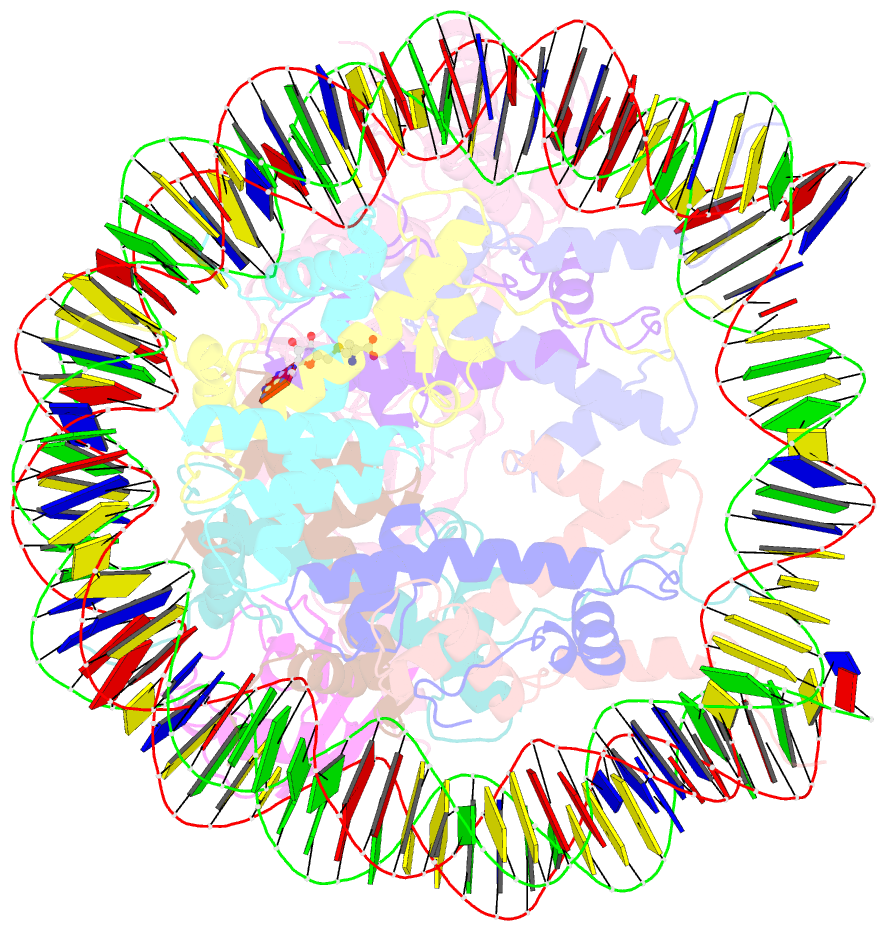
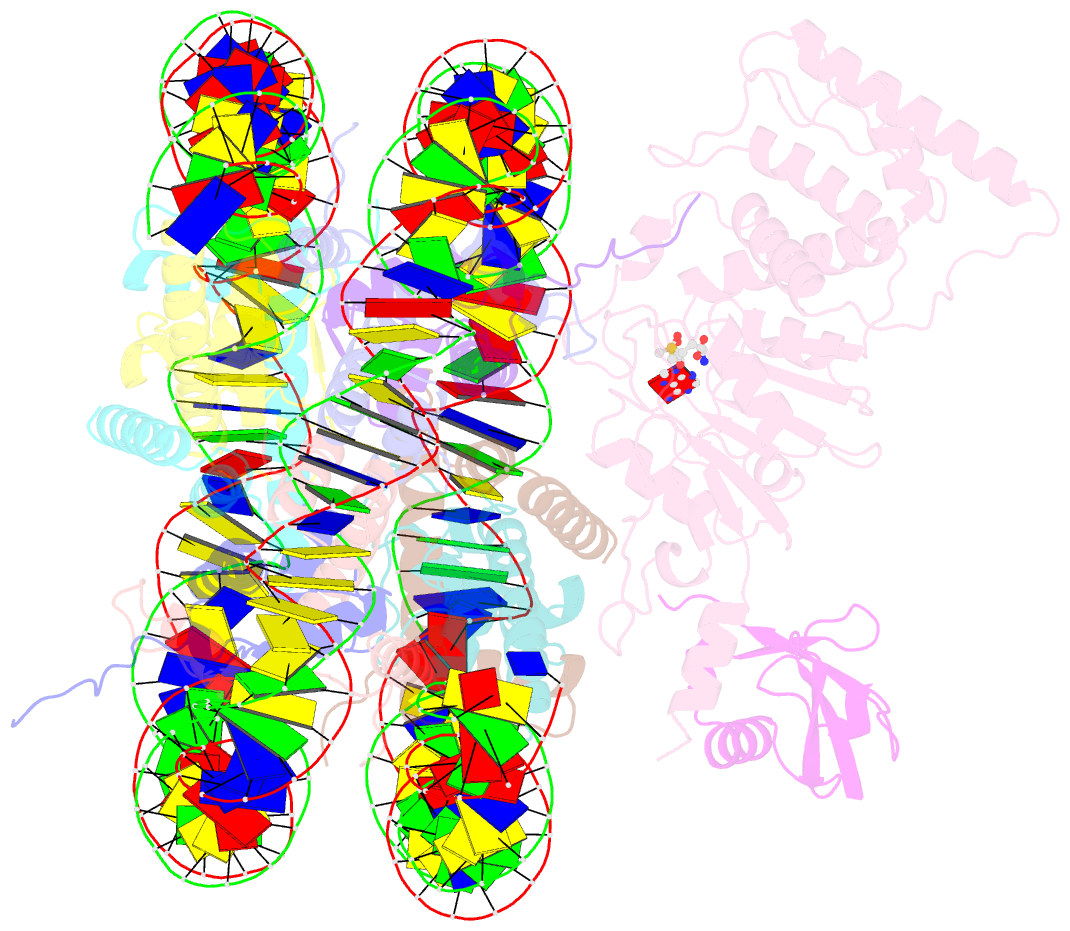
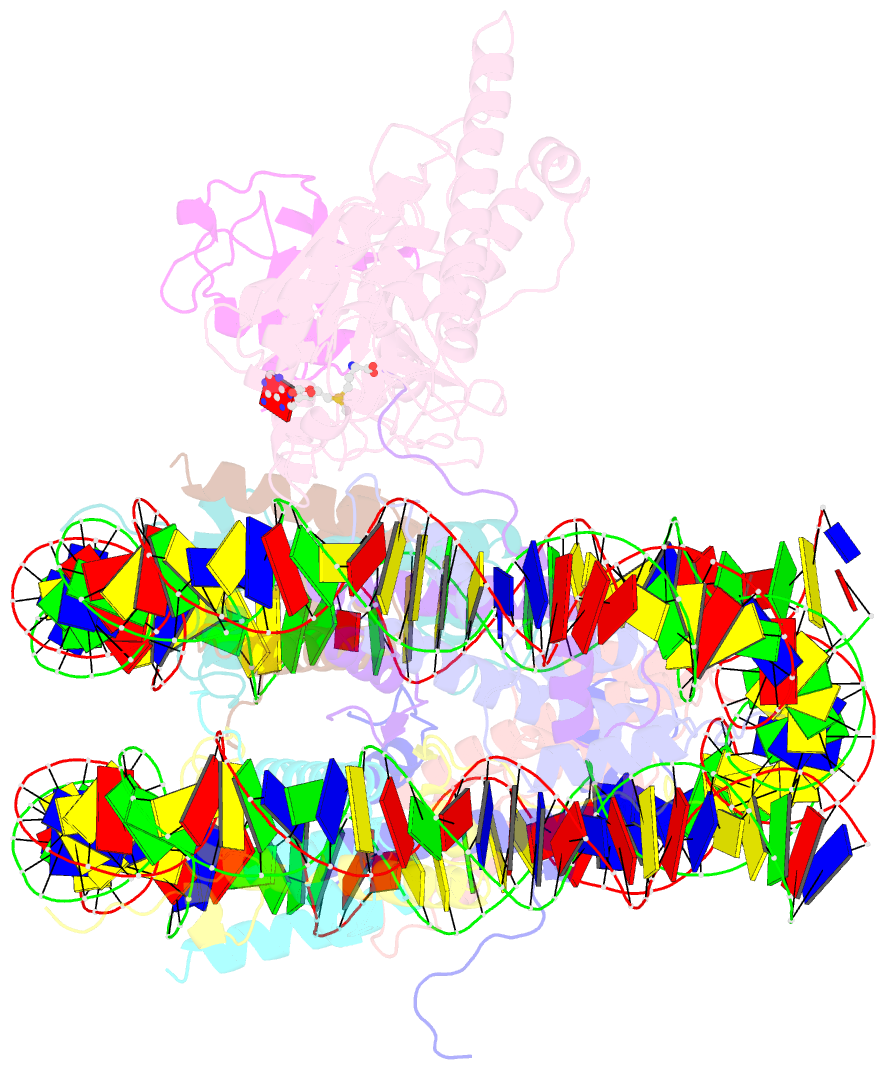
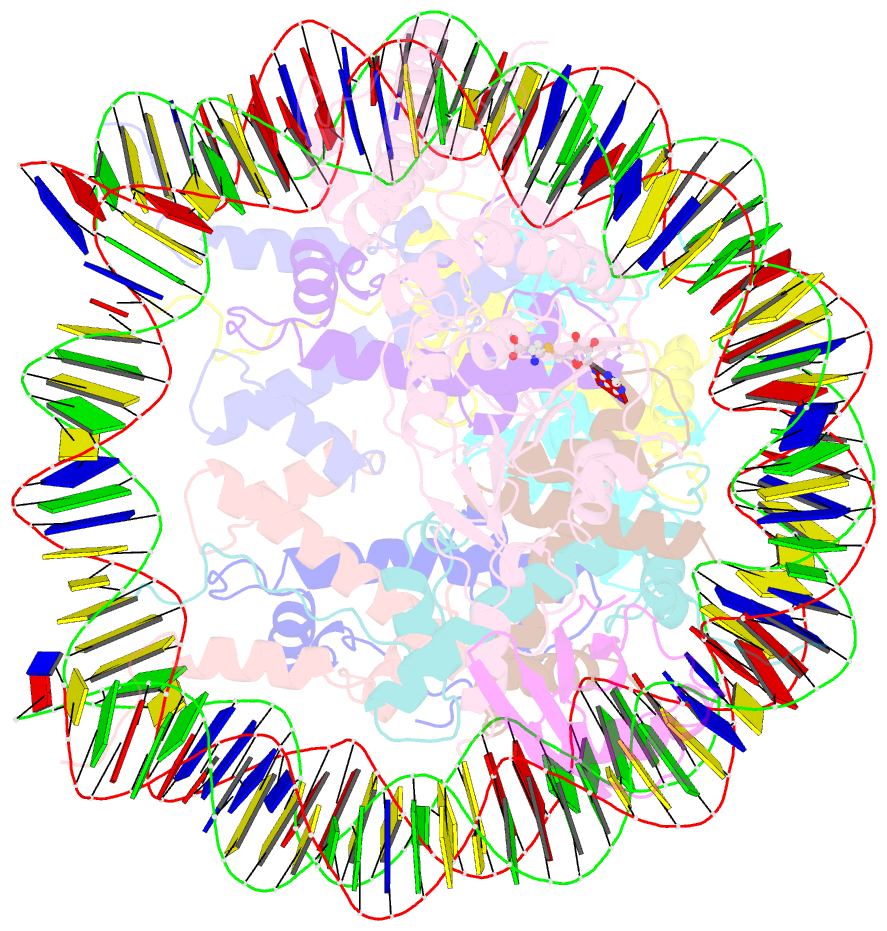
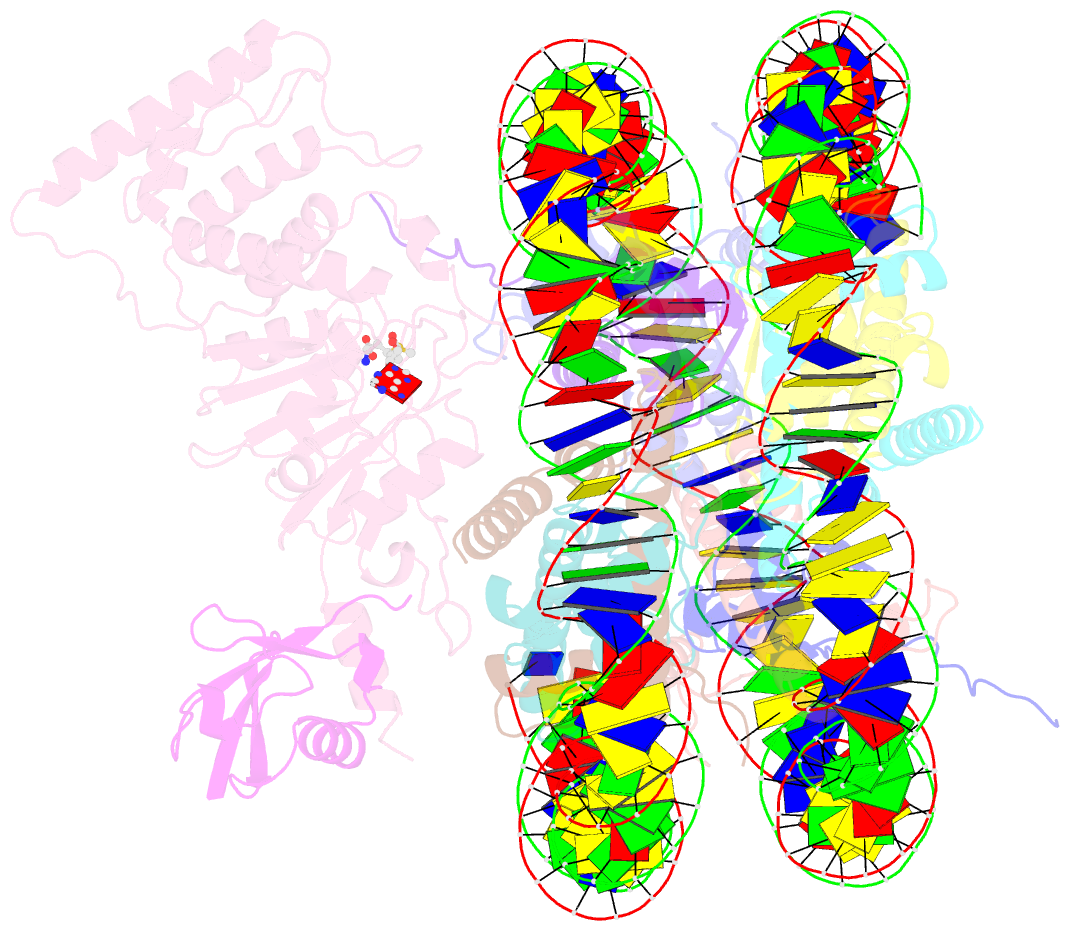
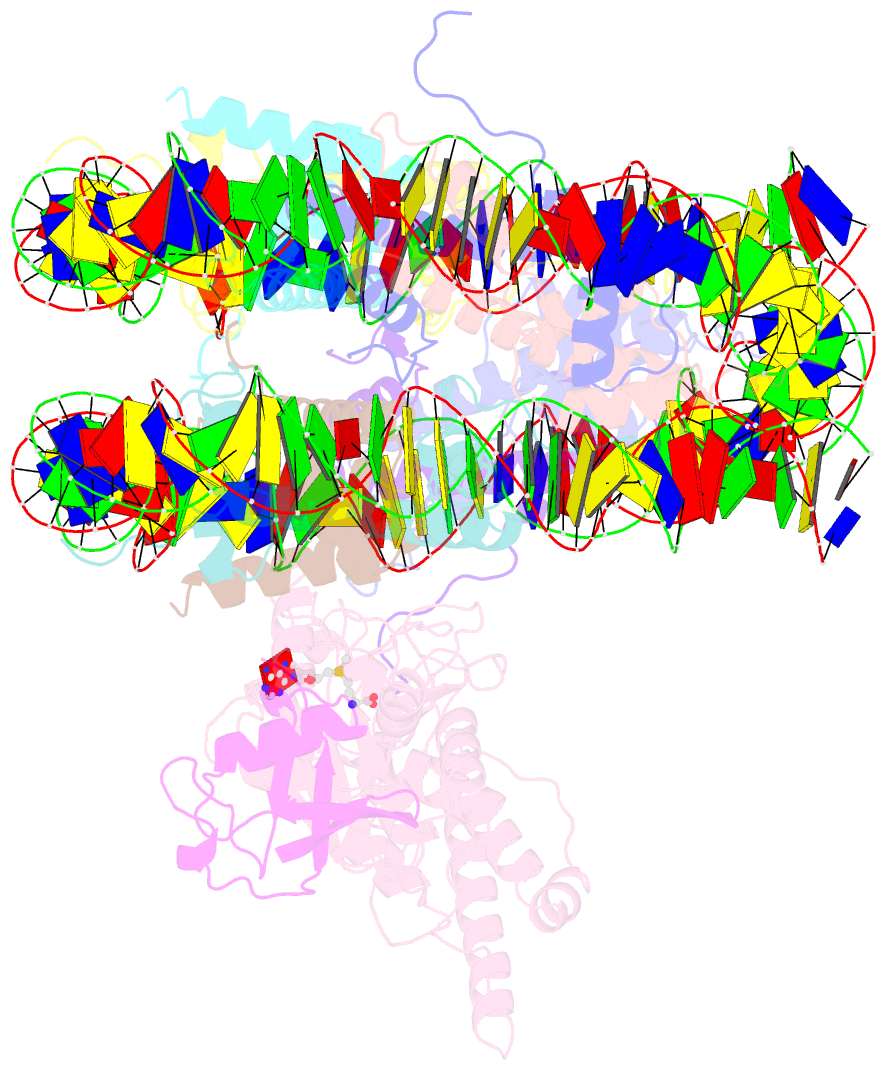
Summary information and primary citation
- PDB-id
- 6nj9; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- structural protein-transferase-DNA
- Method
- cryo-EM (2.96 Å)
- Summary
- Active state dot1l bound to the h2b-ubiquitinated nucleosome, 2-to-1 complex
- Reference
- Worden EJ, Hoffmann NA, Hicks CW, Wolberger C (2019): "Mechanism of Cross-talk between H2B Ubiquitination and H3 Methylation by Dot1L." Cell, 176, 1490-1501.e12. doi: 10.1016/j.cell.2019.02.002.
- Abstract
- Methylation of histone H3 K79 by Dot1L is a hallmark of actively transcribed genes that depends on monoubiquitination of H2B K120 (H2B-Ub) and is an example of histone modification cross-talk that is conserved from yeast to humans. We report here cryo-EM structures of Dot1L bound to ubiquitinated nucleosome that show how H2B-Ub stimulates Dot1L activity and reveal a role for the histone H4 tail in positioning Dot1L. We find that contacts mediated by Dot1L and the H4 tail induce a conformational change in the globular core of histone H3 that reorients K79 from an inaccessible position, thus enabling this side chain to insert into the active site in a position primed for catalysis. Our study provides a comprehensive mechanism of cross-talk between histone ubiquitination and methylation and reveals structural plasticity in histones that makes it possible for histone-modifying enzymes to access residues within the nucleosome core.