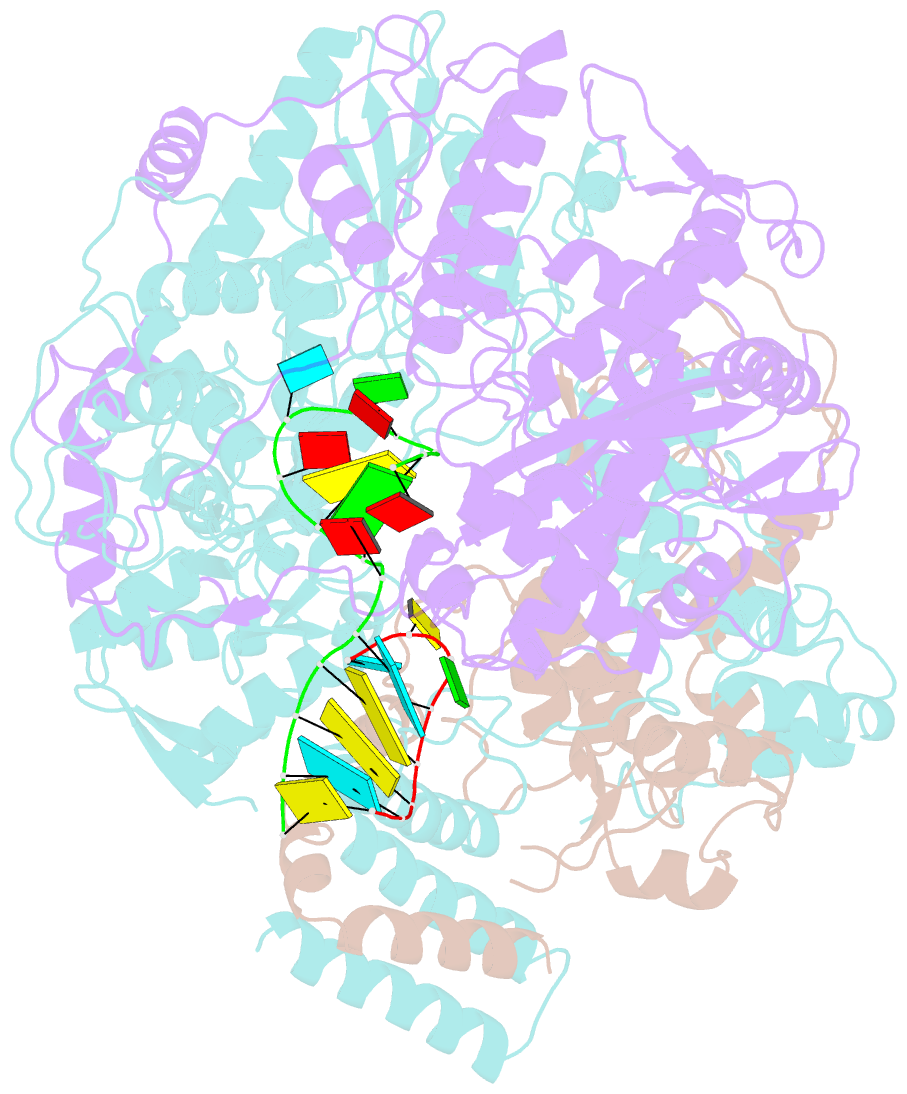
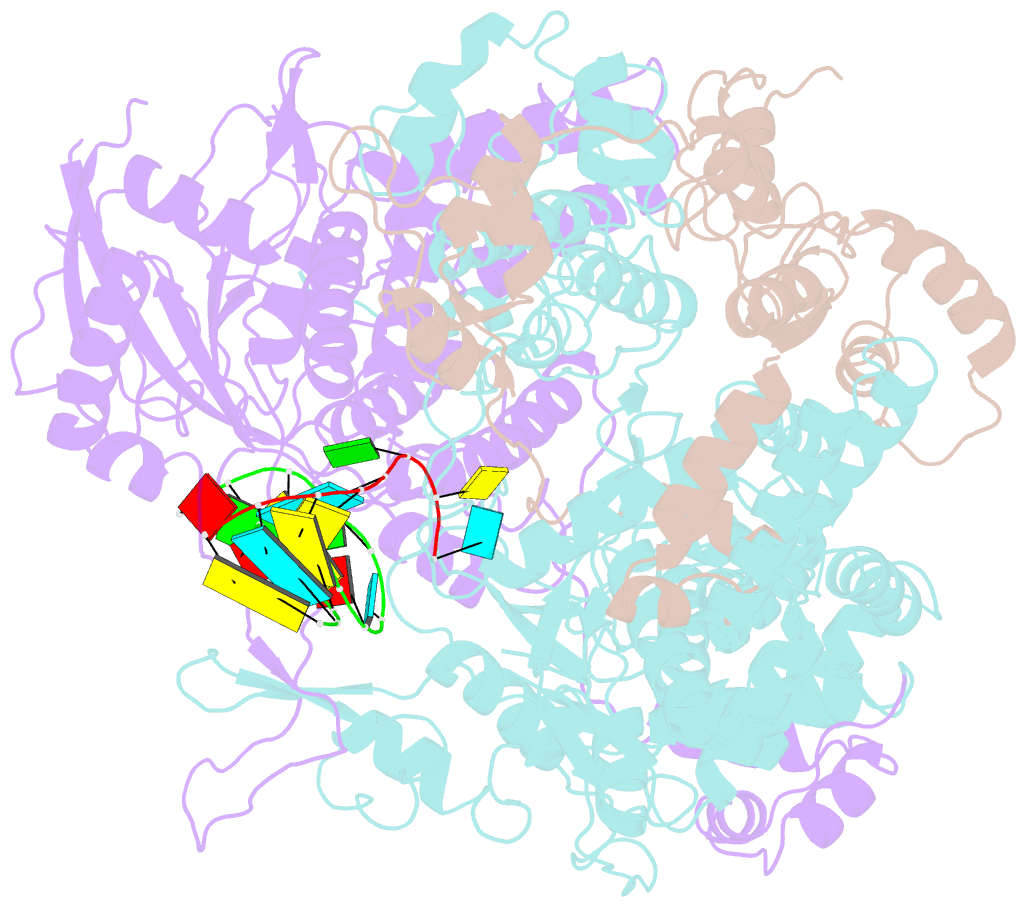
Summary information and primary citation
- PDB-id
- 6kuk; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- viral protein-RNA
- Method
- cryo-EM (3.9 Å)
- Summary
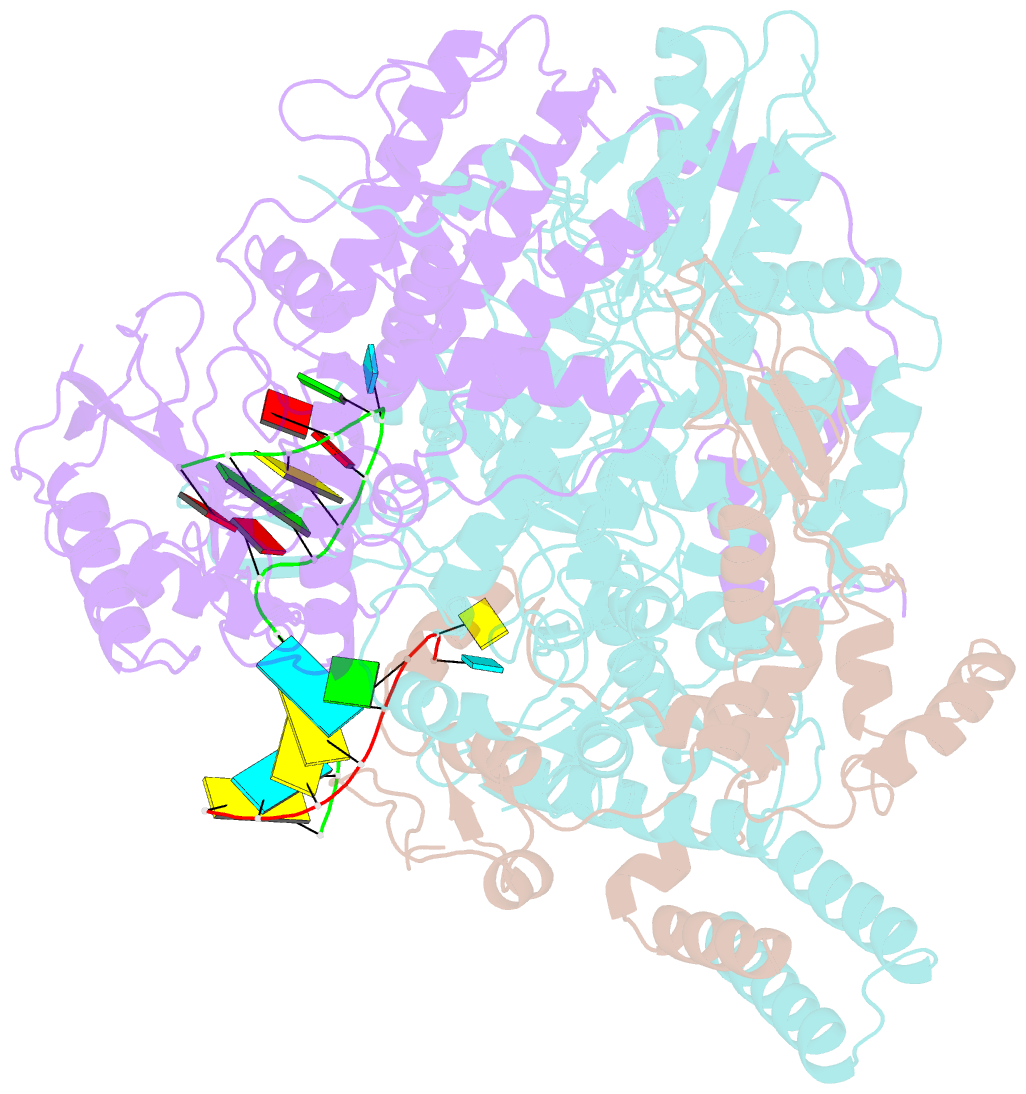
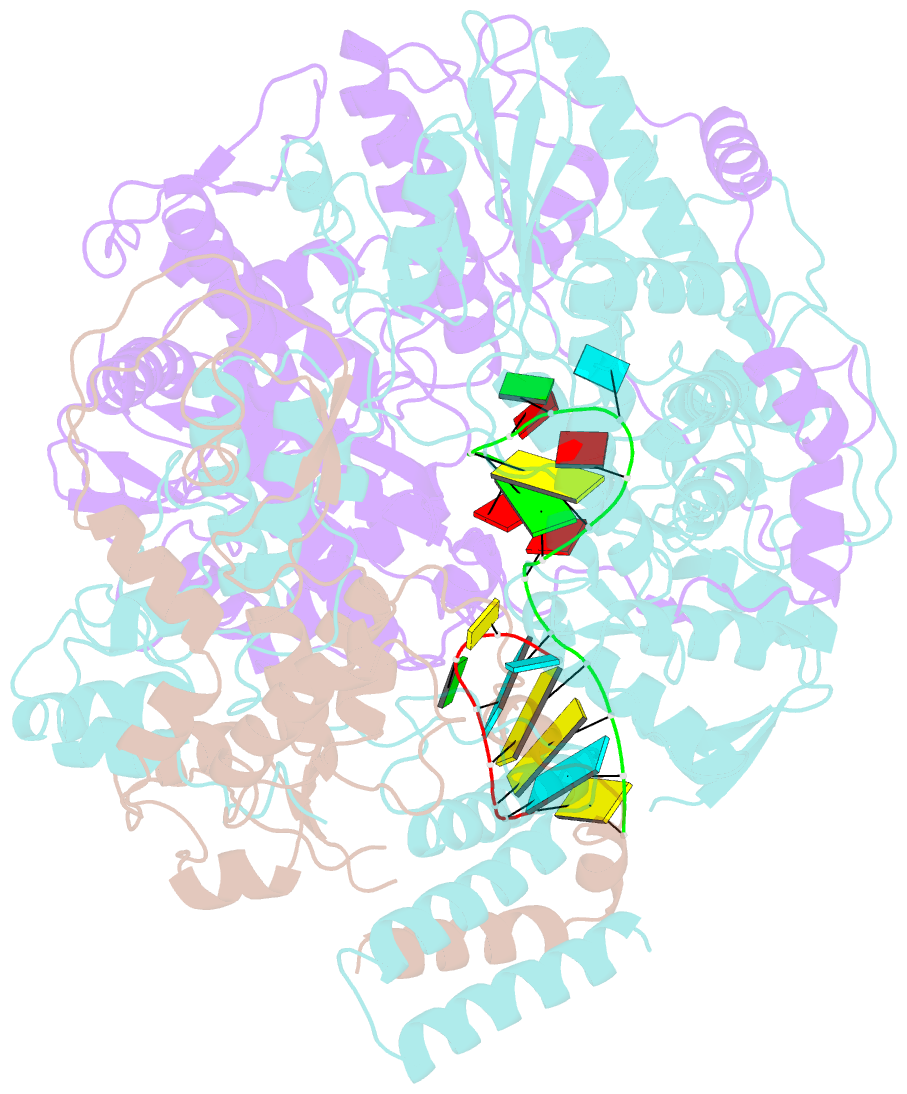
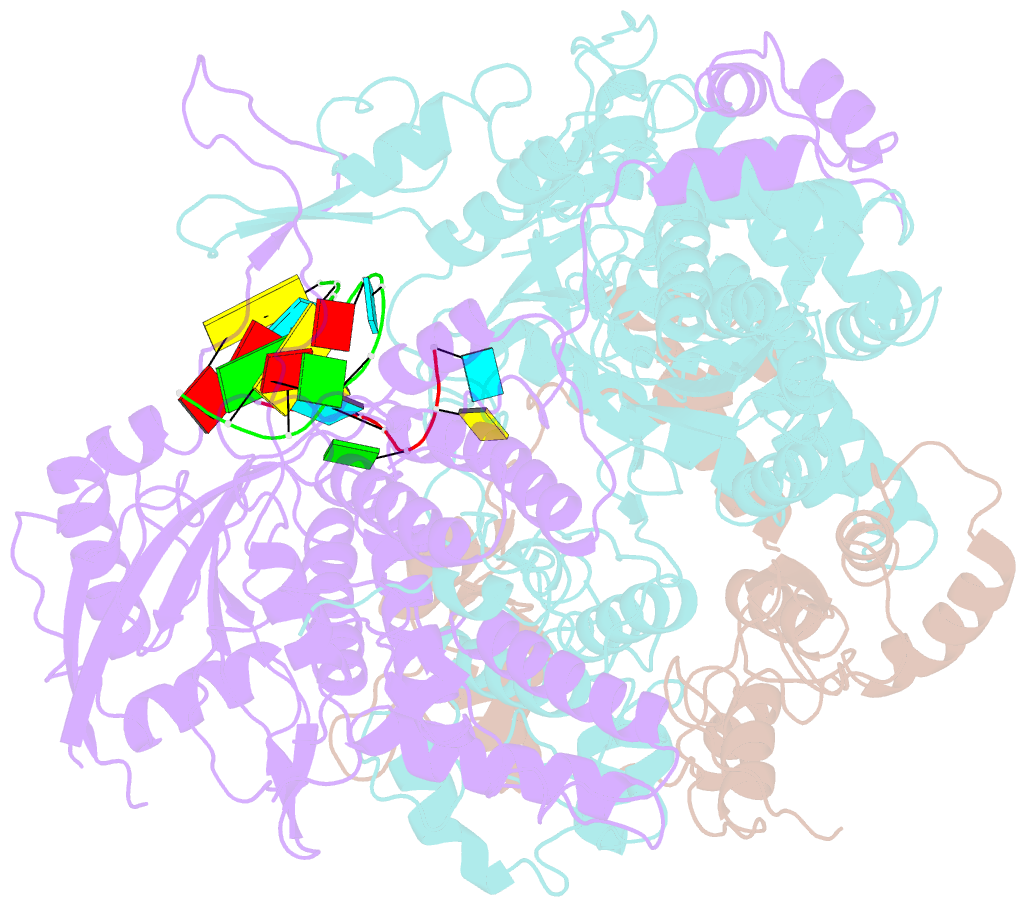
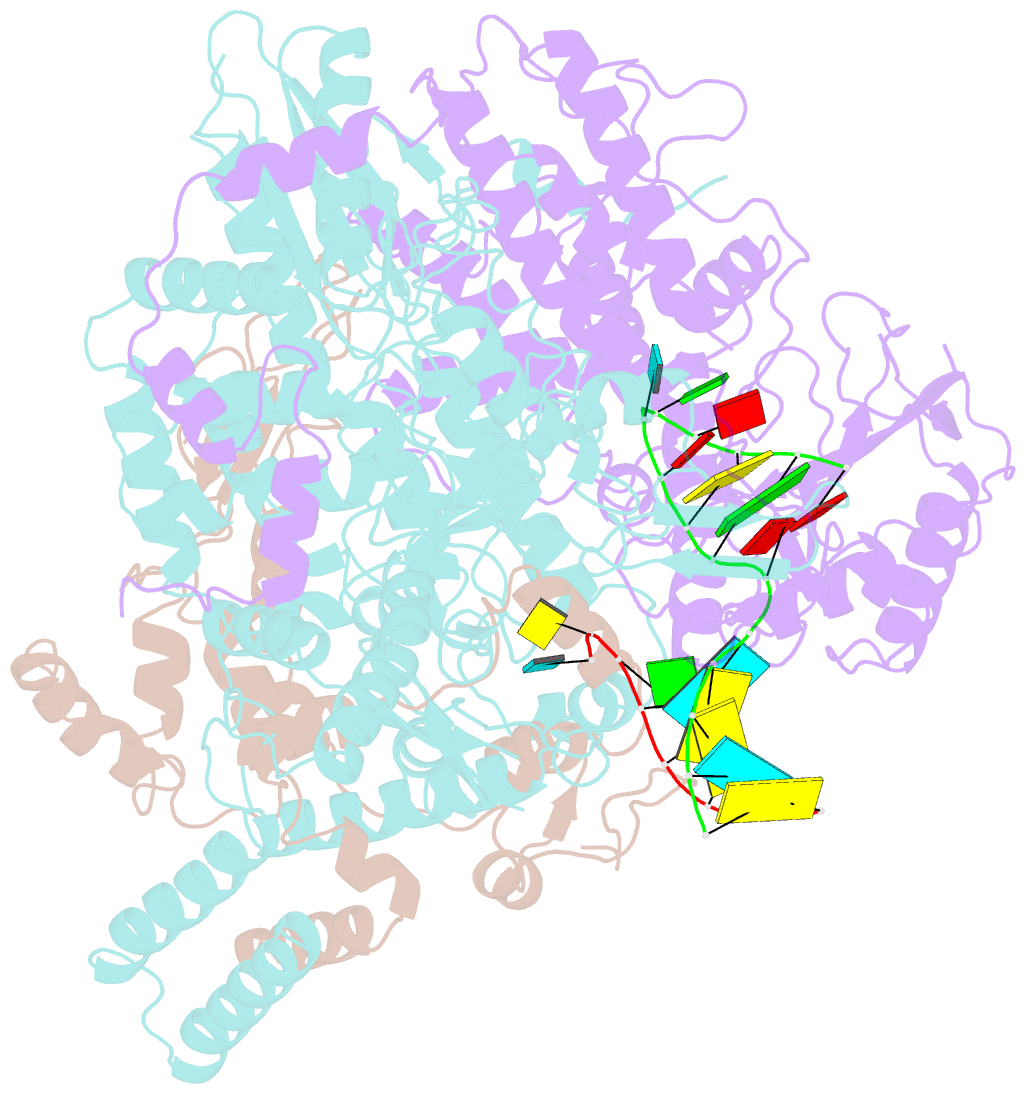
- Structure of influenza d virus polymerase bound to vrna promoter in mode a conformation (class a1)
- Reference
- Peng Q, Liu Y, Peng R, Wang M, Yang W, Song H, Chen Y, Liu S, Han M, Zhang X, Wang P, Yan J, Zhang B, Qi J, Deng T, Gao GF, Shi Y (2019): "Structural insight into RNA synthesis by influenza D polymerase." Nat Microbiol, 4, 1750-1759. doi: 10.1038/s41564-019-0487-5.
- Abstract
- The influenza virus polymerase uses capped RNA primers to initiate transcription, and a combination of terminal and internal de novo initiations for the two-step replication process by binding the conserved viral genomic RNA (vRNA) or complementary RNA (cRNA) promoter. Here, we determined the apo and promoter-bound influenza D polymerase structures using cryo-electron microscopy and found the polymerase has an evolutionarily conserved stable core structure with inherently flexible peripheral domains. Strikingly, two conformations (mode A and B) of the vRNA promoter were observed where the 3'-vRNA end can bind at two different sites, whereas the cRNA promoter only binds in the mode B conformation. Functional studies confirmed the critical role of the mode B conformation for vRNA synthesis via the intermediate cRNA but not for cRNA production, which is mainly regulated by the mode A conformation. Both conformations participate in the regulation of the transcription process. This work advances our understanding of the regulatory mechanisms for the synthesis of different RNA species by influenza virus polymerase and opens new opportunities for antiviral drug design.