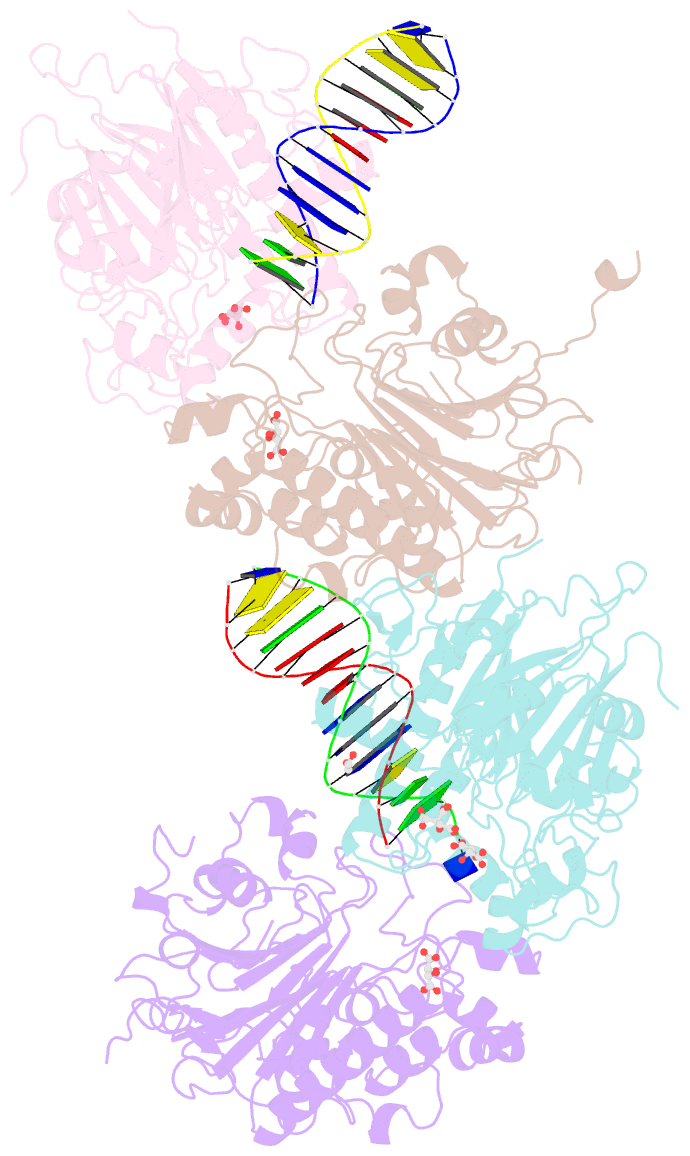
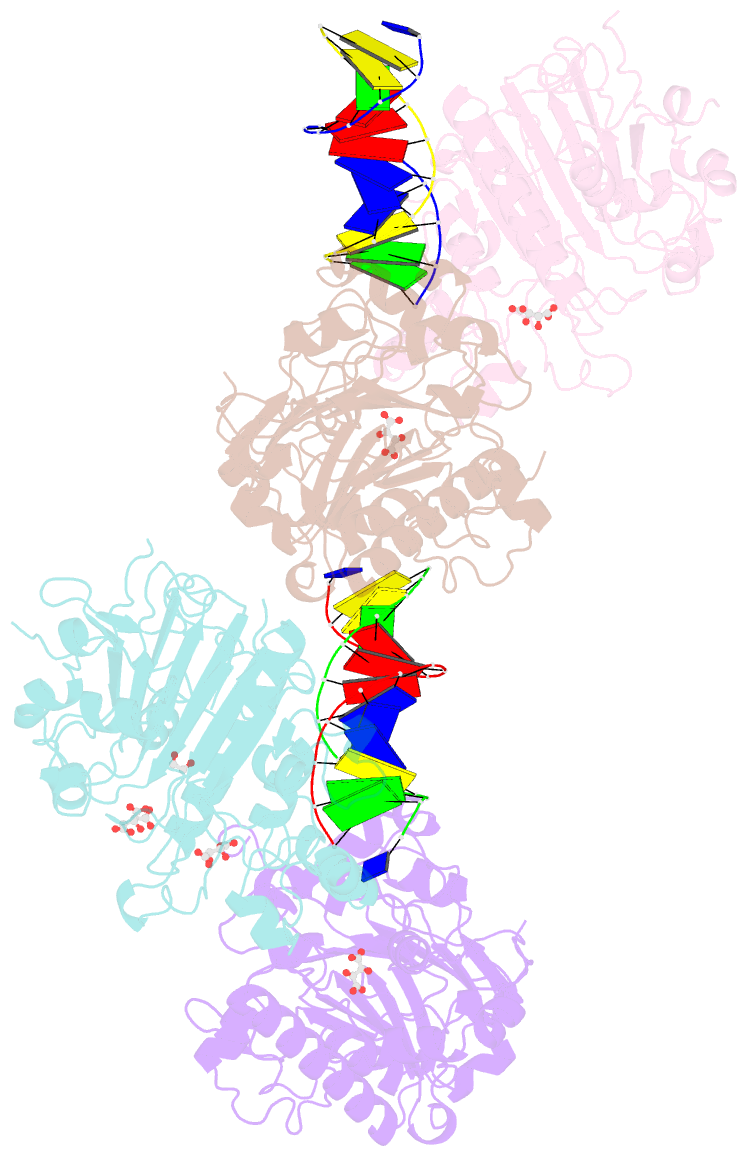
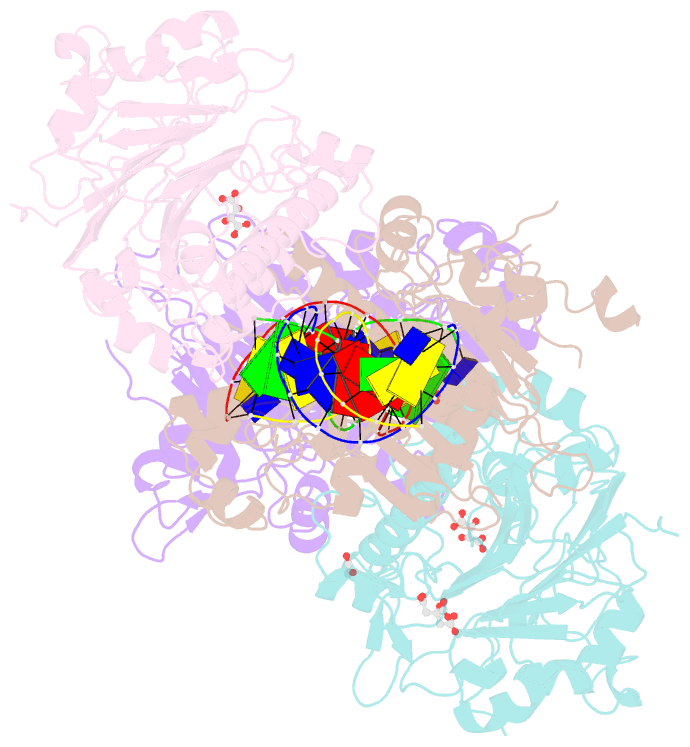
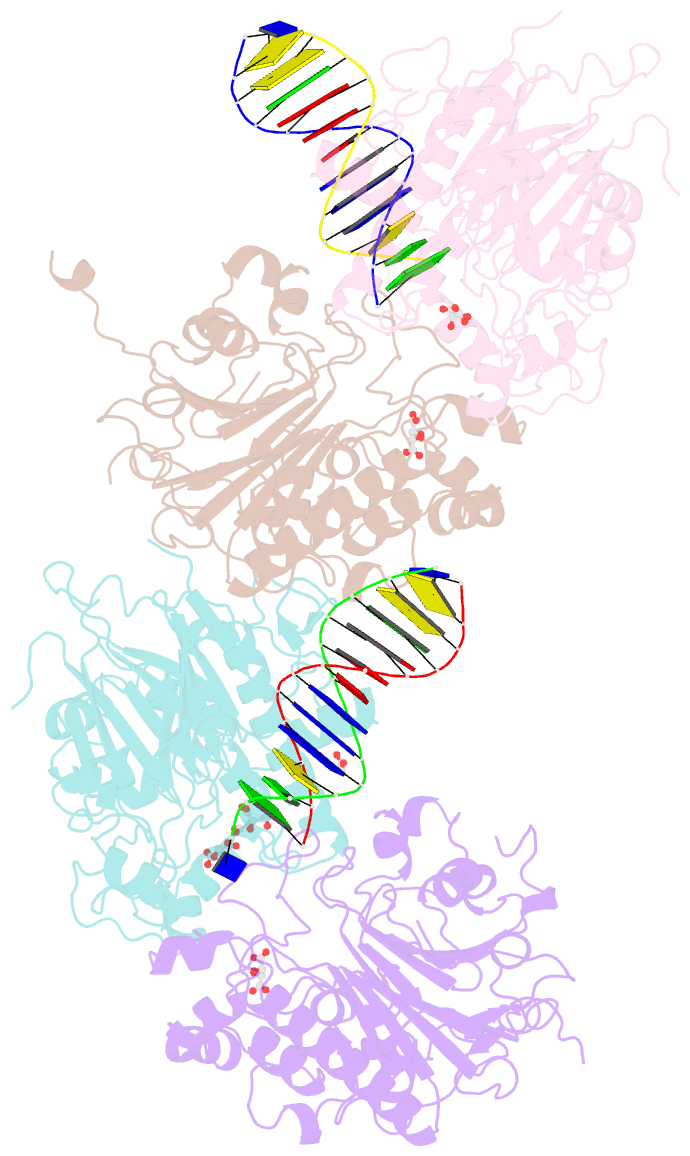
Summary information and primary citation
- PDB-id
- 7n3y; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- hydrolase-DNA
- Method
- X-ray (2.73 Å)
- Summary
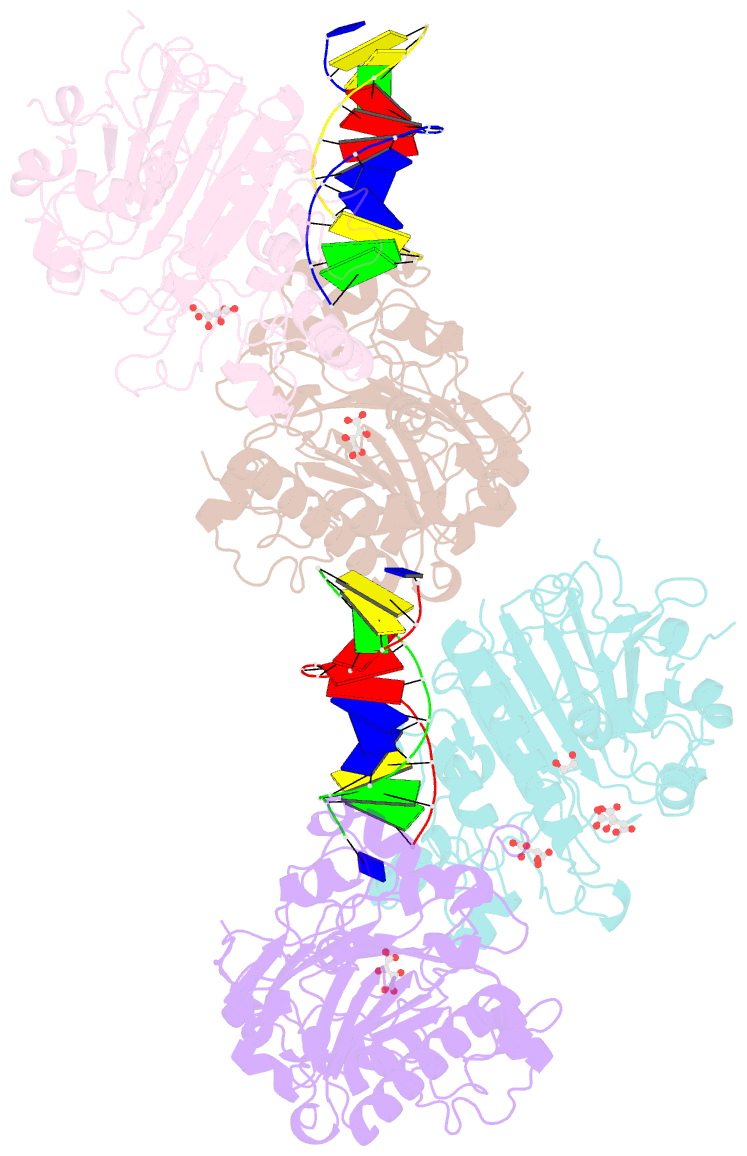
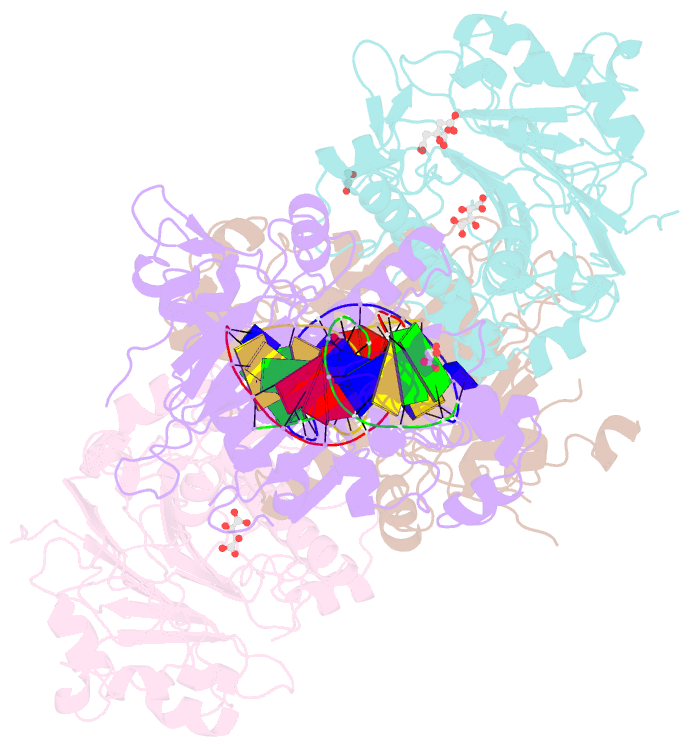
- Crystal structure of saccharomyces cerevisiae apn2 catalytic domain e59q-d222n mutant in complex with DNA
- Reference
- Williams JS, Wojtaszek JL, Appel DC, Krahn J, Wallace BD, Walsh E, Kunkel TA, Williams RS (2022): "Molecular basis for processing of topoisomerase 1-triggered DNA damage by Apn2/APE2." Cell Rep, 41, 111448. doi: 10.1016/j.celrep.2022.111448.
- Abstract
- Topoisomerase 1 (Top1) incises DNA containing ribonucleotides to generate complex DNA lesions that are resolved by APE2 (Apn2 in yeast). How Apn2 engages and processes this DNA damage is unclear. Here, we report X-ray crystal structures and biochemical analysis of Apn2-DNA complexes to demonstrate how Apn2 frays and cleaves 3' DNA termini via a wedging mechanism that facilitates 1-6 nucleotide endonucleolytic cleavages. APN2 deletion and DNA-wedge mutant Saccharomyces cerevisiae strains display mutator phenotypes, cell growth defects, and sensitivity to genotoxic stress in a ribonucleotide excision repair (RER)-defective background harboring a high density of Top1-incised ribonucleotides. Our data implicate a wedge-and-cut mechanism underpinning the broad-specificity Apn2 nuclease activity that mitigates mutagenic and genome instability phenotypes caused by Top1 incision at genomic ribonucleotides incorporated by DNA polymerase epsilon.