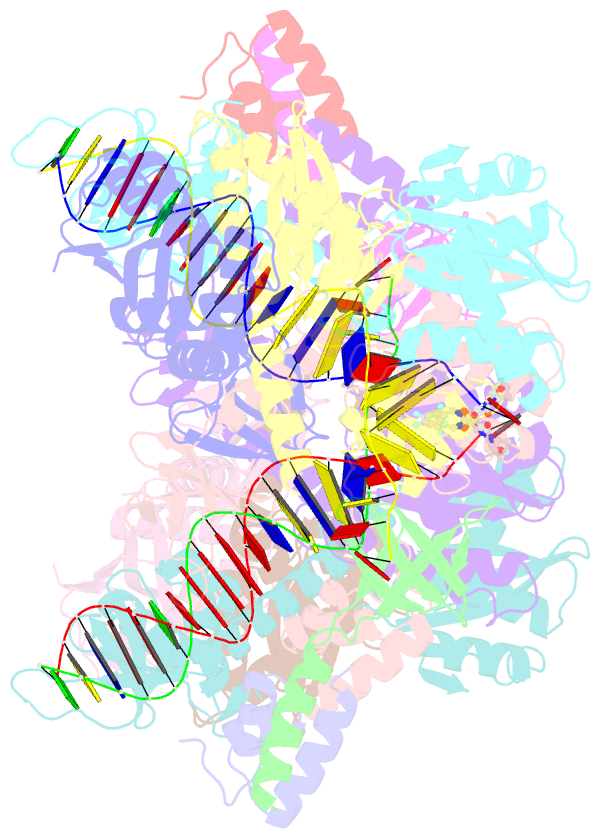
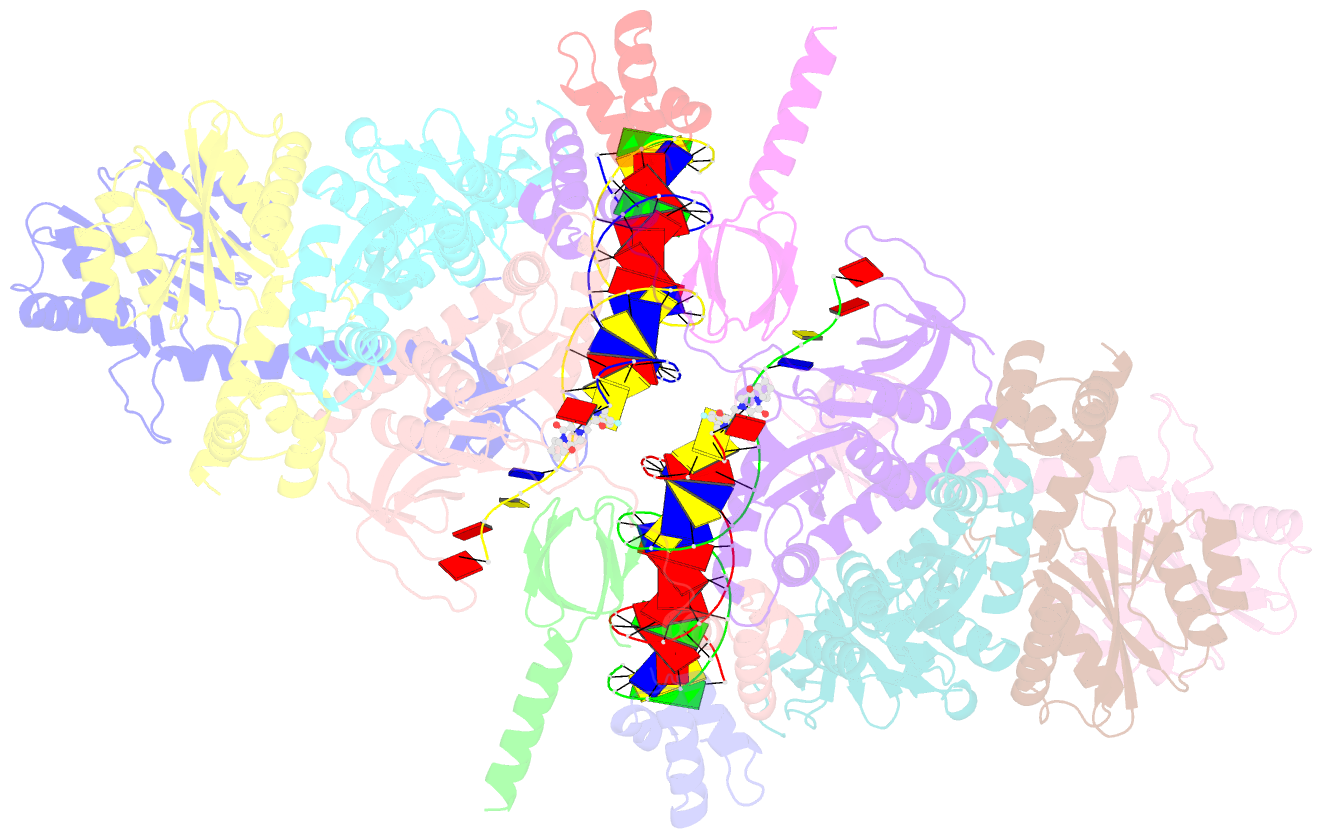
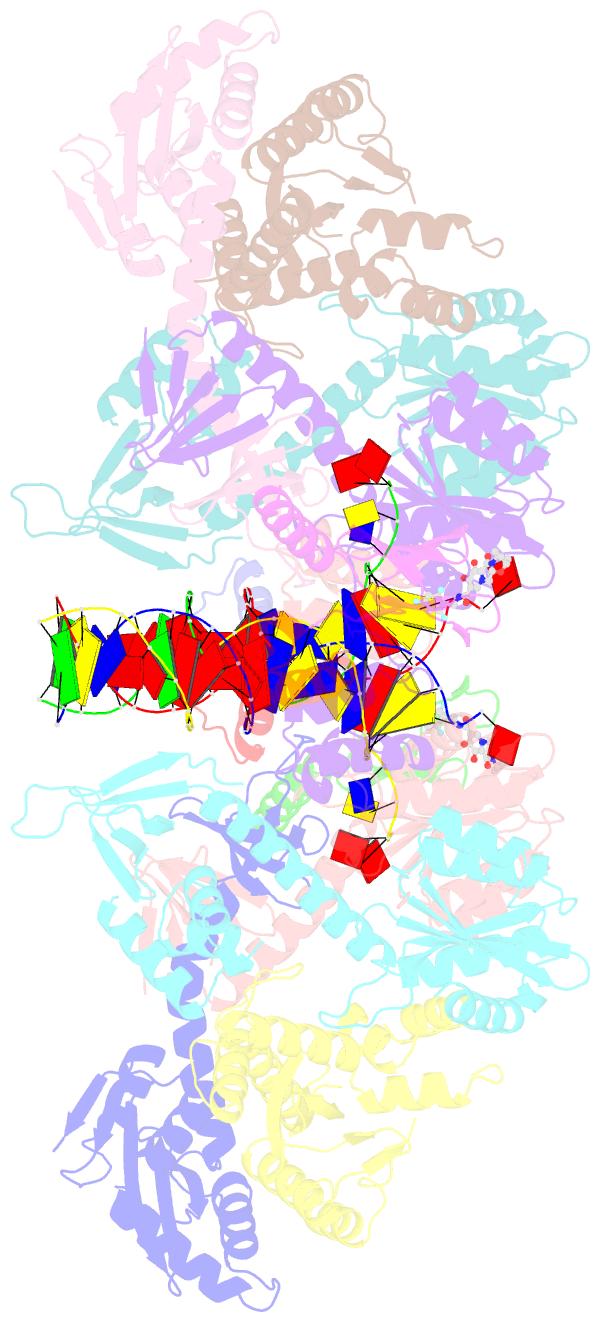
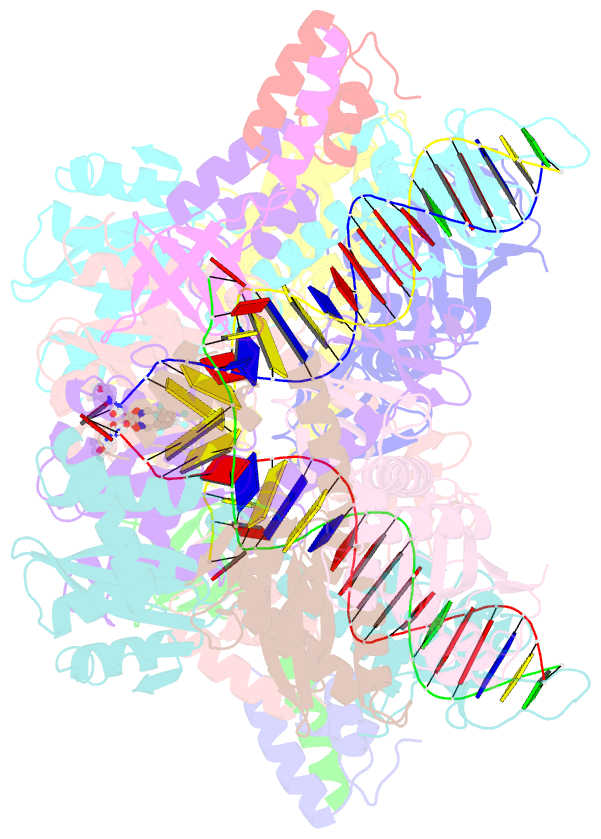
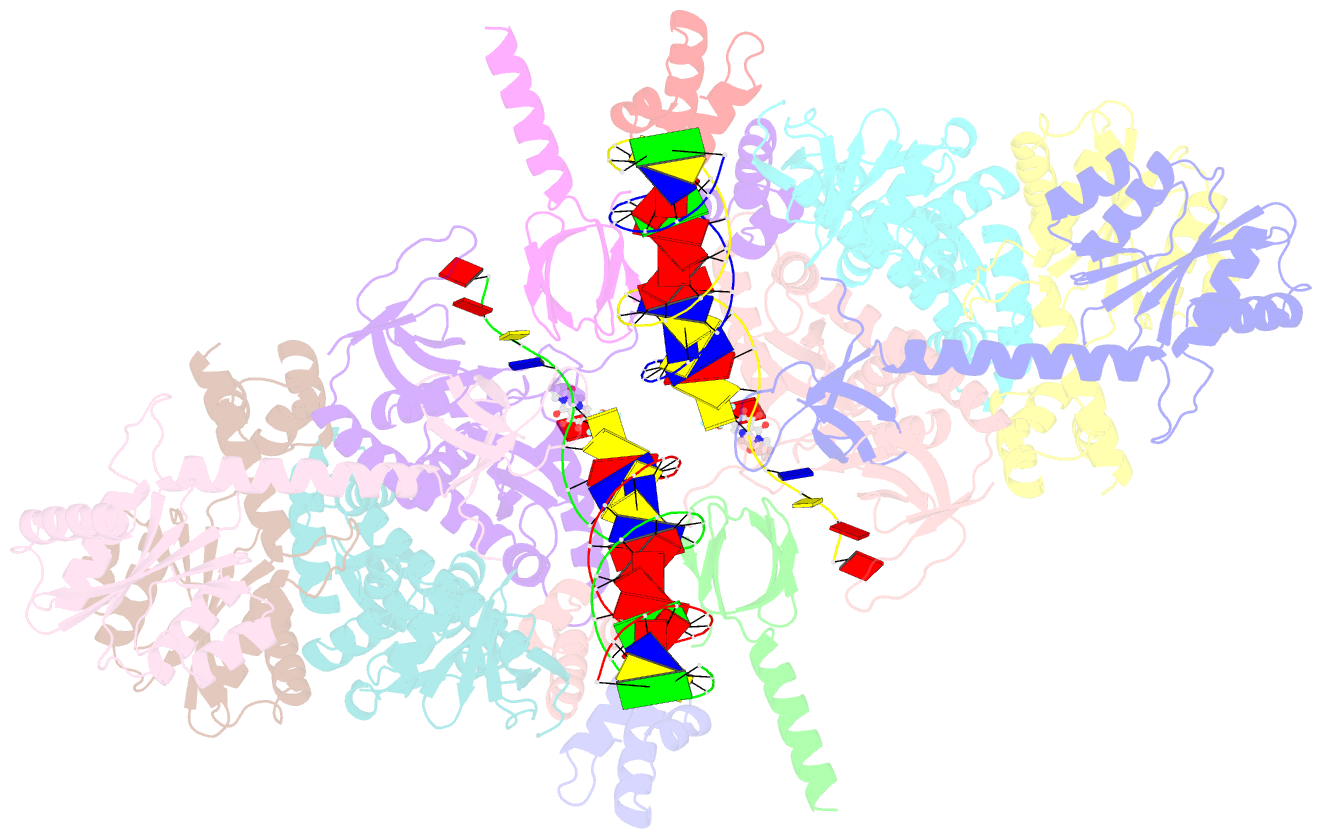
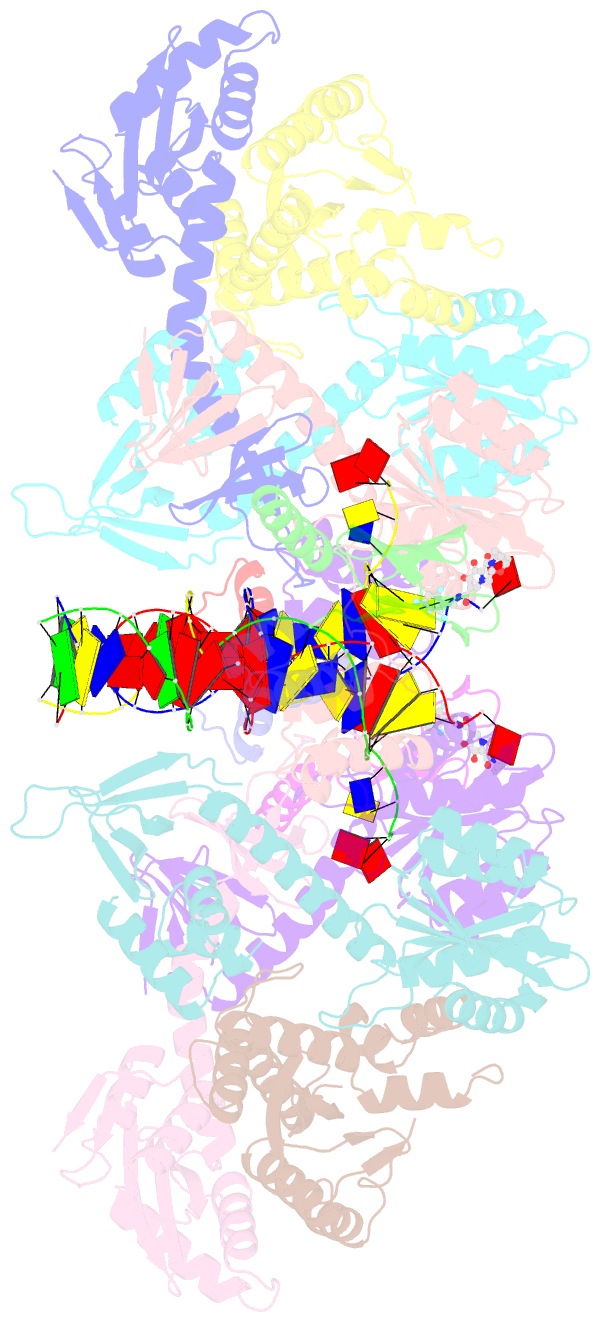
Summary information and primary citation
- PDB-id
- 6rwm; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- recombination
- Method
- cryo-EM (2.81 Å)
- Summary
- Sivrcm intasome in complex with bictegravir
- Reference
- Cook NJ, Li W, Berta D, Badaoui M, Ballandras-Colas A, Nans A, Kotecha A, Rosta E, Engelman AN, Cherepanov P (2020): "Structural basis of second-generation HIV integrase inhibitor action and viral resistance." Science, 367, 806-810. doi: 10.1126/science.aay4919.
- Abstract
- Although second-generation HIV integrase strand-transfer inhibitors (INSTIs) are prescribed throughout the world, the mechanistic basis for the superiority of these drugs is poorly understood. We used single-particle cryo-electron microscopy to visualize the mode of action of the advanced INSTIs dolutegravir and bictegravir at near-atomic resolution. Glutamine-148→histidine (Q148H) and glycine-140→serine (G140S) amino acid substitutions in integrase that result in clinical INSTI failure perturb optimal magnesium ion coordination in the enzyme active site. The expanded chemical scaffolds of second-generation compounds mediate interactions with the protein backbone that are critical for antagonizing viruses containing the Q148H and G140S mutations. Our results reveal that binding to magnesium ions underpins a fundamental weakness of the INSTI pharmacophore that is exploited by the virus to engender resistance and provide a structural framework for the development of this class of anti-HIV/AIDS therapeutics.