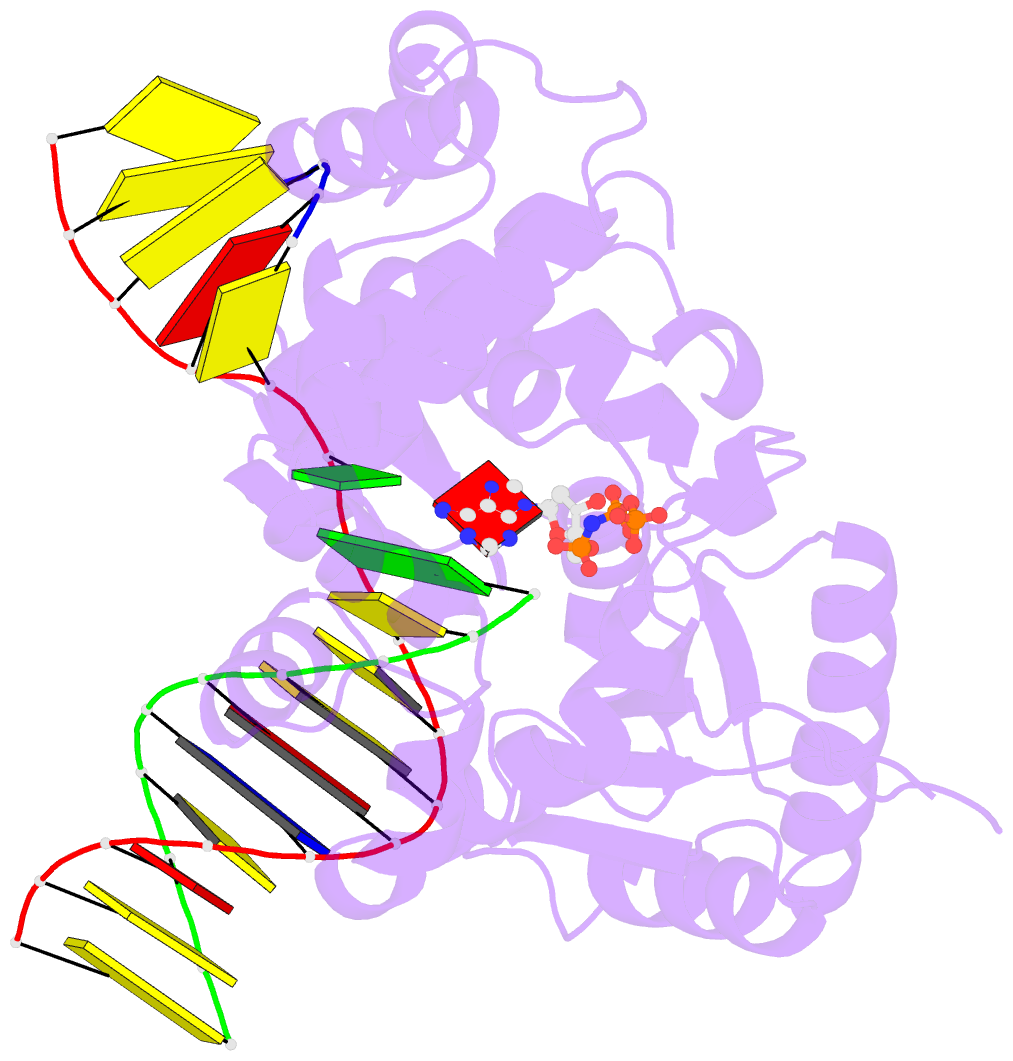
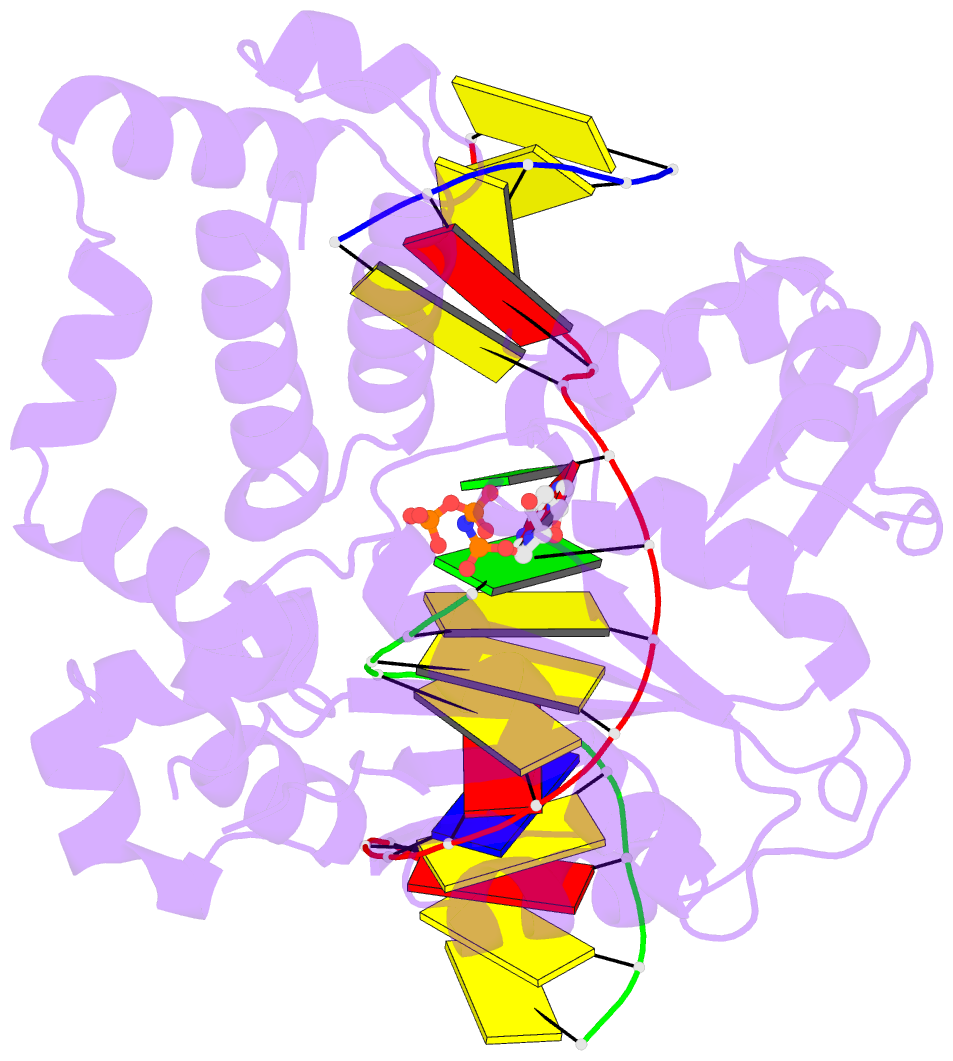
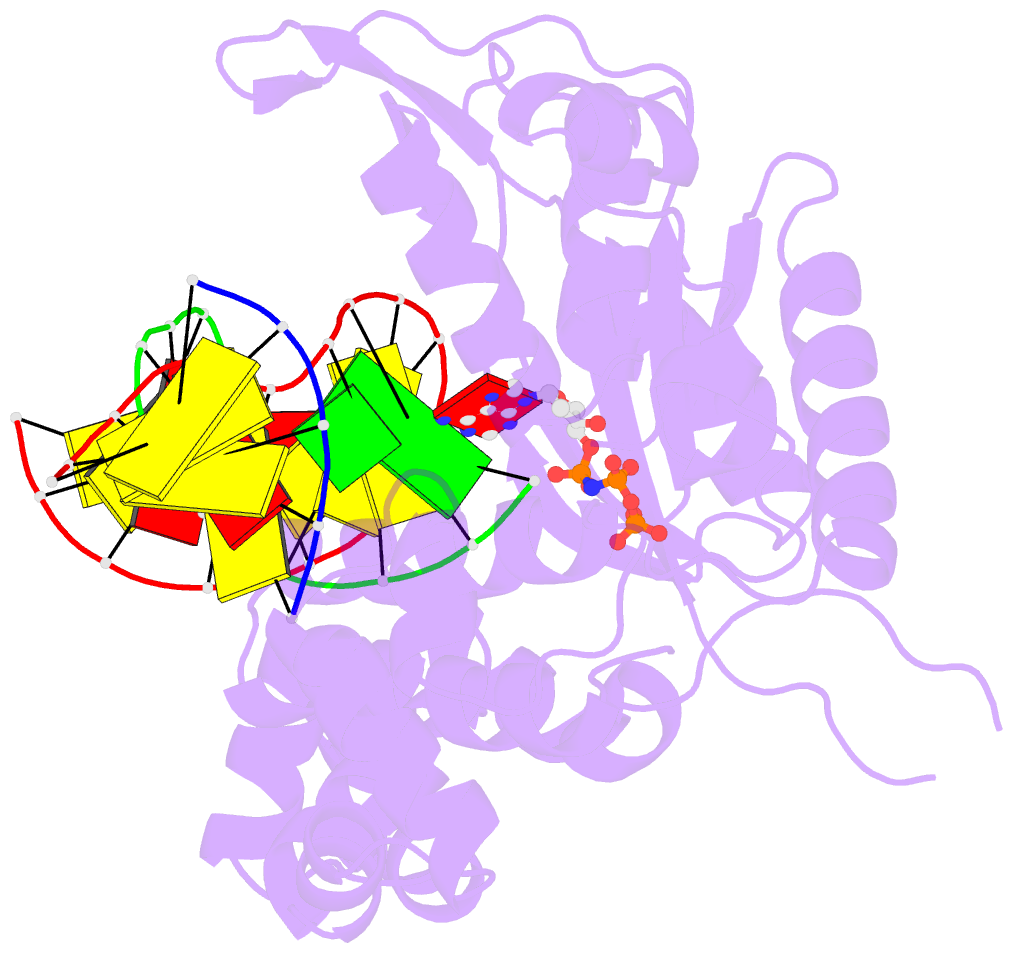
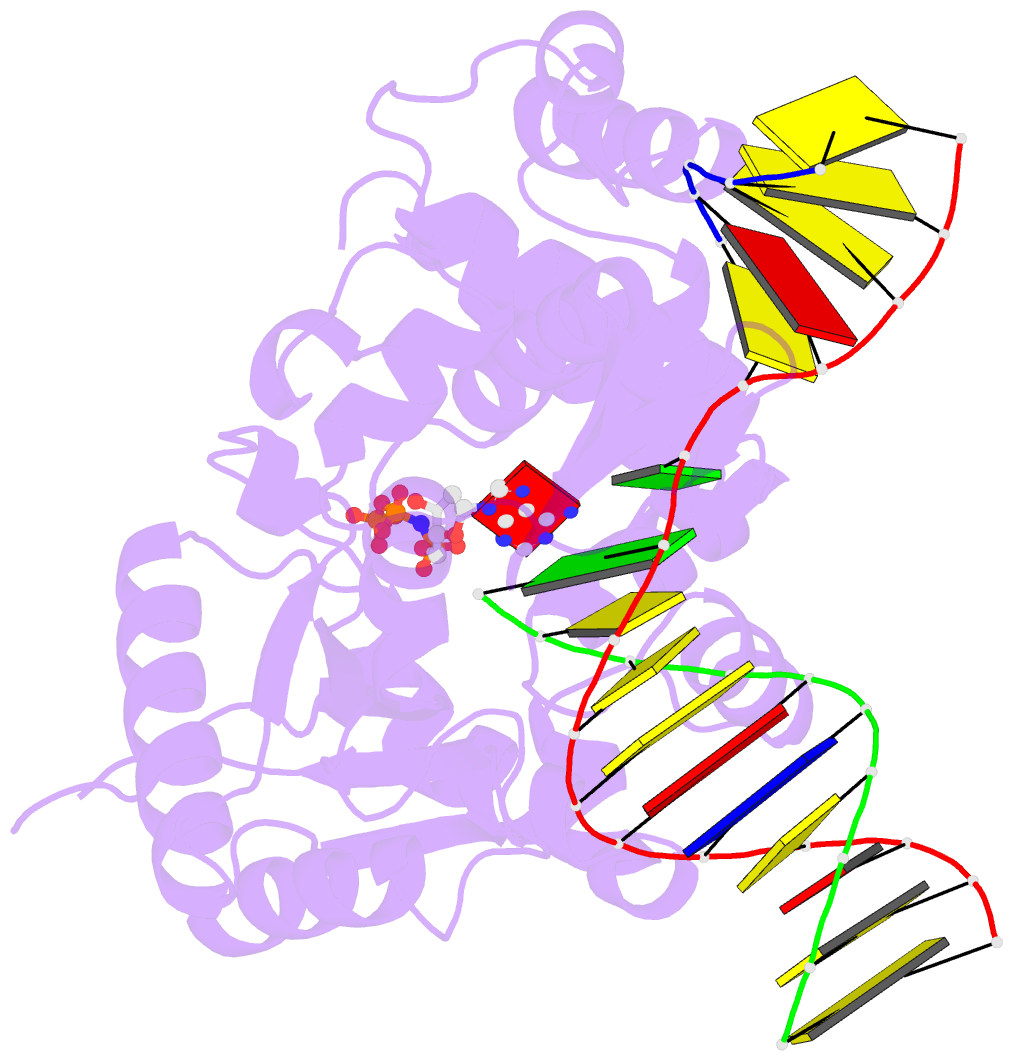
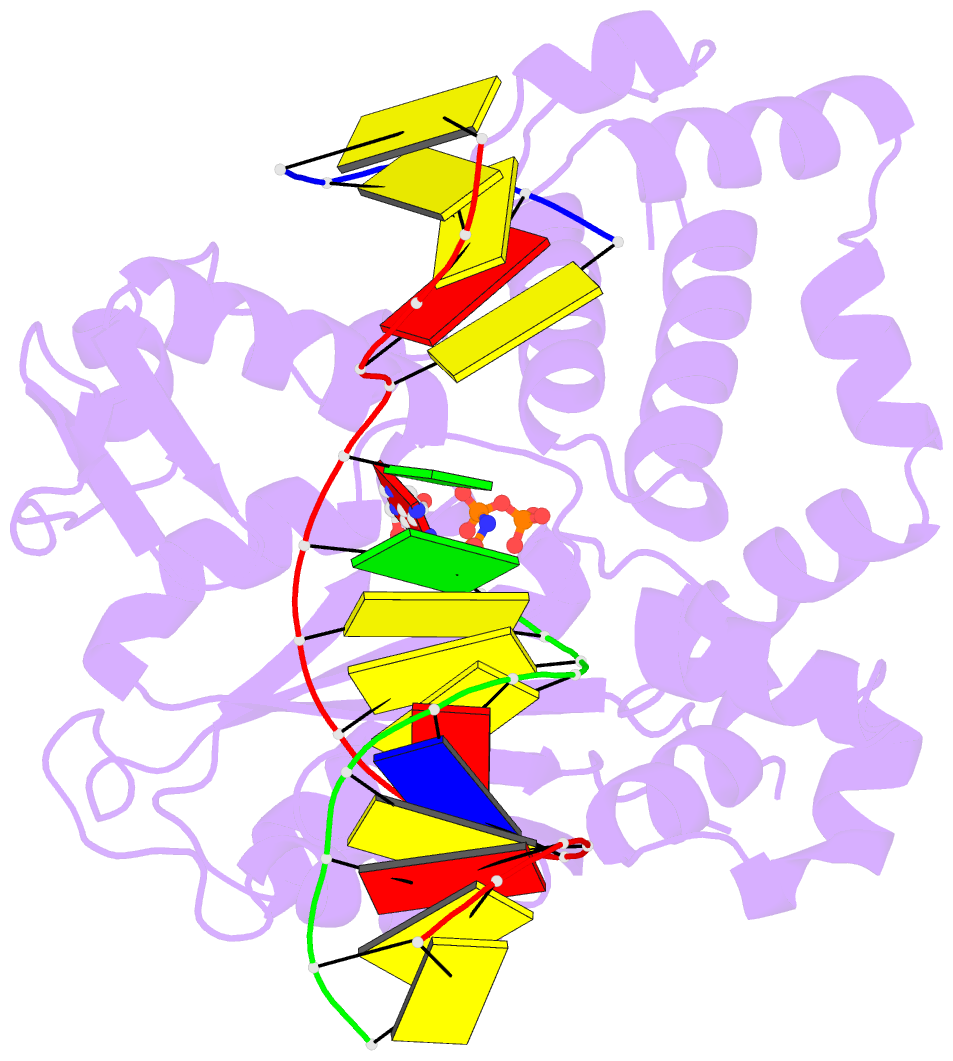
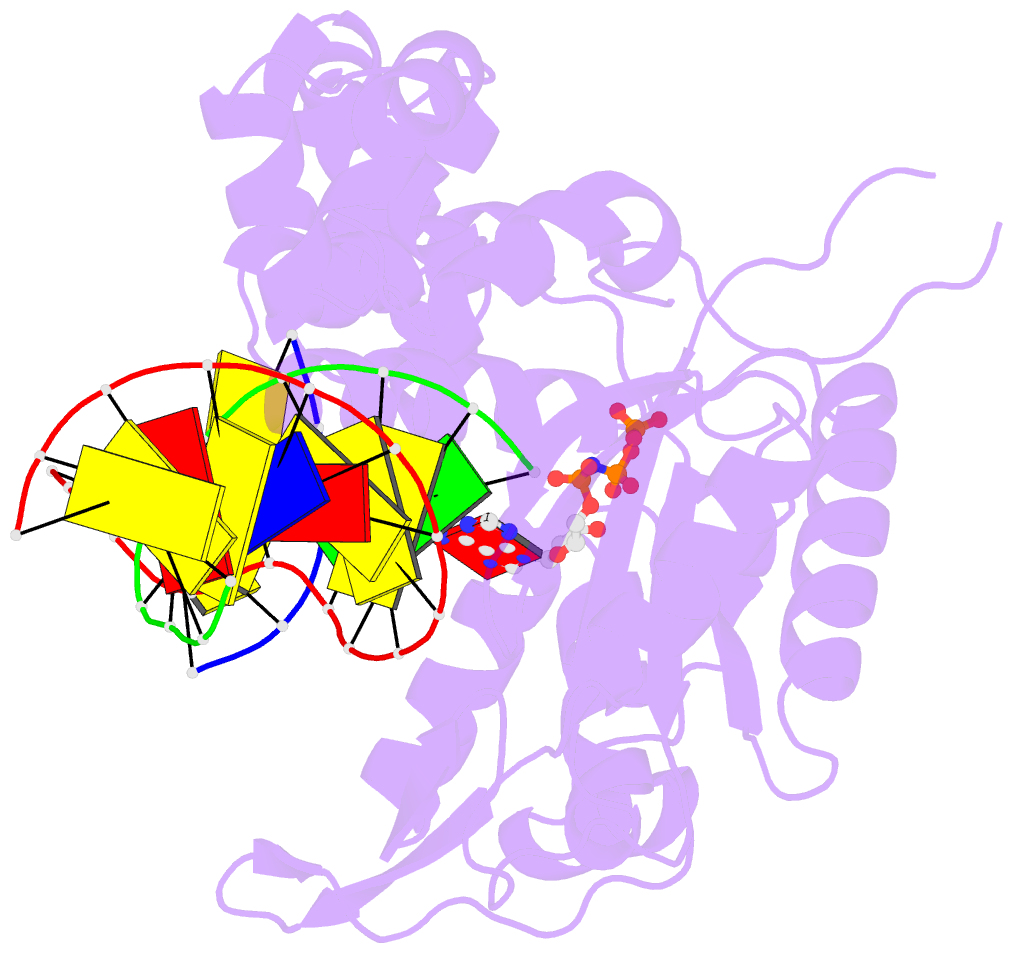
Summary information and primary citation
- PDB-id
- 6u6b; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- transferase-DNA
- Method
- X-ray (3.108 Å)
- Summary
- Structure of human DNA polymerase beta misinserting dampnpp opposite the 5'g of the cisplatin pt-gg intrastrand crosslink with manganese in the active site
- Reference
- Ouzon-Shubeita H, Vilas CK, Lee S (2020): "Structural insights into the promutagenic bypass of the major cisplatin-induced DNA lesion." Biochem.J., 477, 937-951. doi: 10.1042/BCJ20190906.
- Abstract
- The cisplatin-1,2-d(GpG) (Pt-GG) intrastrand cross-link is the predominant DNA lesion generated by cisplatin. Cisplatin has been shown to predominantly induce G to T mutations and Pt-GG permits significant misincorporation of dATP by human DNA polymerase β (polβ). In agreement, polβ overexpression, which is frequently observed in cancer cells, is linked to cisplatin resistance and a mutator phenotype. However, the structural basis for the misincorporation of dATP opposite Pt-GG is unknown. Here, we report the first structures of a DNA polymerase inaccurately bypassing Pt-GG. We solved two structures of polβ misincorporating dATP opposite the 5'-dG of Pt-GG in the presence of Mg2+ or Mn2+. The Mg2+-bound structure exhibits a sub-optimal conformation for catalysis, while the Mn2+-bound structure is in a catalytically more favorable semi-closed conformation. In both structures, dATP does not form a coplanar base pairing with Pt-GG. In the polβ active site, the syn-dATP opposite Pt-GG appears to be stabilized by protein templating and pi stacking interactions, which resembles the polβ-mediated dATP incorporation opposite an abasic site. Overall, our results suggest that the templating Pt-GG in the polβ active site behaves like an abasic site, promoting the insertion of dATP in a non-instructional manner.