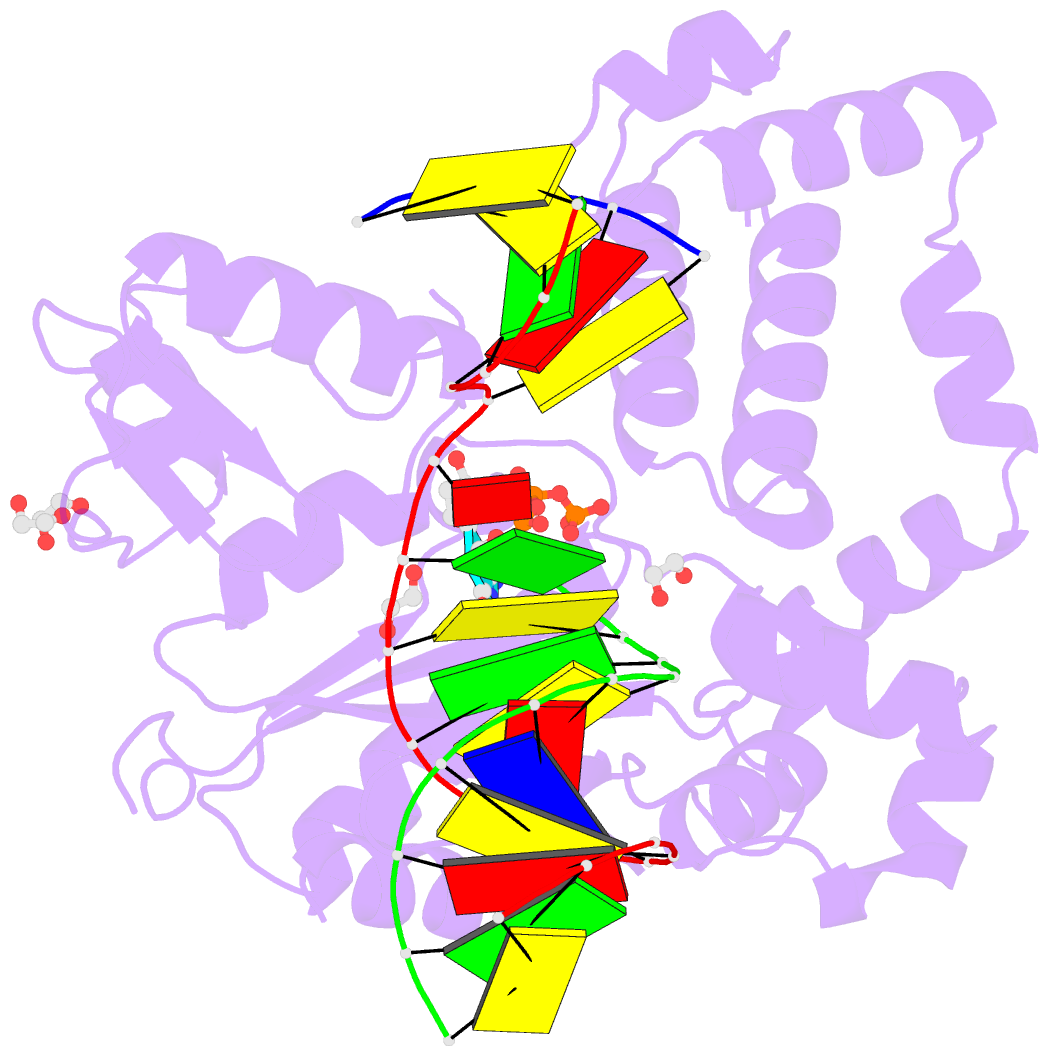
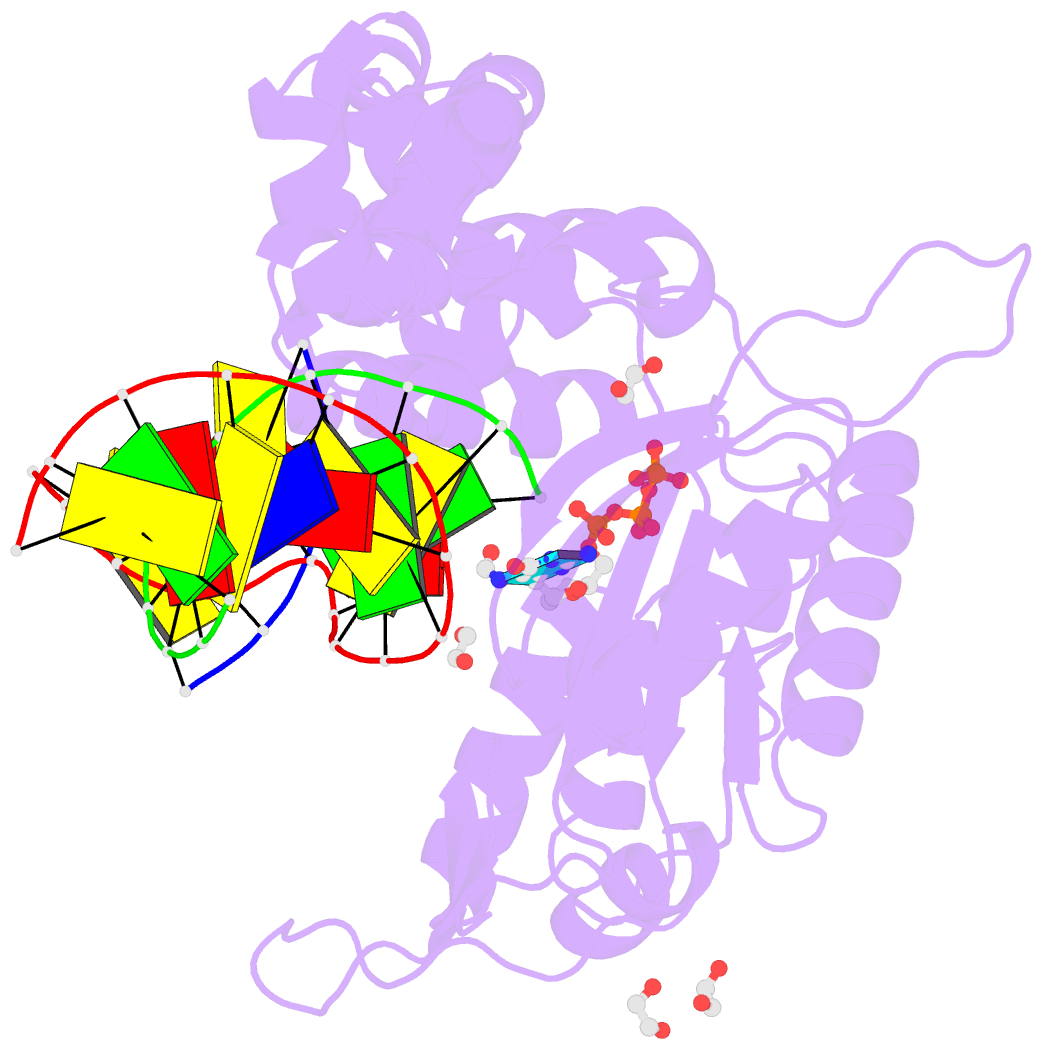
Summary information and primary citation
- PDB-id
- 6mr7; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- DNA binding protein-DNA
- Method
- X-ray (2.144 Å)
- Summary
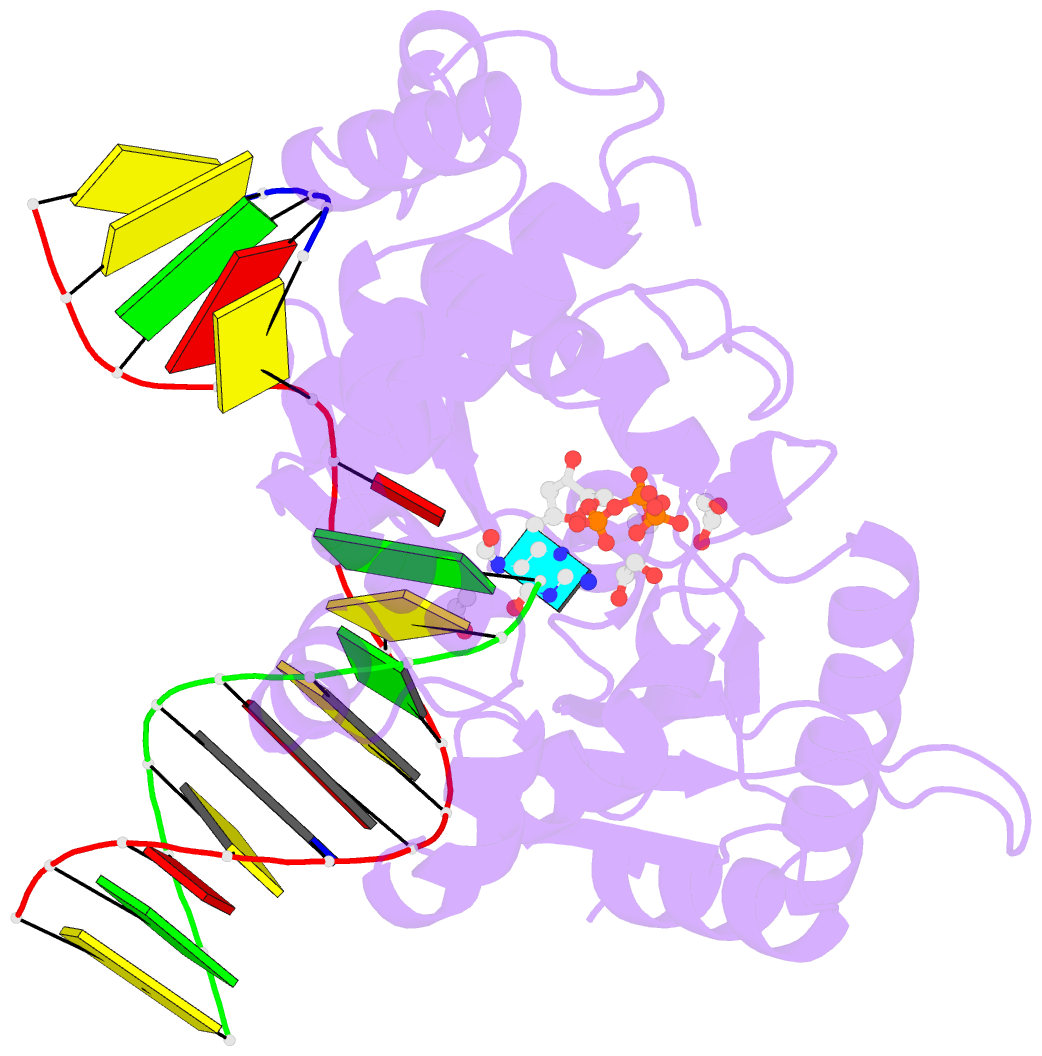
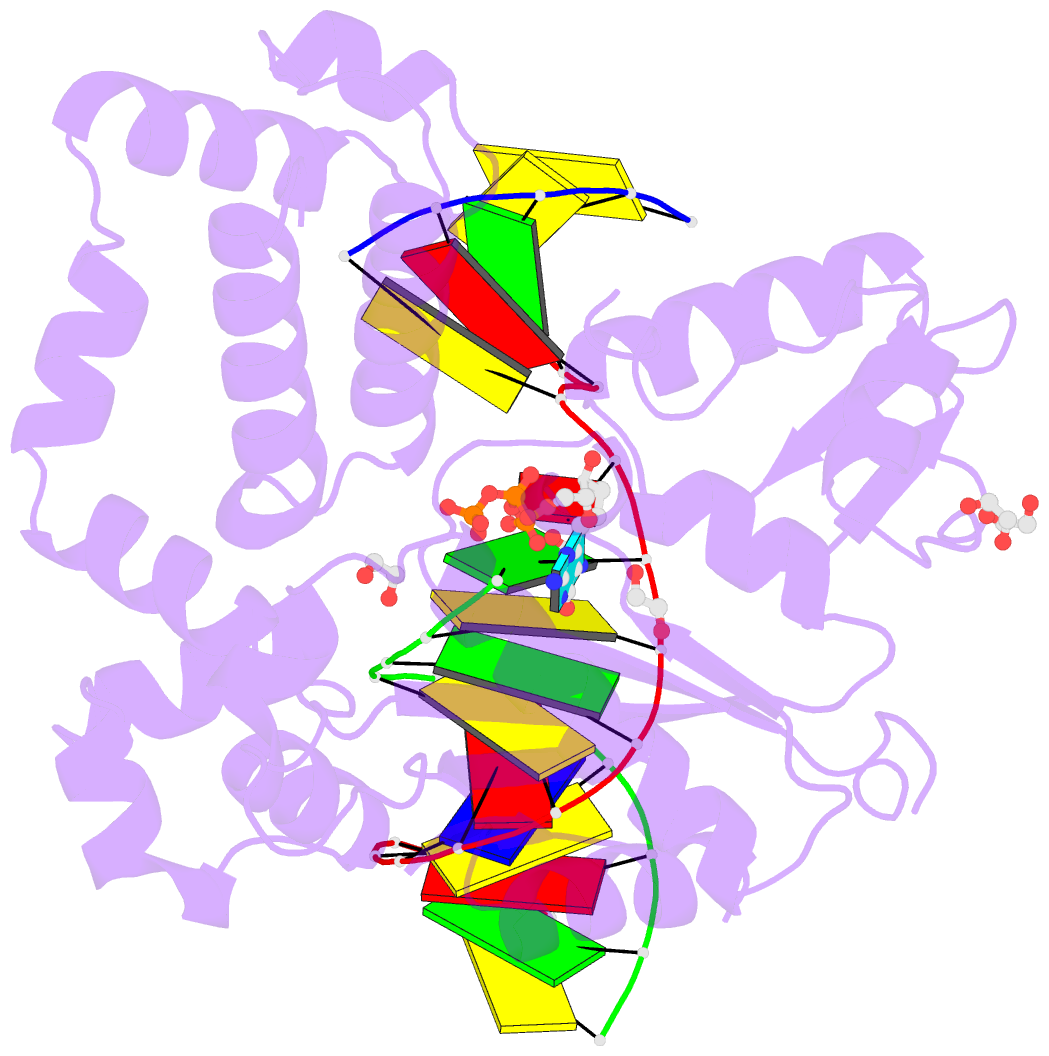
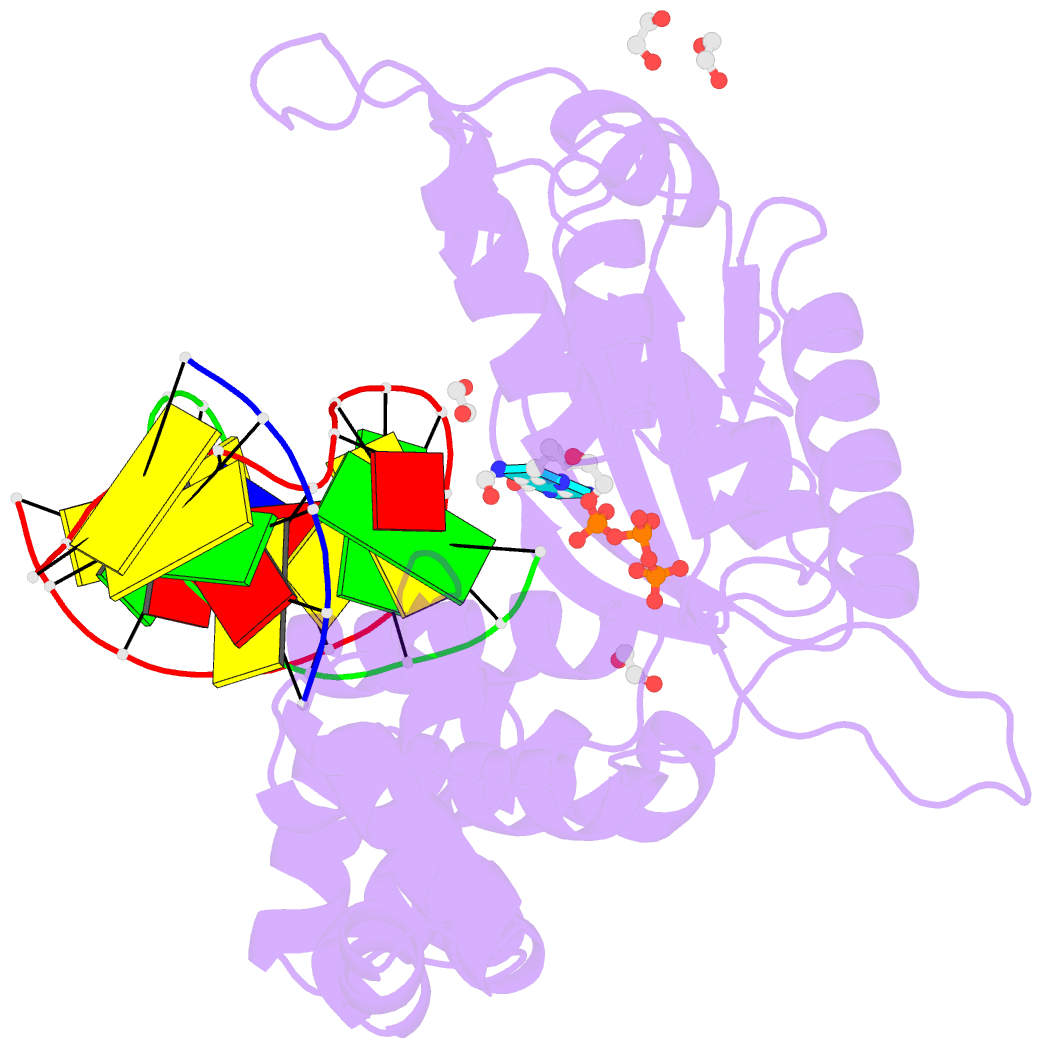
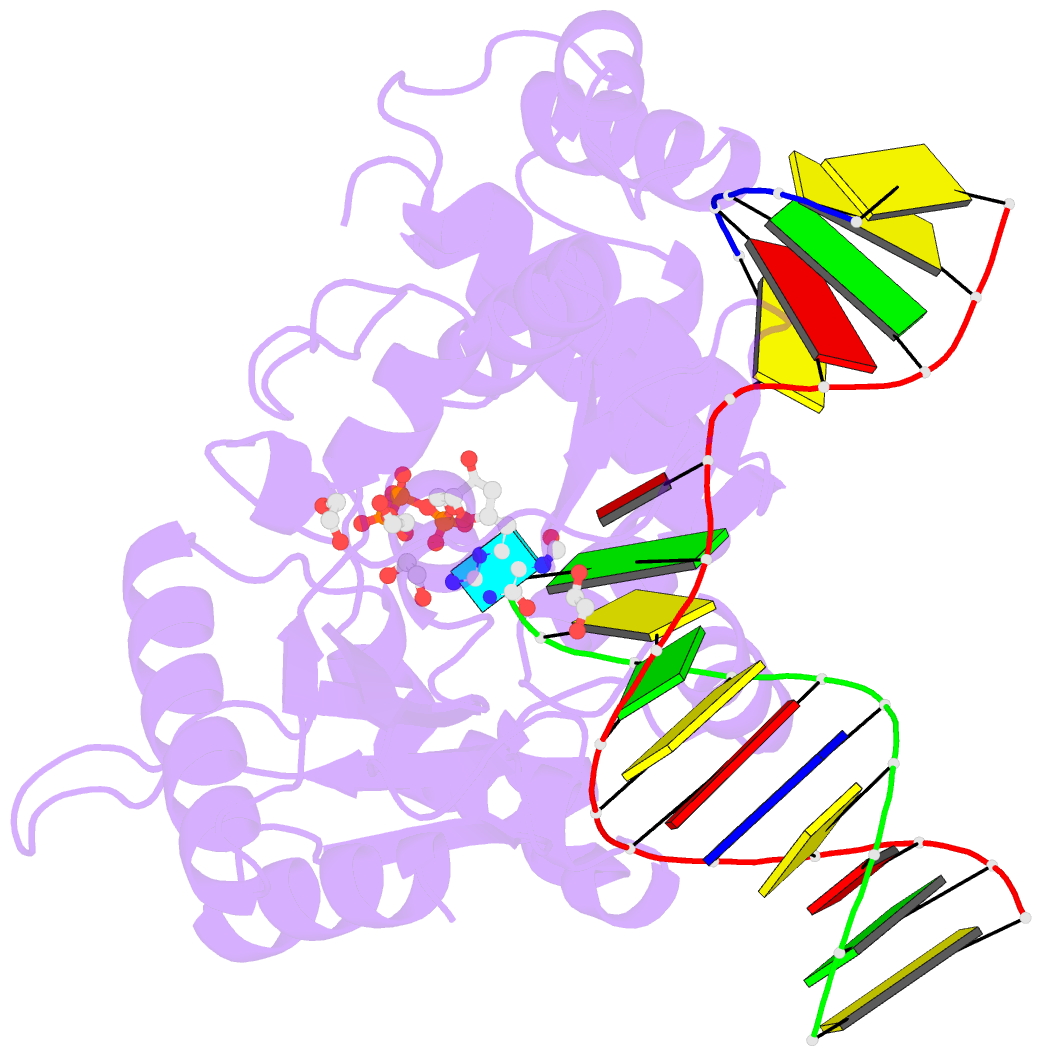
- DNA polymerase beta substrate complex with templating adenine and incoming fapy-dgtp analog
- Reference
- Smith MR, Shock DD, Beard WA, Greenberg MM, Freudenthal BD, Wilson SH (2019): "A guardian residue hinders insertion of a Fapy•dGTP analog by modulating the open-closed DNA polymerase transition." Nucleic Acids Res., 47, 3197-3207. doi: 10.1093/nar/gkz002.
- Abstract
- 4,6-Diamino-5-formamidopyrimidine (Fapy•dG) is an abundant form of oxidative DNA damage that is mutagenic and contributes to the pathogenesis of human disease. When Fapy•dG is in its nucleotide triphosphate form, Fapy•dGTP, it is inefficiently cleansed from the nucleotide pool by the responsible enzyme in Escherichia coli MutT and its mammalian homolog MTH1. Therefore, under oxidative stress conditions, Fapy•dGTP could become a pro-mutagenic substrate for insertion into the genome by DNA polymerases. Here, we evaluated insertion kinetics and high-resolution ternary complex crystal structures of a configurationally stable Fapy•dGTP analog, β-C-Fapy•dGTP, with DNA polymerase β. The crystallographic snapshots and kinetic data indicate that binding of β-C-Fapy•dGTP impedes enzyme closure, thus hindering insertion. The structures reveal that an active site residue, Asp276, positions β-C-Fapy•dGTP so that it distorts the geometry of critical catalytic atoms. Removal of this guardian side chain permits enzyme closure and increases the efficiency of β-C-Fapy•dG insertion opposite dC. These results highlight the stringent requirements necessary to achieve a closed DNA polymerase active site poised for efficient nucleotide incorporation and illustrate how DNA polymerase β has evolved to hinder Fapy•dGTP insertion.