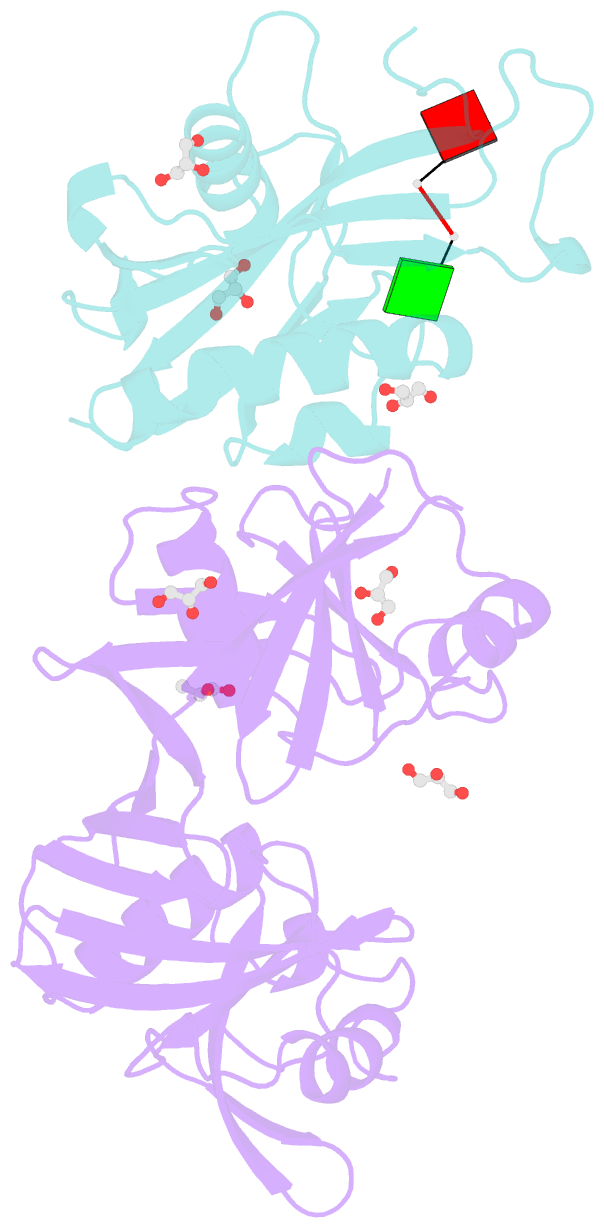
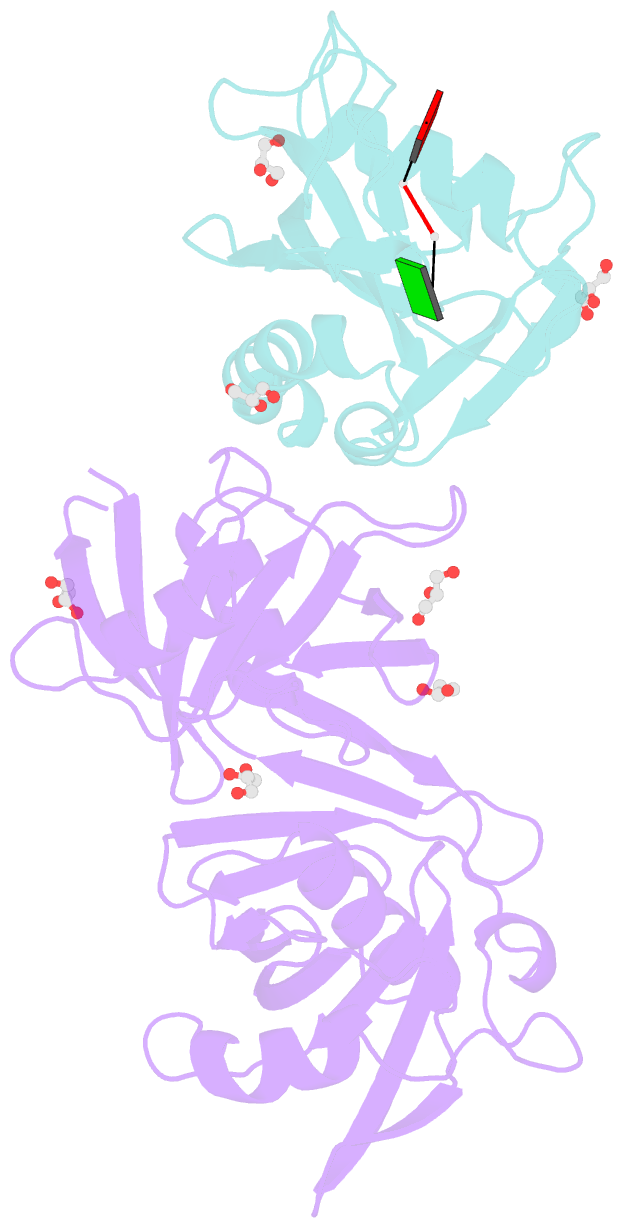
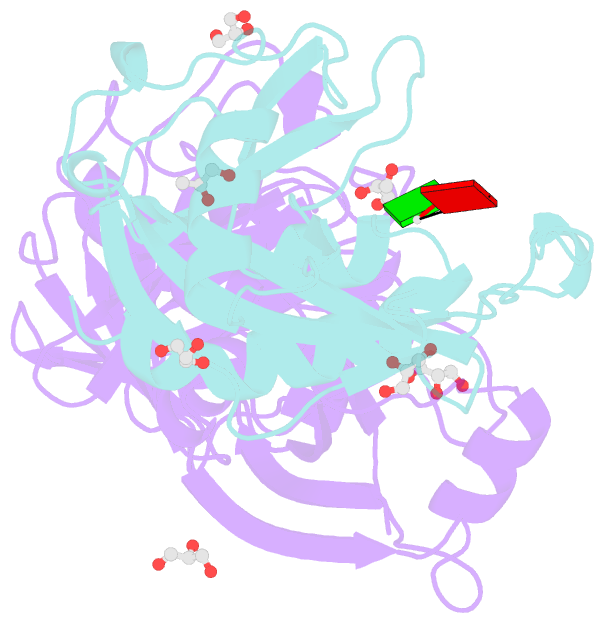
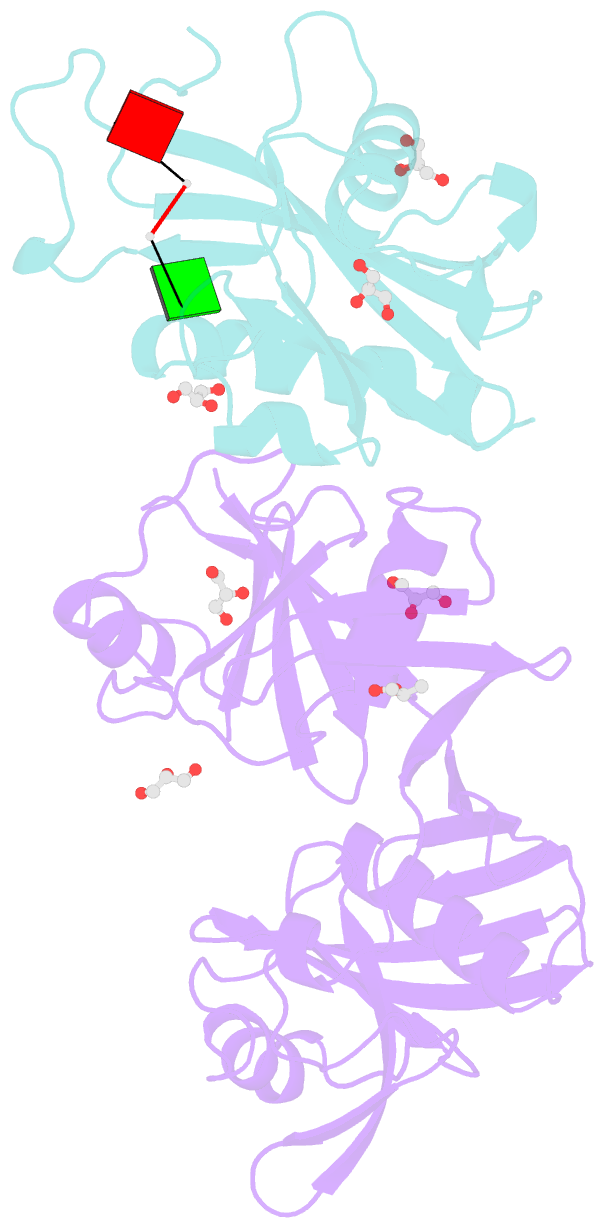
Summary information and primary citation
- PDB-id
- 6d1v; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- isomerase-hydrolase-RNA
- Method
- X-ray (1.81 Å)
- Summary
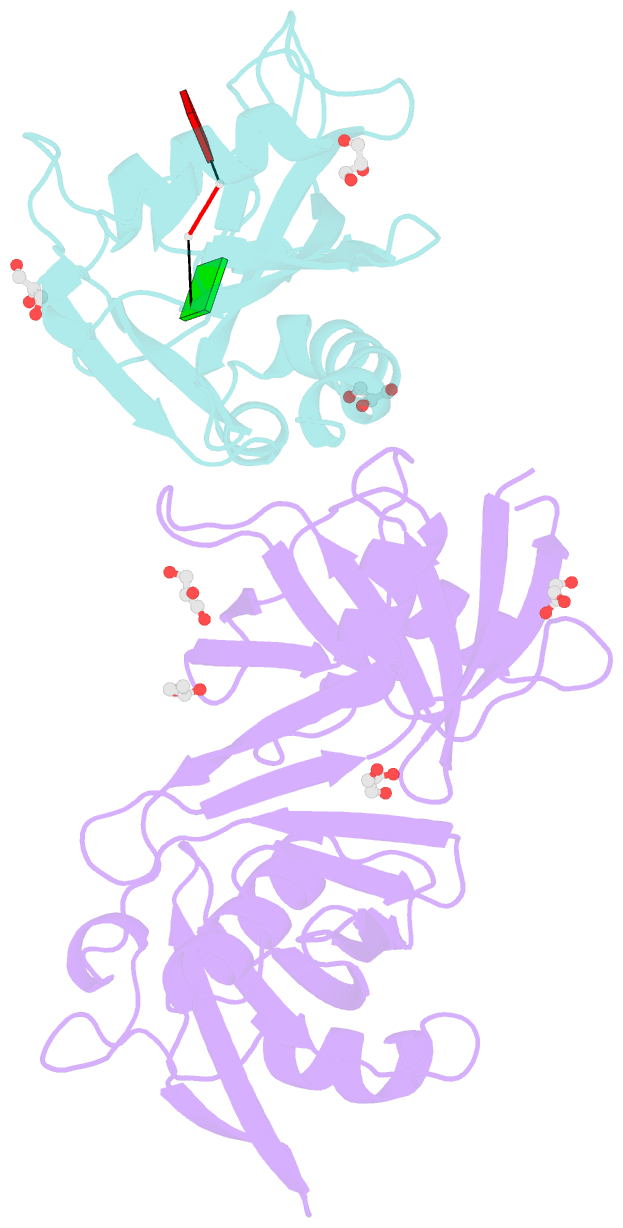
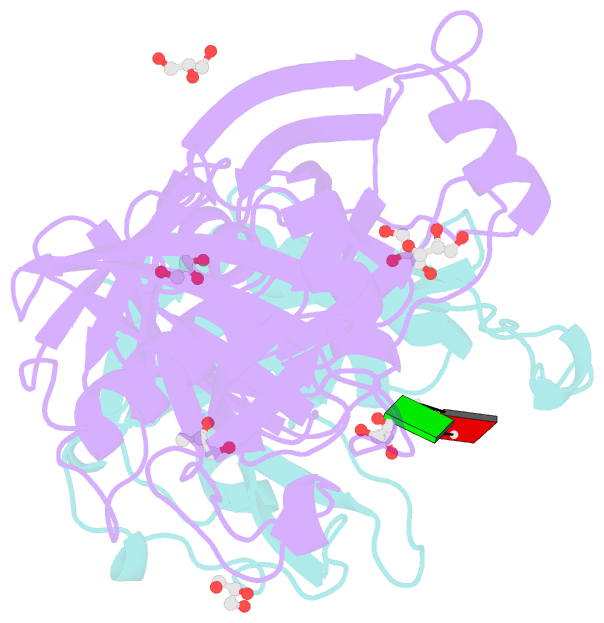
- Crystal structure of e. coli rpph-dapf complex, monomer bound to RNA
- Reference
- Gao A, Vasilyev N, Luciano DJ, Levenson-Palmer R, Richards J, Marsiglia WM, Traaseth NJ, Belasco JG, Serganov A (2018): "Structural and kinetic insights into stimulation of RppH-dependent RNA degradation by the metabolic enzyme DapF." Nucleic Acids Res., 46, 6841-6856. doi: 10.1093/nar/gky327.
- Abstract
- Vitally important for controlling gene expression in eukaryotes and prokaryotes, the deprotection of mRNA 5' termini is governed by enzymes whose activity is modulated by interactions with ancillary factors. In Escherichia coli, 5'-end-dependent mRNA degradation begins with the generation of monophosphorylated 5' termini by the RNA pyrophosphohydrolase RppH, which can be stimulated by DapF, a diaminopimelate epimerase involved in amino acid and cell wall biosynthesis. We have determined crystal structures of RppH-DapF complexes and measured rates of RNA deprotection. These studies show that DapF potentiates RppH activity in two ways, depending on the nature of the substrate. Its stimulatory effect on the reactivity of diphosphorylated RNAs, the predominant natural substrates of RppH, requires a substrate long enough to reach DapF in the complex, while the enhanced reactivity of triphosphorylated RNAs appears to involve DapF-induced changes in RppH itself and likewise increases with substrate length. This study provides a basis for understanding the intricate relationship between cellular metabolism and mRNA decay and reveals striking parallels with the stimulation of decapping activity in eukaryotes.