Summary information and primary citation
- PDB-id
- 6ir8; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transcription-DNA
- Method
- X-ray (2.3 Å)
- Summary
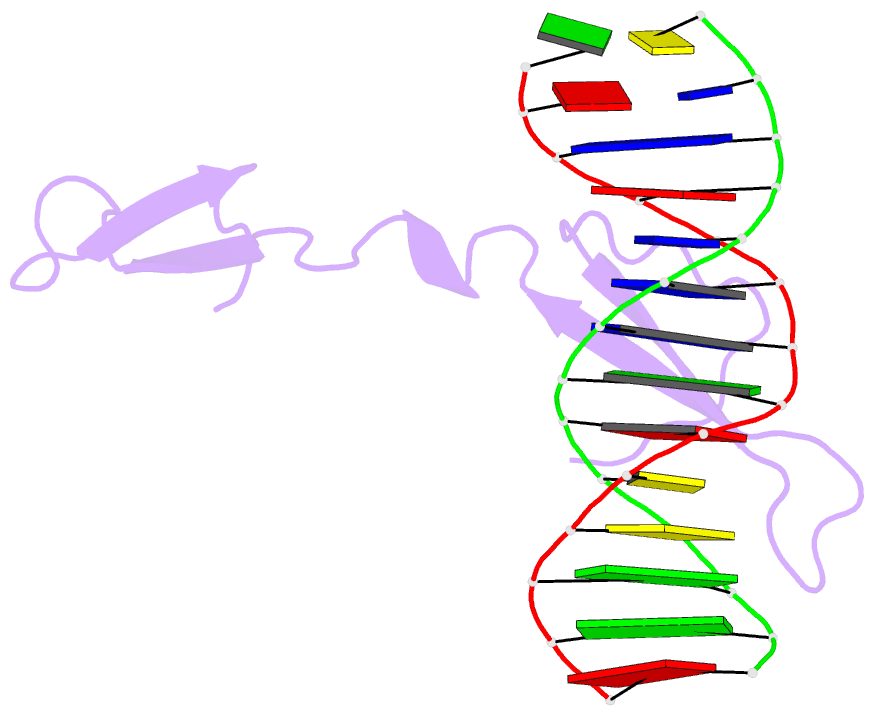
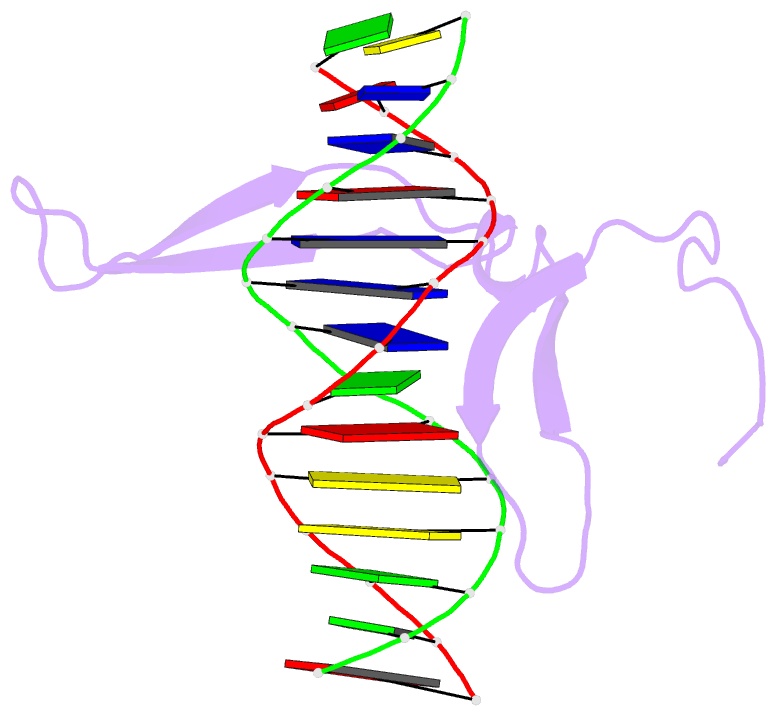
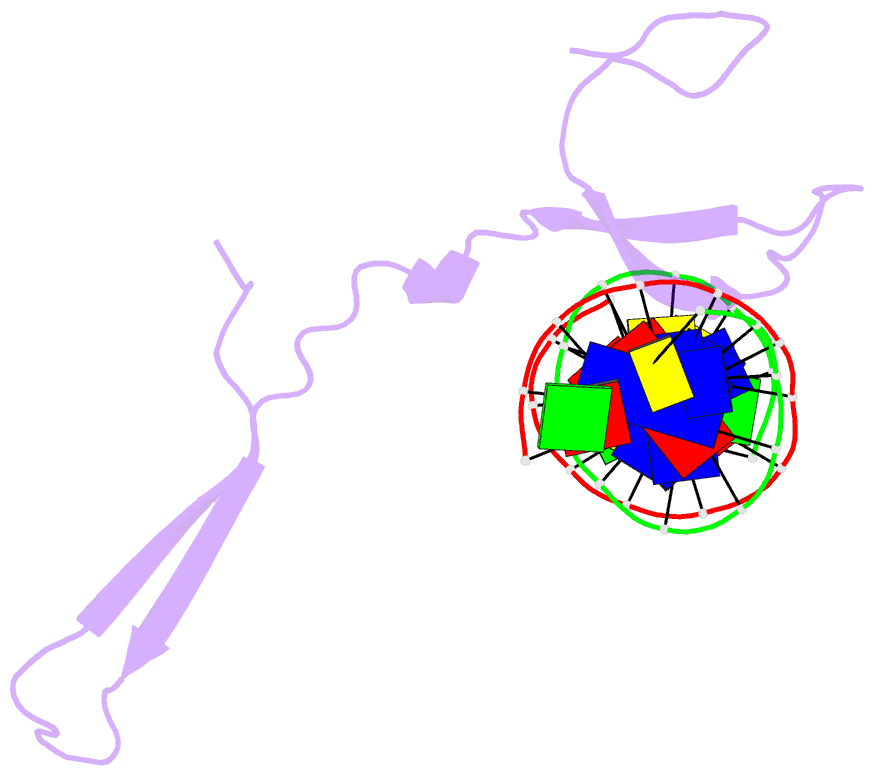
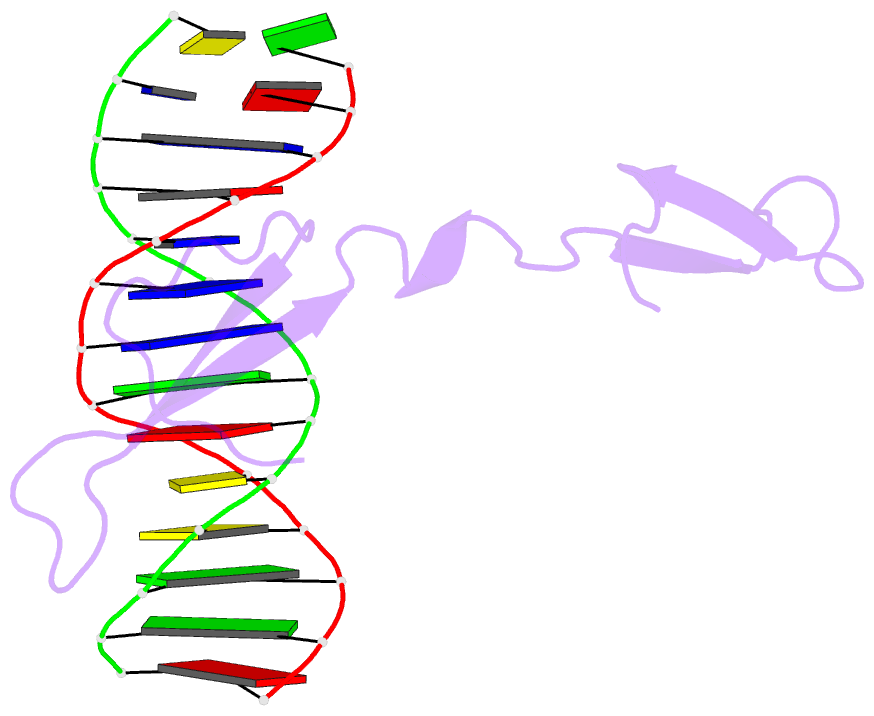
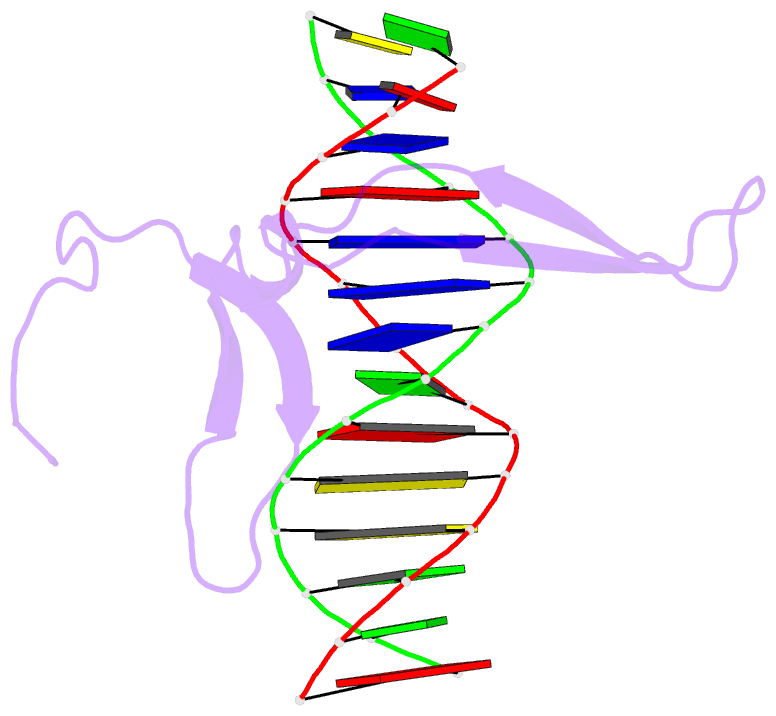
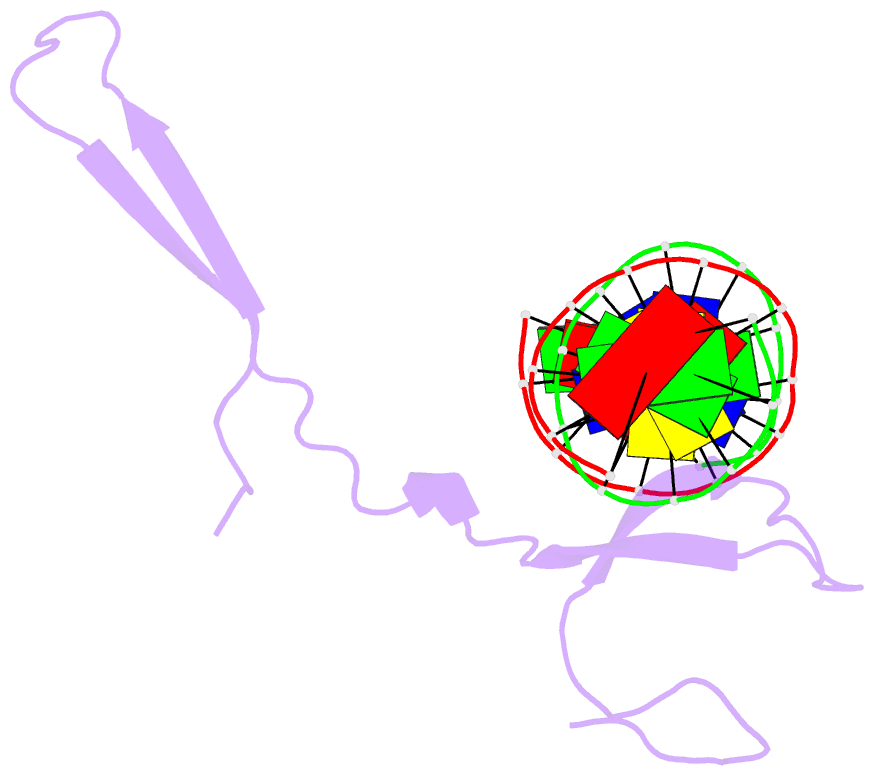
- Rice wrky-DNA complex
- Reference
- Cheng X, Zhao Y, Jiang Q, Yang J, Zhao W, Taylor IA, Peng YL, Wang D, Liu J (2019): "Structural basis of dimerization and dual W-box DNA recognition by rice WRKY domain." Nucleic Acids Res., 47, 4308-4318. doi: 10.1093/nar/gkz113.
- Abstract
- In rice, the critical regulator of the salicylic acid signalling pathway is OsWRKY45, a transcription factor (TF) of the WRKY TF family that functions by binding to the W-box of gene promoters, but the structural basis of OsWRKY45/W-box DNA recognition is unknown. Here, we show the crystal structure of the DNA binding domain of OsWRKY45 (OsWRKY45-DBD, i.e. the WRKY and zinc finger domain) in complex with a W-box DNA. Surprisingly, two OsWRKY45-DBD molecules exchange β4-β5 strands to form a dimer. The domain swapping occurs at the hinge region between the β3 and β4 strands, and is bridged and stabilized by zinc ion via coordinating residues from different chains. The dimer contains two identical DNA binding domains that interact with the major groove of W-box DNA. In addition to hydrophobic and direct hydrogen bonds, water mediated hydrogen bonds are also involved in base-specific interaction between protein and DNA. Finally, we discussed the cause and consequence of domain swapping of OsWRKY45-DBD, and based on our work and that of previous studies present a detailed mechanism of W-box recognition by WRKY TFs. This work reveals a novel dimerization and DNA-binding mode of WRKY TFs, and an intricate picture of the WRKY/W-box DNA recognition.