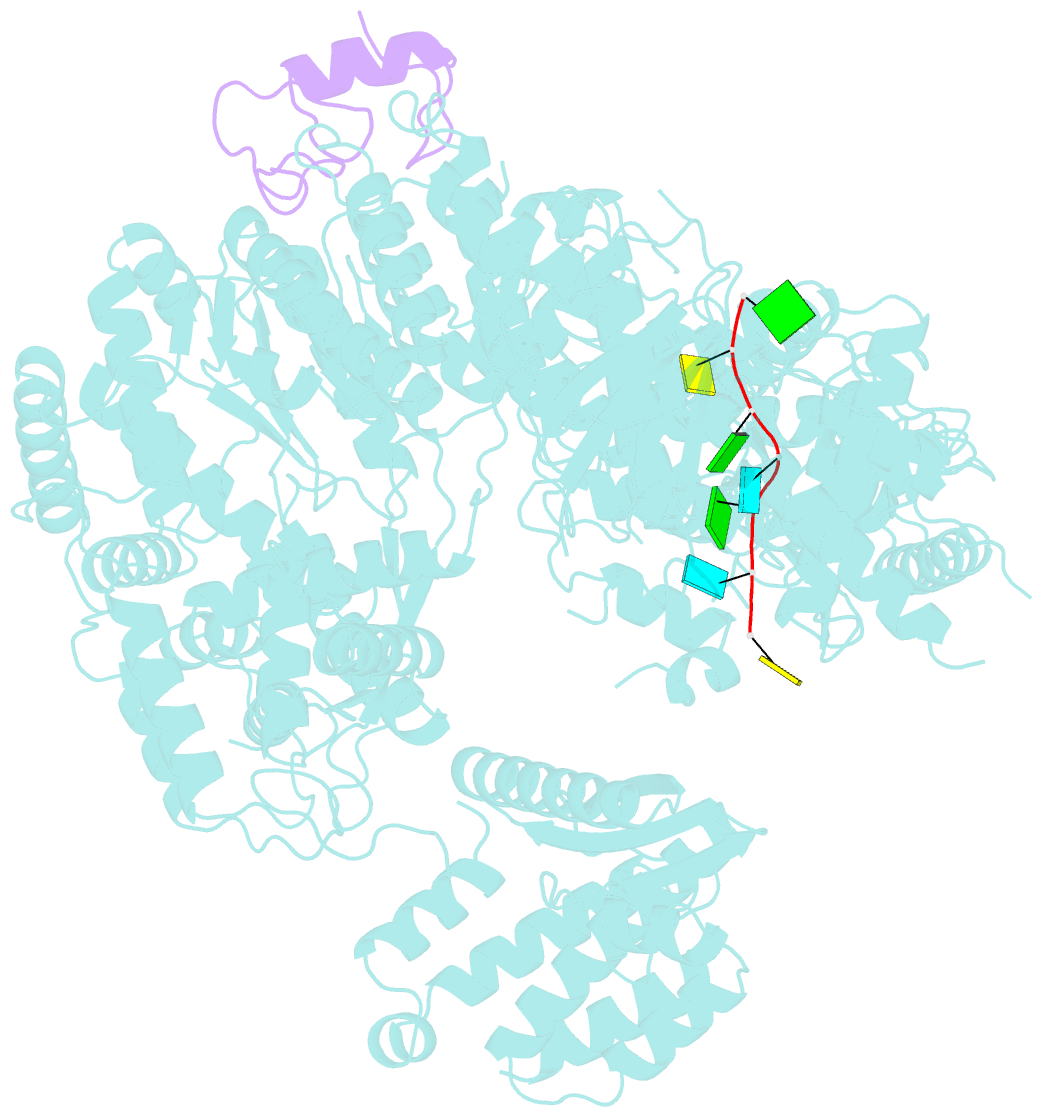
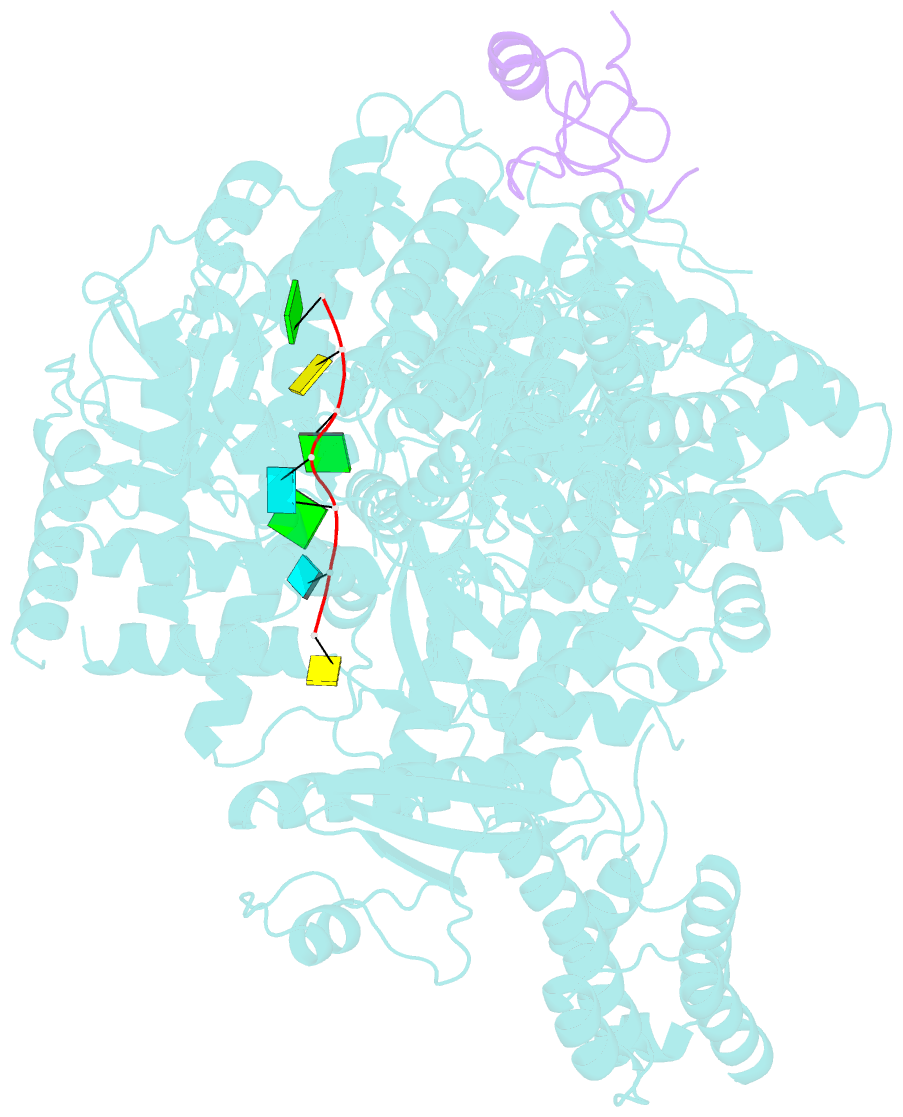
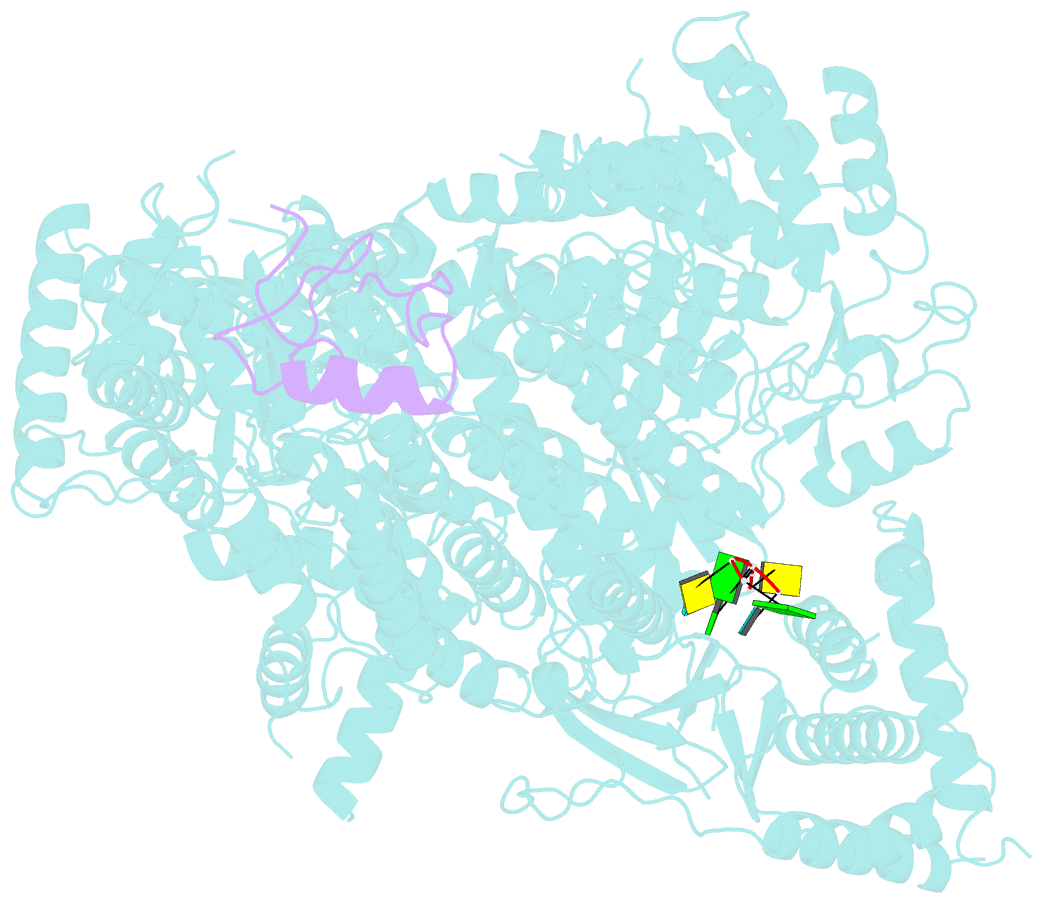
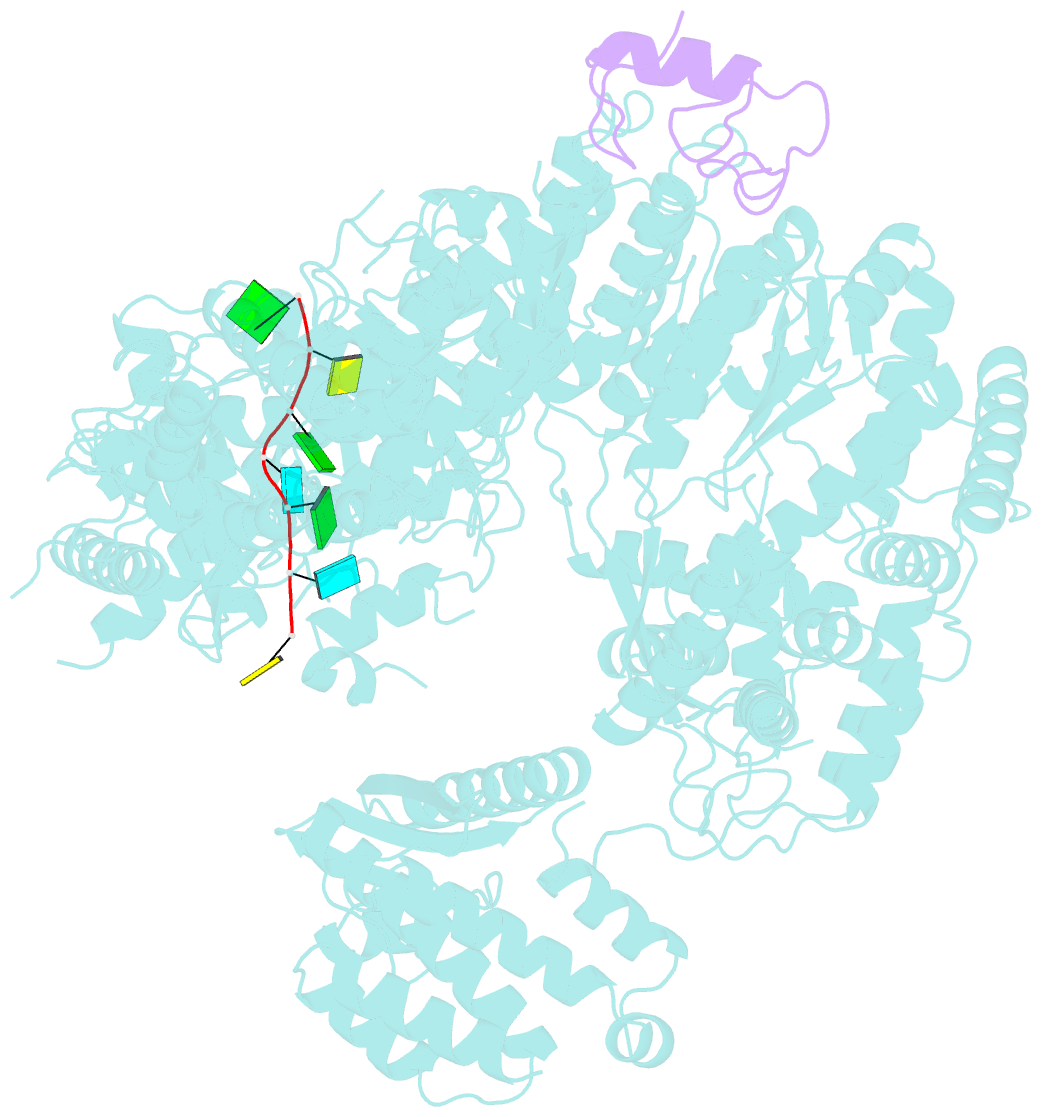
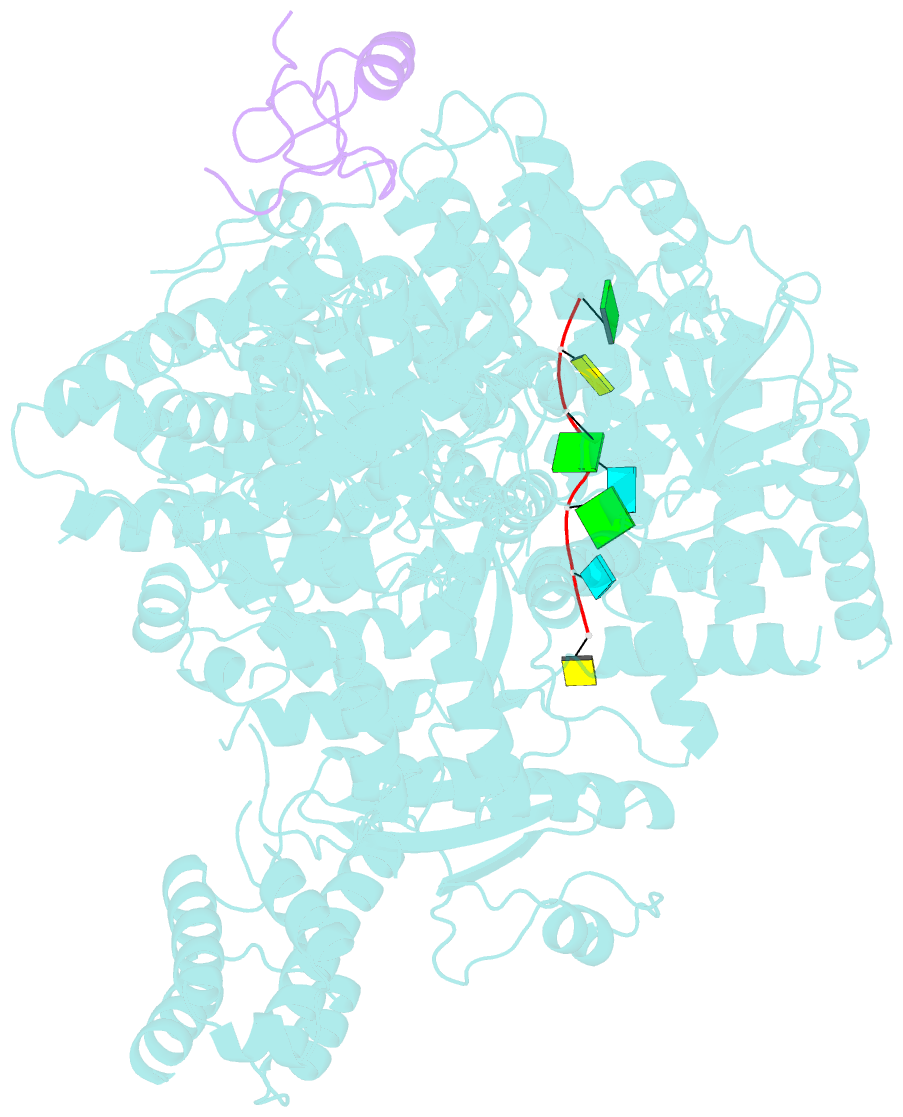
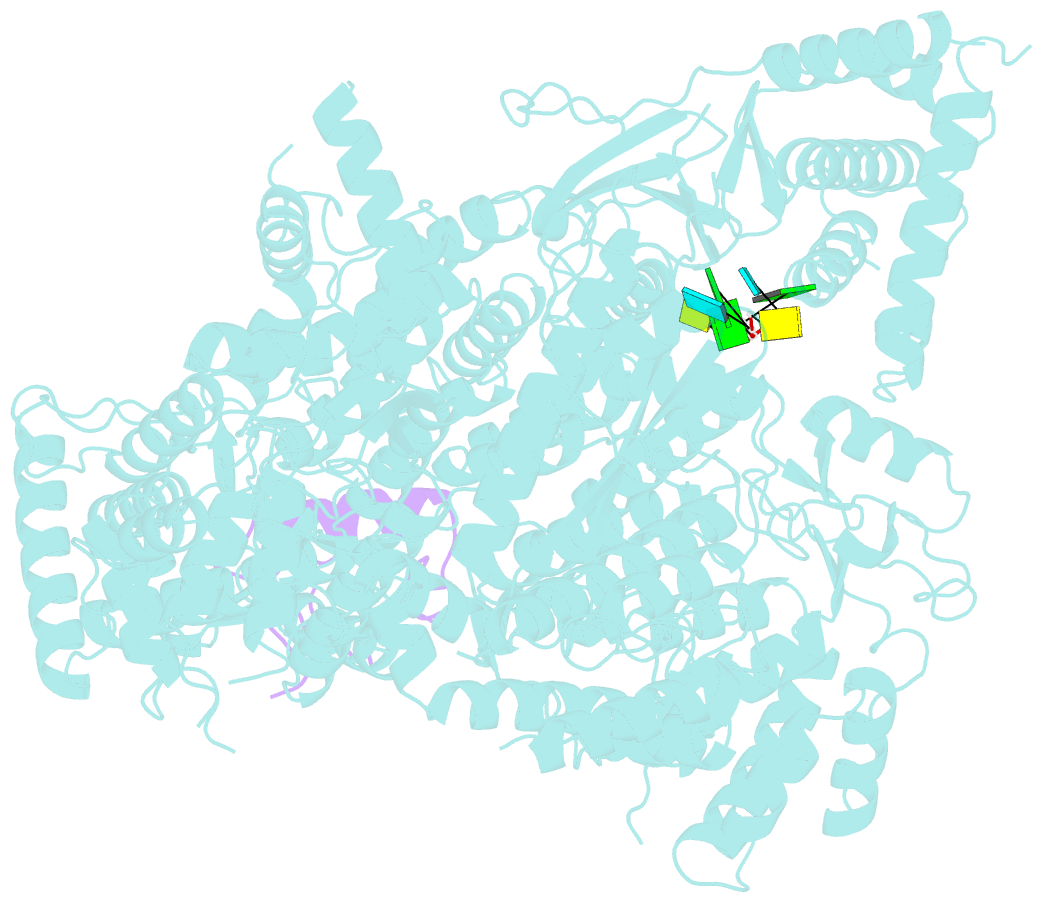
Summary information and primary citation
- PDB-id
- 7ela; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- viral protein-RNA
- Method
- cryo-EM (3.4 Å)
- Summary
- Structure of lassa virus polymerase in complex with 3'-vrna and z mutant (f36a)
- Reference
- Xu X, Peng R, Peng Q, Wang M, Xu Y, Liu S, Tian X, Deng H, Tong Y, Hu X, Zhong J, Wang P, Qi J, Gao GF, Shi Y (2021): "Cryo-EM structures of Lassa and Machupo virus polymerases complexed with cognate regulatory Z proteins identify targets for antivirals." Nat Microbiol, 6, 921-931. doi: 10.1038/s41564-021-00916-w.
- Abstract
- Zoonotic arenaviruses can lead to life-threating diseases in humans. These viruses encode a large (L) polymerase that transcribes and replicates the viral genome. At the late stage of replication, the multifunctional Z protein interacts with the L polymerase to shut down RNA synthesis and initiate virion assembly. However, the mechanism by which the Z protein regulates the activity of L polymerase is unclear. Here, we used cryo-electron microscopy to resolve the structures of both Lassa and Machupo virus L polymerases in complex with their cognate Z proteins, and viral RNA, to 3.1-3.9 Å resolutions. These structures reveal that Z protein binding induces conformational changes in two catalytic motifs of the L polymerase, and restrains their conformational dynamics to inhibit RNA synthesis, which is supported by hydrogen-deuterium exchange mass spectrometry analysis. Importantly, we show, by in vitro polymerase reactions, that Z proteins of Lassa and Machupo viruses can cross-inhibit their L polymerases, albeit with decreased inhibition efficiencies. This cross-reactivity results from a highly conserved determinant motif at the contacting interface, but is affected by other variable auxiliary motifs due to the divergent evolution of Old World and New World arenaviruses. These findings could provide promising targets for developing broad-spectrum antiviral drugs.