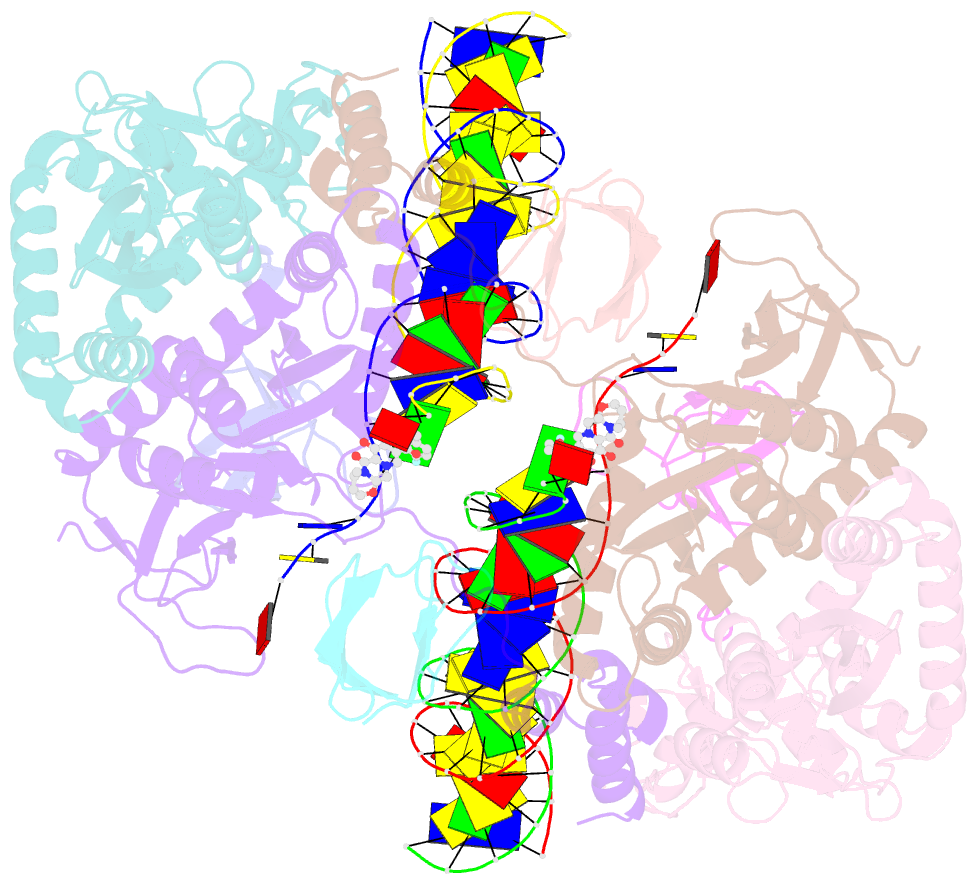
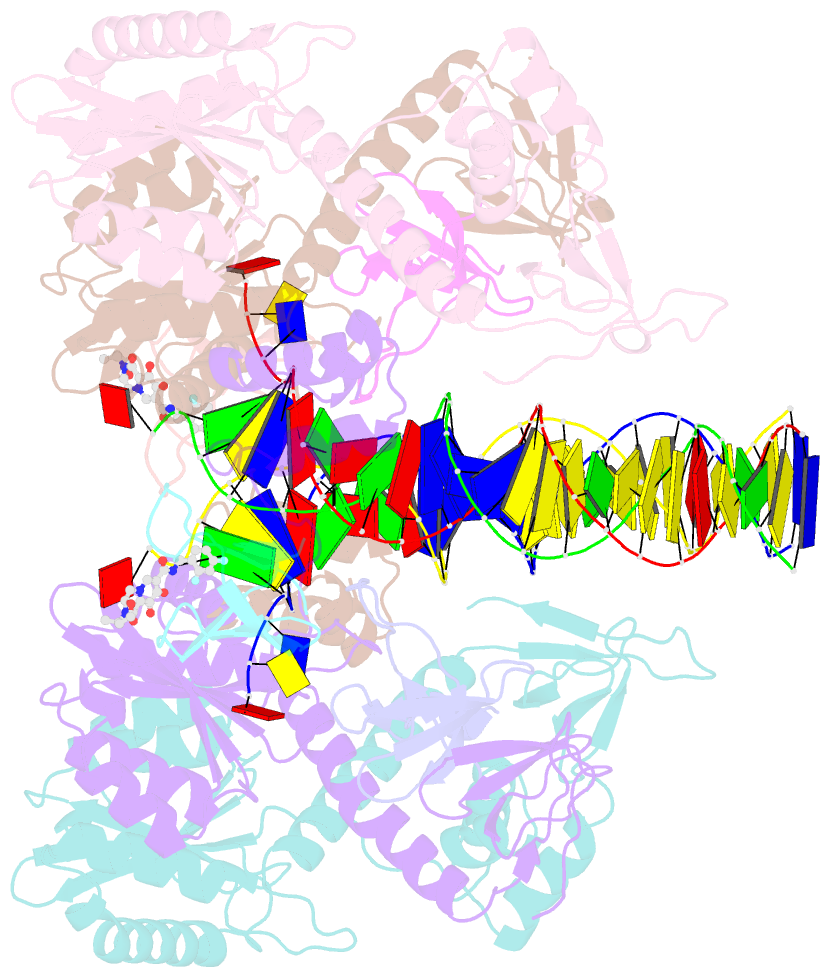
Summary information and primary citation
- PDB-id
-
6vdk;
DSSR-derived features in text and
JSON formats; DNAproDB
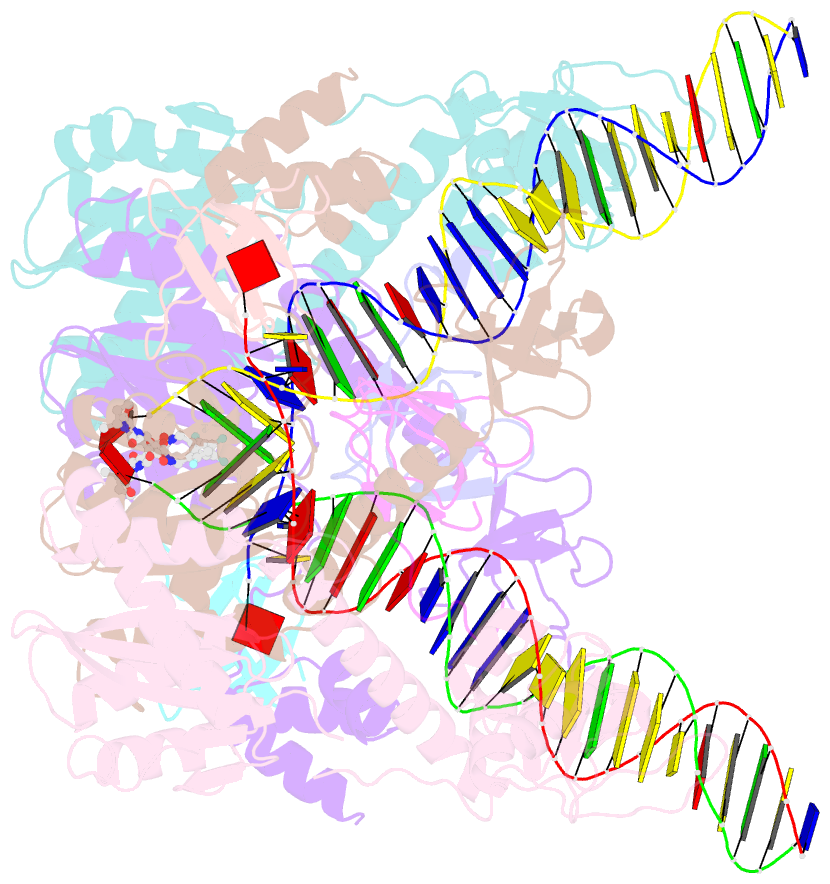
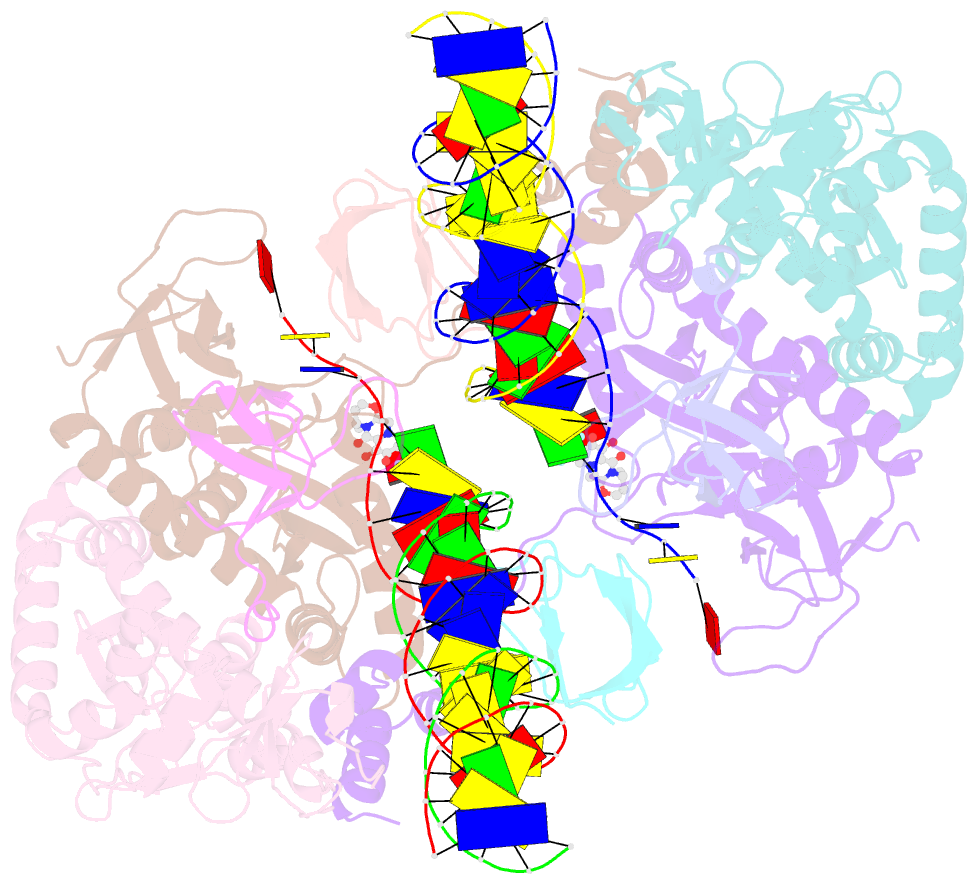
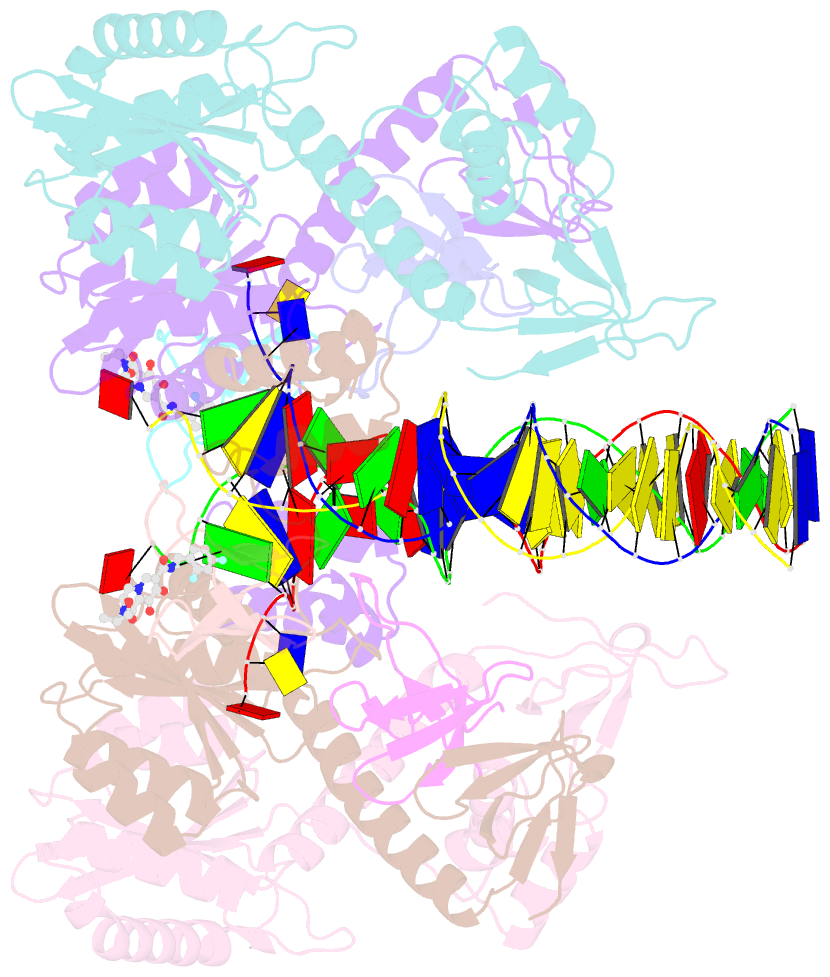
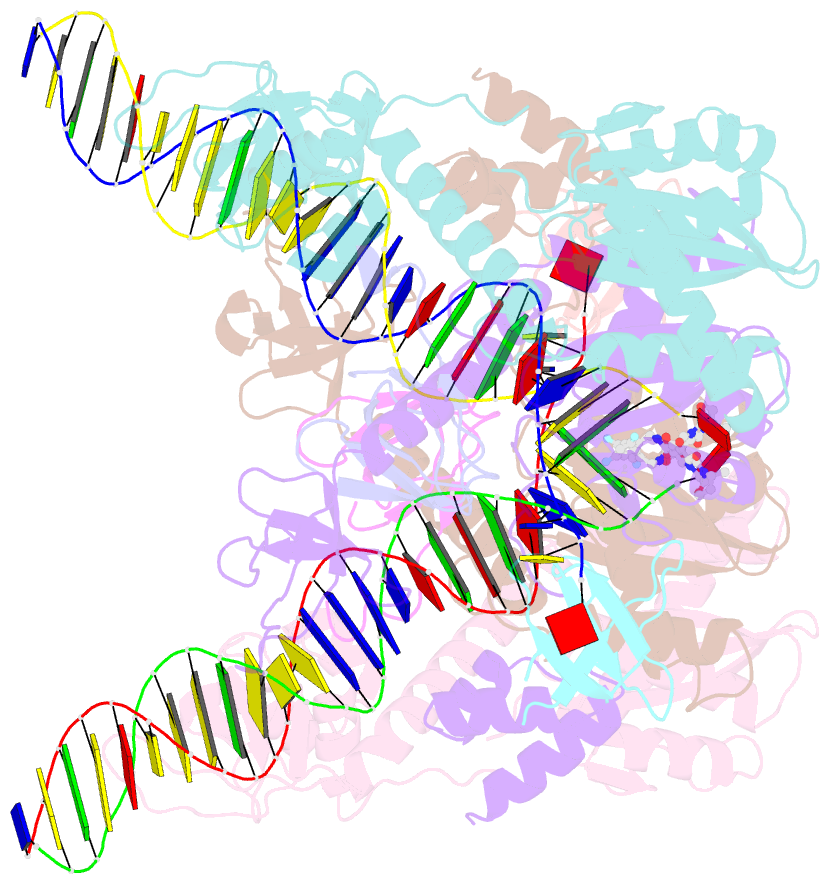
- Class
- transferase-DNA
- Method
- cryo-EM (4.5 Å)
- Summary
- Cryoem structure of hiv-1 conserved intasome core
- Reference
-
Li M, Chen X, Wang H, Jurado KA, Engelman AN, Craigie R
(2020): "A Peptide
Derived from Lens Epithelium-Derived Growth Factor
Stimulates HIV-1 DNA Integration and Facilitates Intasome
Structural Studies." J.Mol.Biol.,
432, 2055-2066. doi: 10.1016/j.jmb.2020.01.040.
- Abstract
- The low solubility and aggregation properties of HIV-1
integrase (IN) are major obstacles for biochemical and
structural studies. The lens epithelium-derived growth
factor (LEDGF) is a cellular factor that binds IN and
tethers preintegration complexes to chromatin before
integration. The LEDGF also stimulates HIV-1 IN DNA strand
transfer activity and improves its solubility in vitro. We
show that these properties are conferred by a short peptide
spanning residues 178 to 197 of the LEDGF that encompasses
its AT-hook DNA-binding elements. The peptide stimulates
HIV-1 IN activity both in trans and in cis. Fusion of the
peptide to either the N- or C-terminus of IN results in
maximal stimulation of concerted integration activity and
greatly improves the solubility of the protein and
nucleoprotein complexes of IN with viral DNA ends
(intasomes). High-resolution structures of HIV-1 intasomes
are required to understand the mechanism of IN strand
transfer inhibitors (INSTIs), which are front-line drugs
for the treatment of HIV-1, and how the virus can develop
resistance to INSTIs. We have previously determined the
structure of the HIV-1 strand transfer complex intasome.
The improved biophysical properties of intasomes assembled
with LEDGF peptide fusion IN have enabled us to determine
the structure of the cleaved synaptic
complex intasome, which is the direct target of
INSTIs.