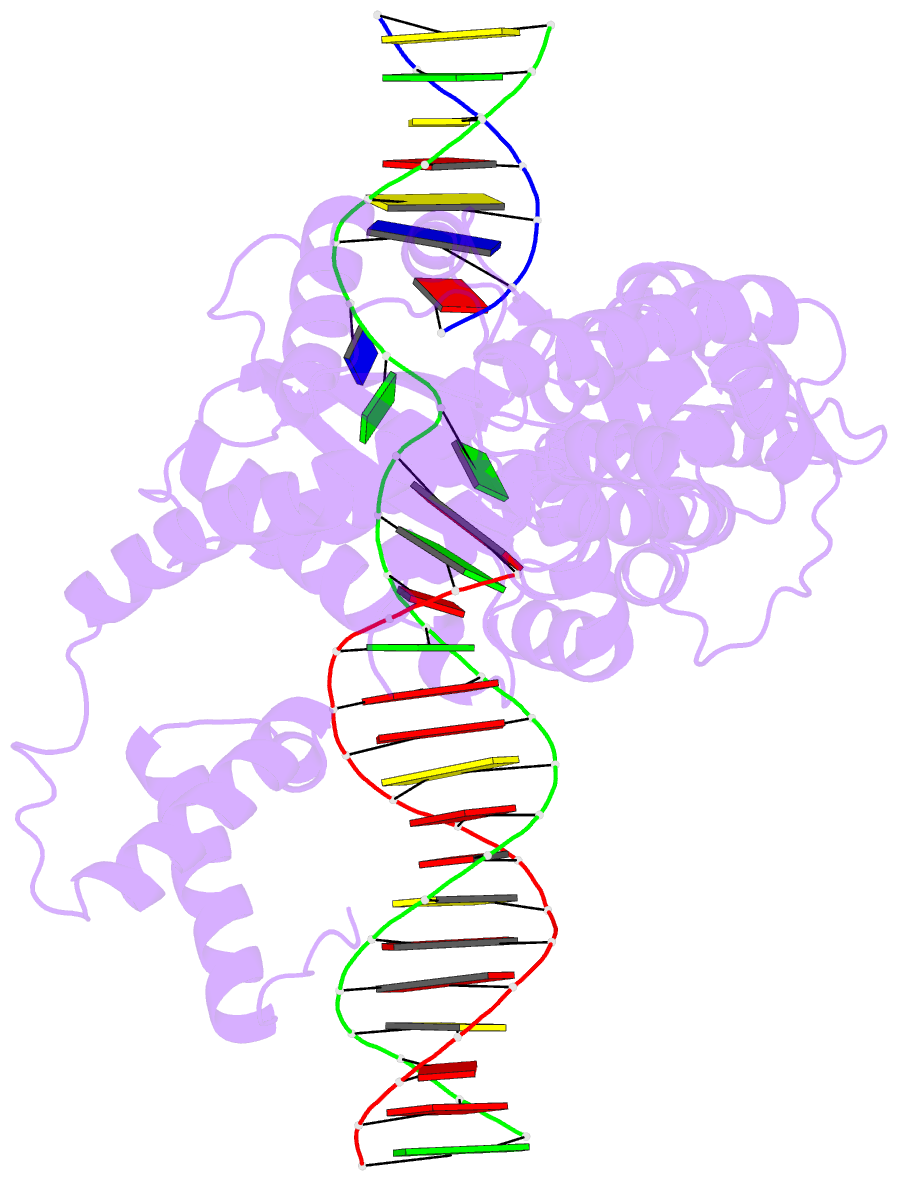
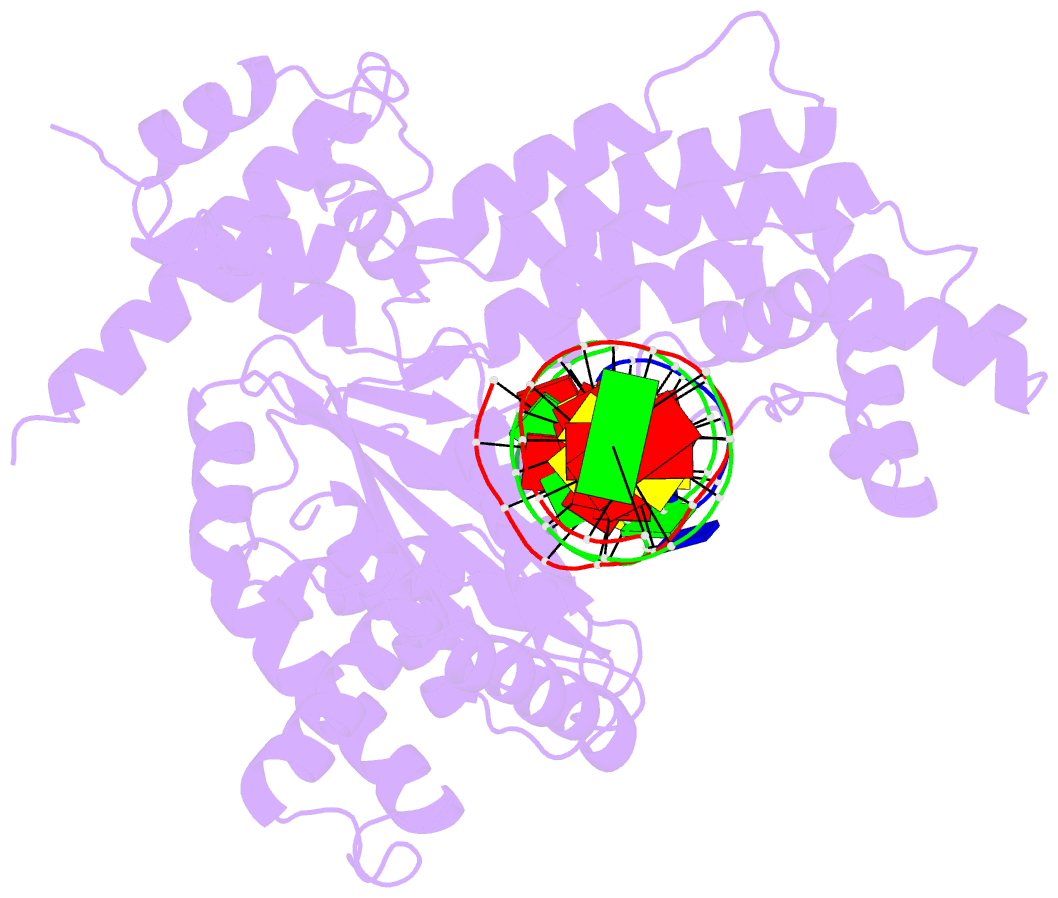
Summary information and primary citation
- PDB-id
- 6dww; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- DNA binding protein-DNA
- Method
- X-ray (2.851 Å)
- Summary
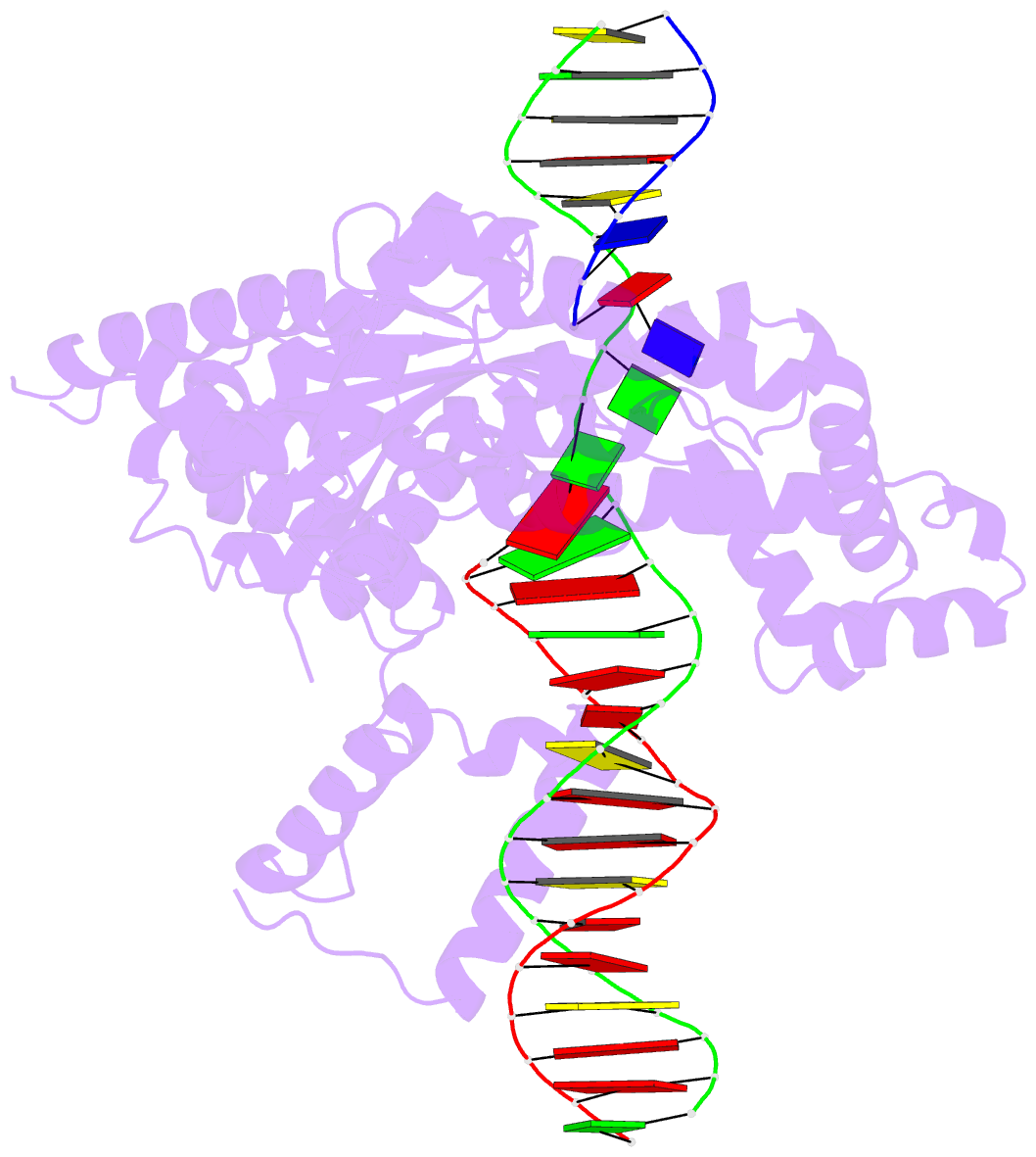
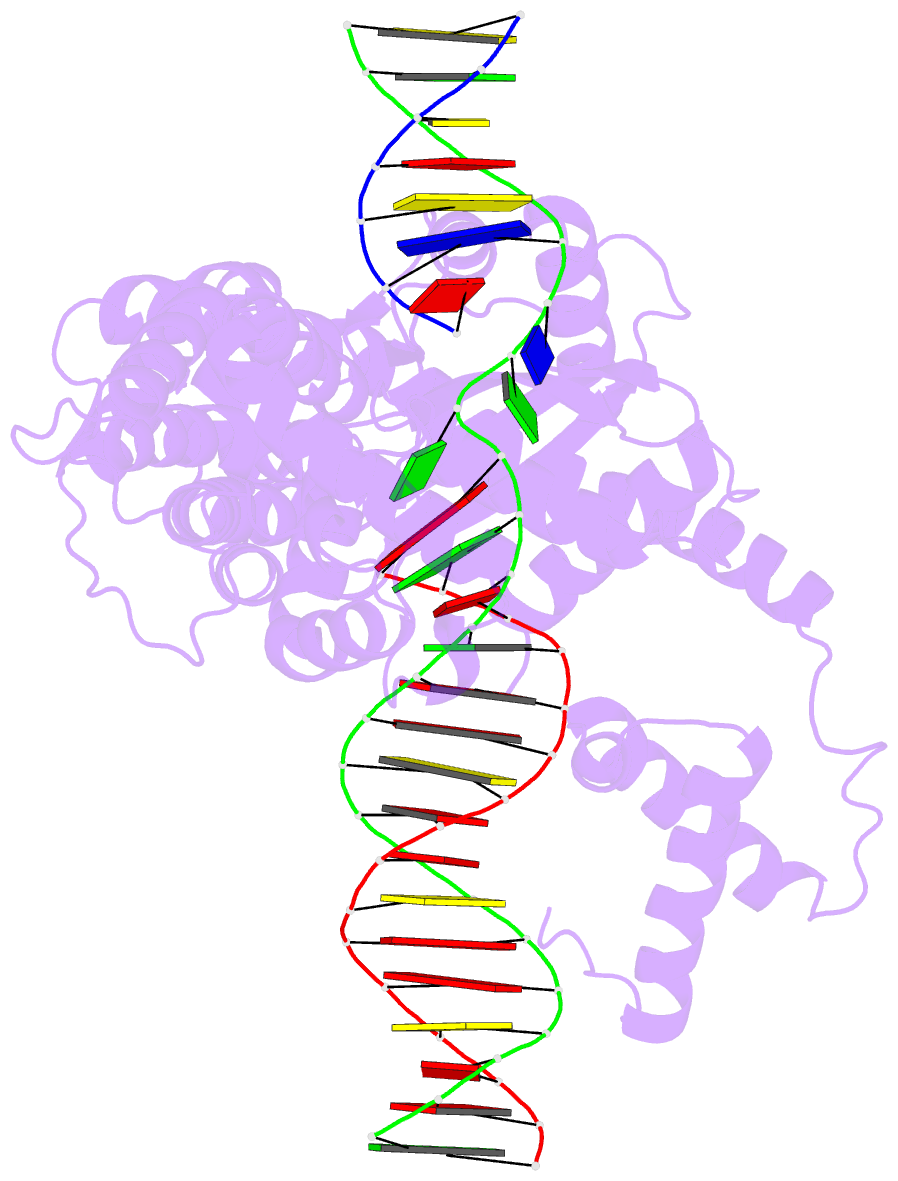
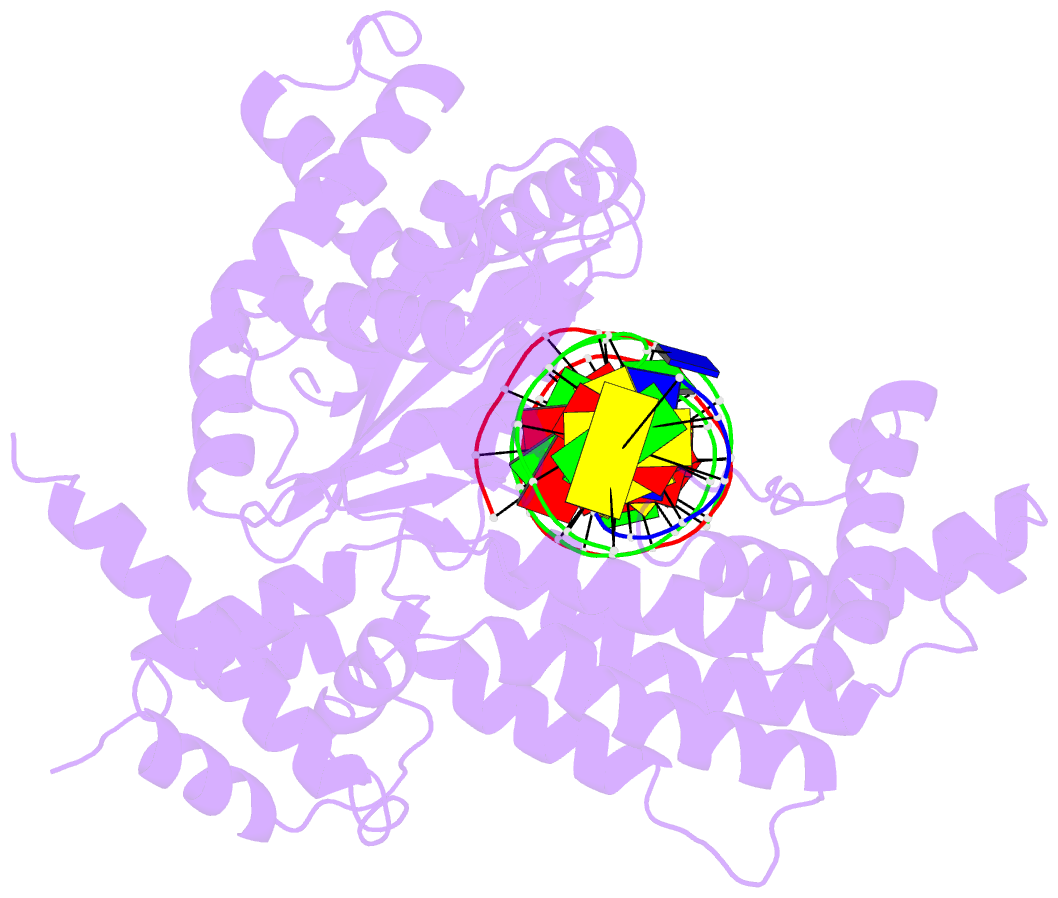
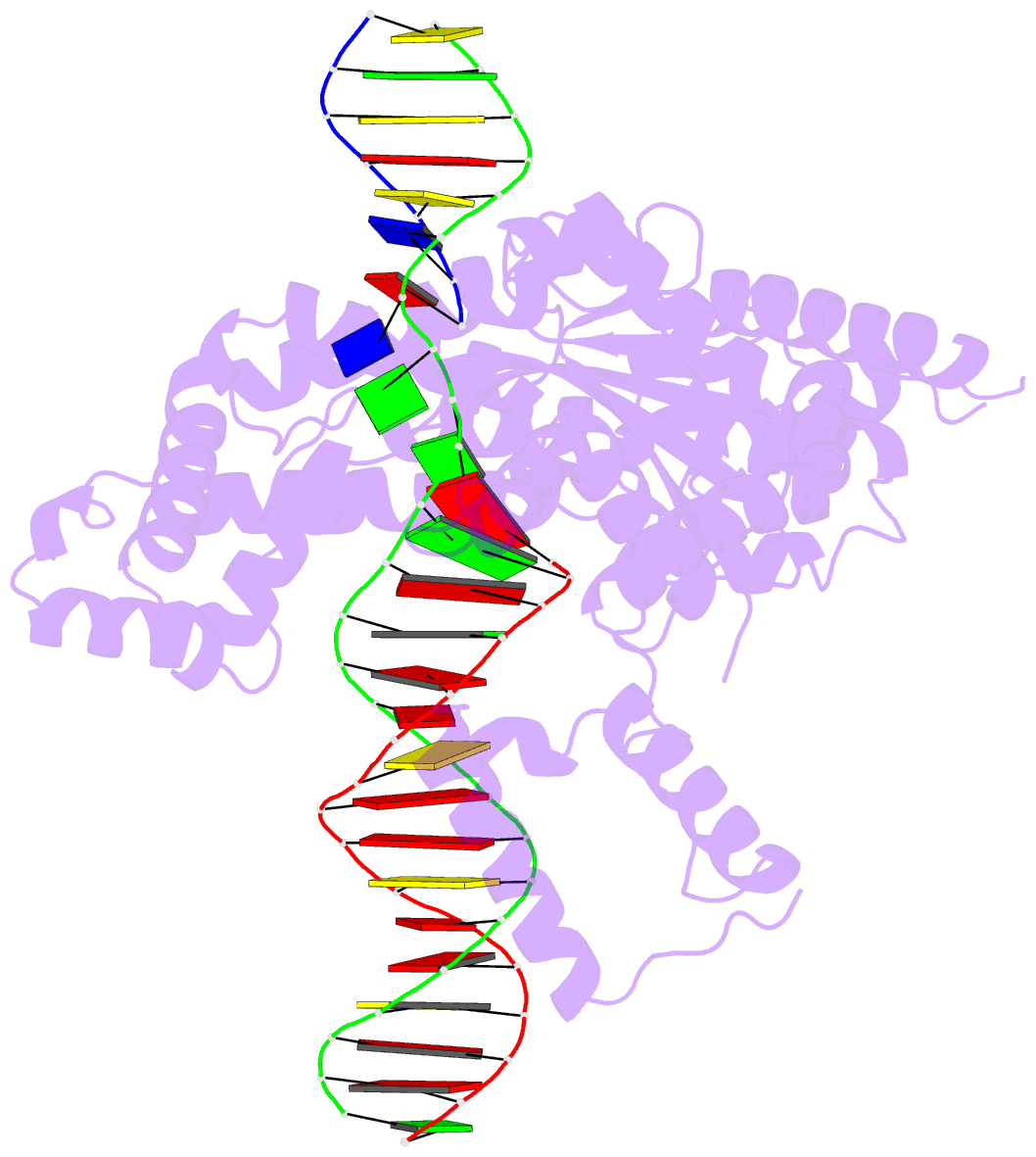
- Hermes transposase deletion dimer complex with (a-t) DNA and mn2+
- Reference
- Hickman AB, Voth AR, Ewis H, Li X, Craig NL, Dyda F (2018): "Structural insights into the mechanism of double strand break formation by Hermes, a hAT family eukaryotic DNA transposase." Nucleic Acids Res., 46, 10286-10301. doi: 10.1093/nar/gky838.
- Abstract
- Some DNA transposons relocate from one genomic location to another using a mechanism that involves generating double-strand breaks at their transposon ends by forming hairpins on flanking DNA. The same double-strand break mode is employed by the V(D)J recombinase at signal-end/coding-end junctions during the generation of antibody diversity. How flanking hairpins are formed during DNA transposition has remained elusive. Here, we describe several co-crystal structures of the Hermes transposase bound to DNA that mimics the reaction step immediately prior to hairpin formation. Our results reveal a large DNA conformational change between the initial cleavage step and subsequent hairpin formation that changes which strand is acted upon by a single active site. We observed that two factors affect the conformational change: the complement of divalent metal ions bound by the catalytically essential DDE residues, and the identity of the -2 flanking base pair. Our data also provides a mechanistic link between the efficiency of hairpin formation (an A:T basepair is favored at the -2 position) and Hermes' strong target site preference. Furthermore, we have established that the histidine residue within a conserved C/DxxH motif present in many transposase families interacts directly with the scissile phosphate, suggesting a crucial role in catalysis.