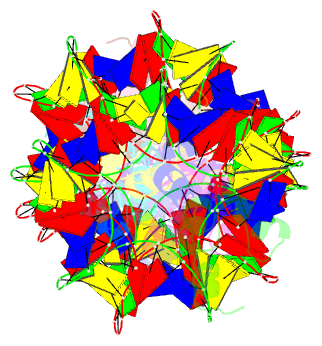
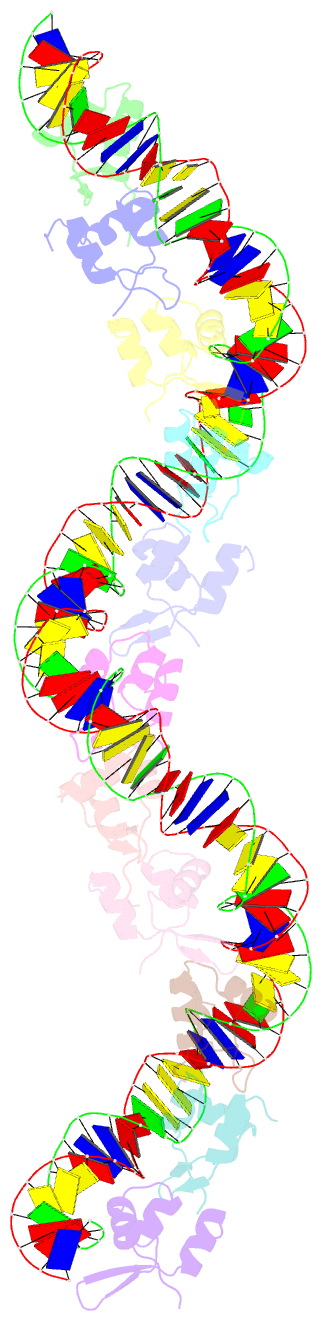
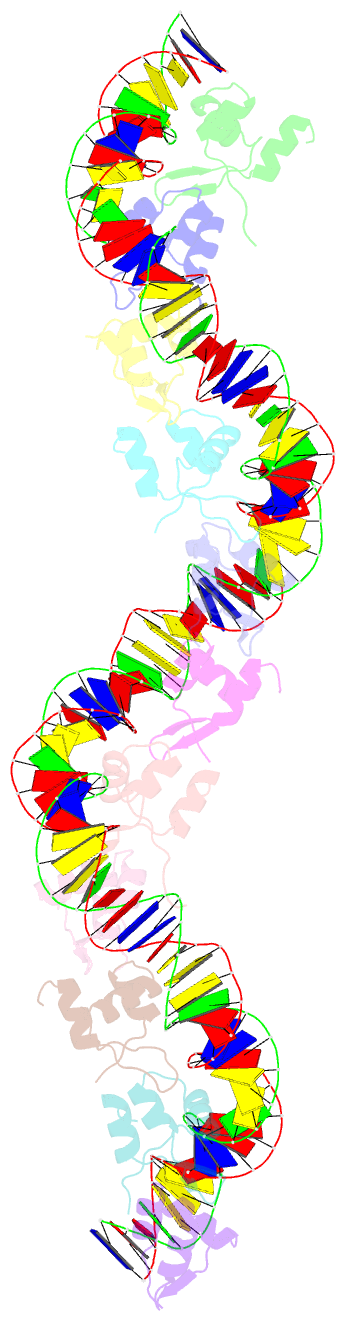
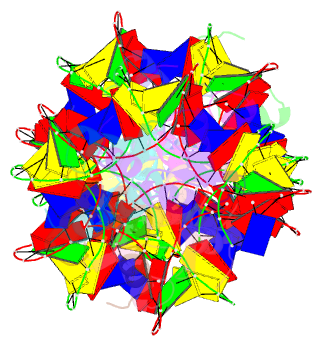
Summary information and primary citation
- PDB-id
- 6ama; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- DNA binding protein-DNA
- Method
- X-ray (3.09 Å)
- Summary
- Structure of s. coelicolor-s. venezuelae bldc-smea-ssfa complex to 3.09 angstrom
- Reference
- Schumacher MA, den Hengst CD, Bush MJ, Le TBK, Tran NT, Chandra G, Zeng W, Travis B, Brennan RG, Buttner MJ (2018): "The MerR-like protein BldC binds DNA direct repeats as cooperative multimers to regulate Streptomyces development." Nat Commun, 9, 1139. doi: 10.1038/s41467-018-03576-3.
- Abstract
- Streptomycetes are notable for their complex life cycle and production of most clinically important antibiotics. A key factor that controls entry into development and the onset of antibiotic production is the 68-residue protein, BldC. BldC is a putative DNA-binding protein related to MerR regulators, but lacks coiled-coil dimerization and effector-binding domains characteristic of classical MerR proteins. Hence, the molecular function of the protein has been unclear. Here we show that BldC is indeed a DNA-binding protein and controls a regulon that includes other key developmental regulators. Intriguingly, BldC DNA-binding sites vary significantly in length. Our BldC-DNA structures explain this DNA-binding capability by revealing that BldC utilizes a DNA-binding mode distinct from MerR and other known regulators, involving asymmetric head-to-tail oligomerization on DNA direct repeats that results in dramatic DNA distortion. Notably, BldC-like proteins radiate throughout eubacteria, establishing BldC as the founding member of a new structural family of regulators.