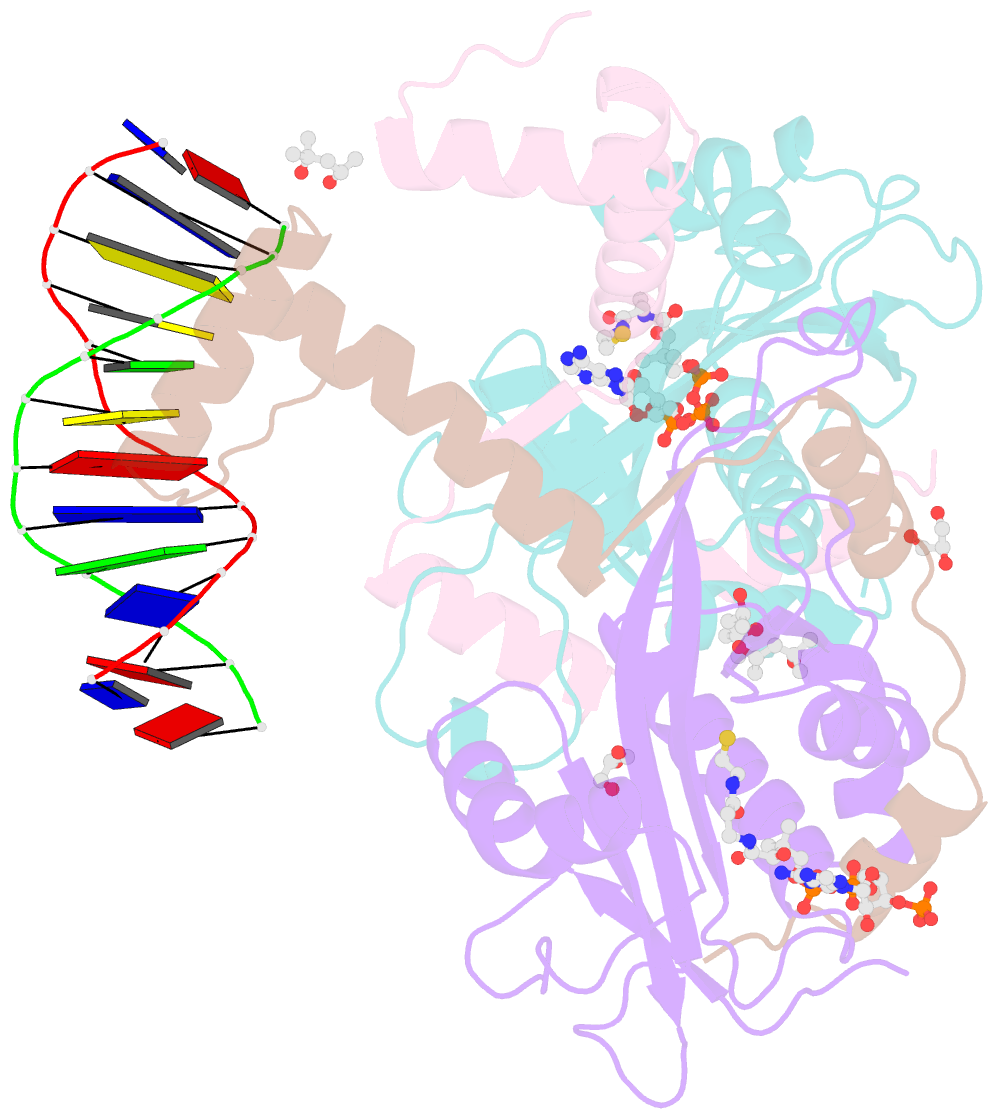
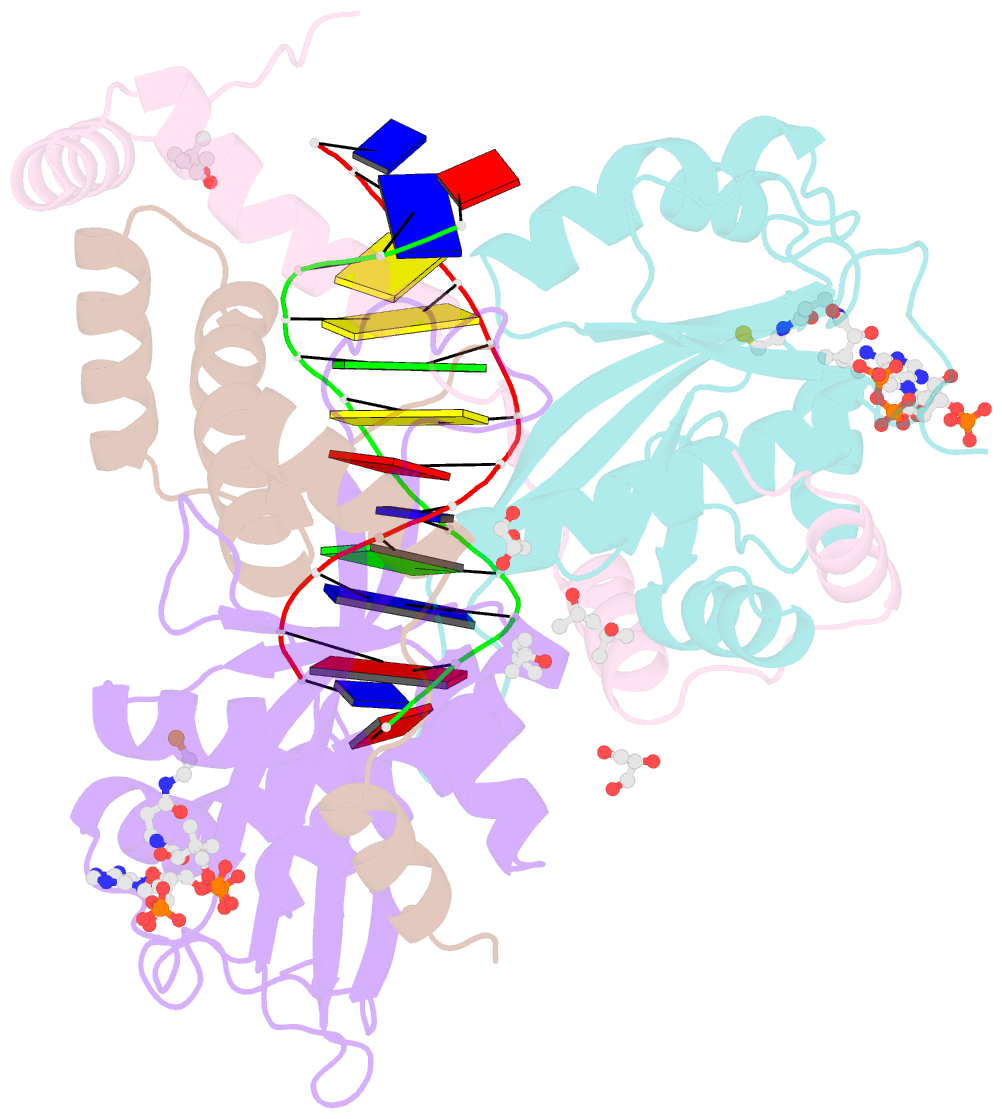
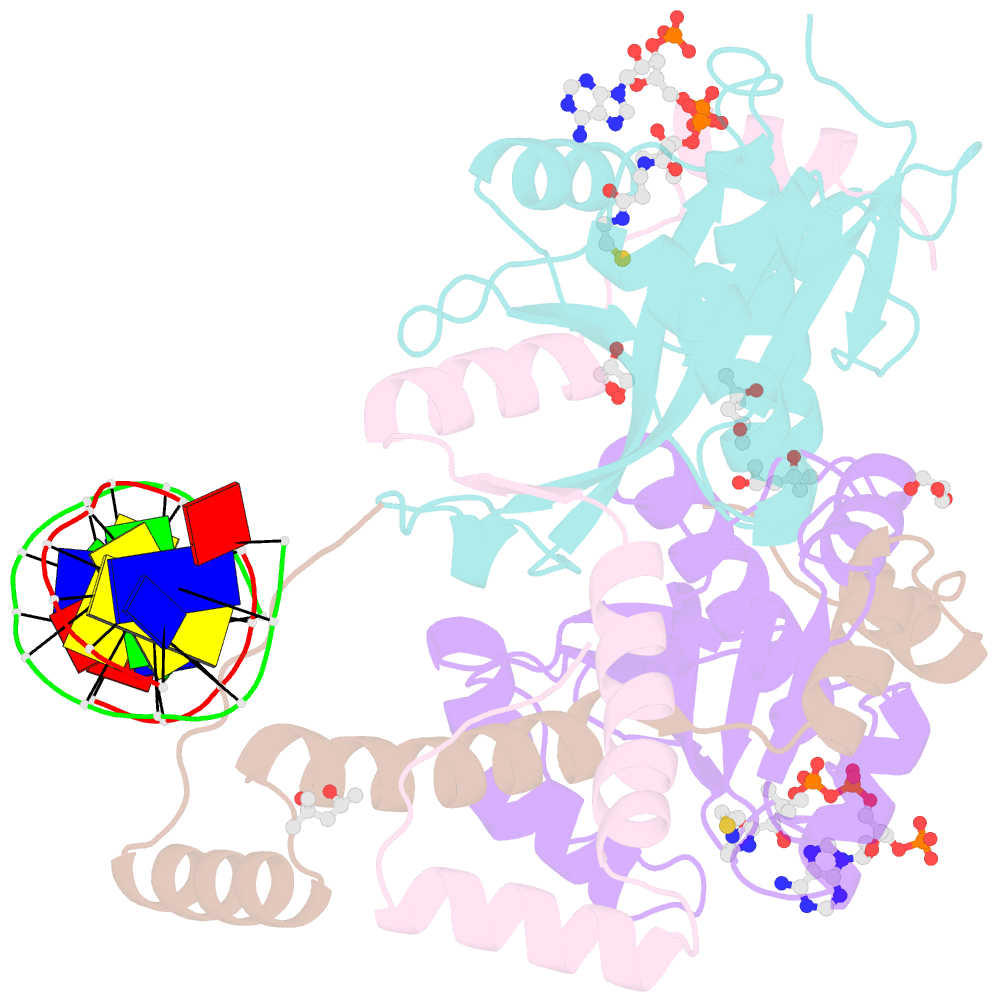
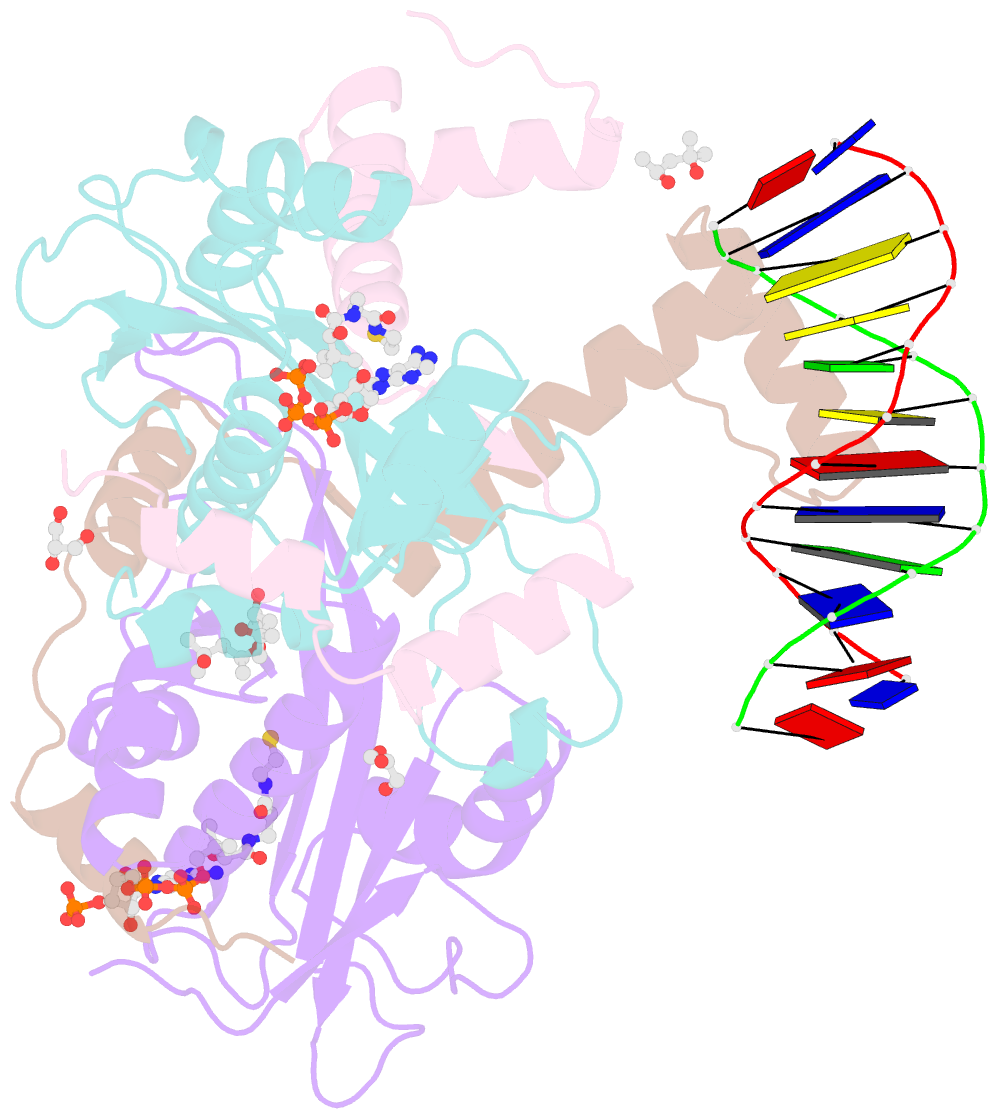
Summary information and primary citation
- PDB-id
- 7zg5; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- toxin
- Method
- X-ray (2.0 Å)
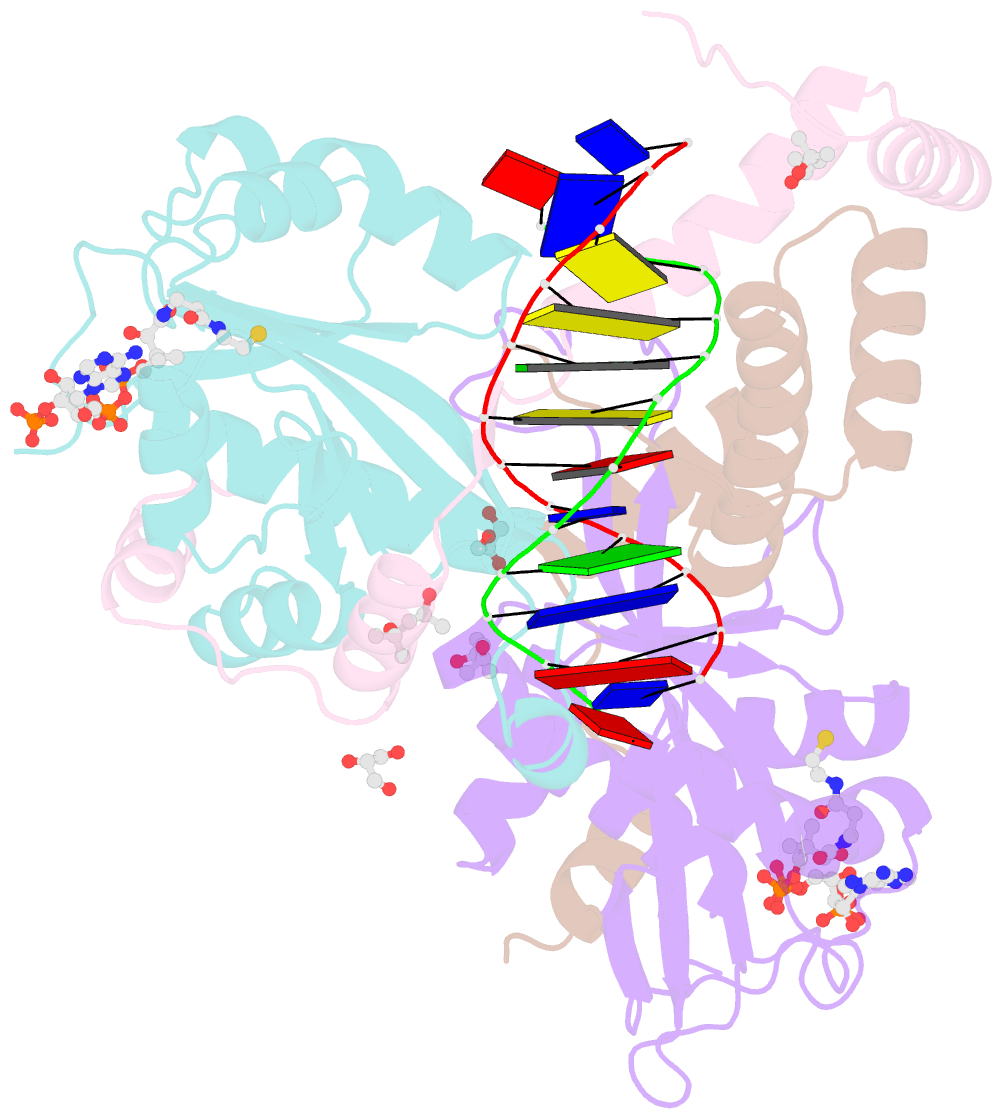
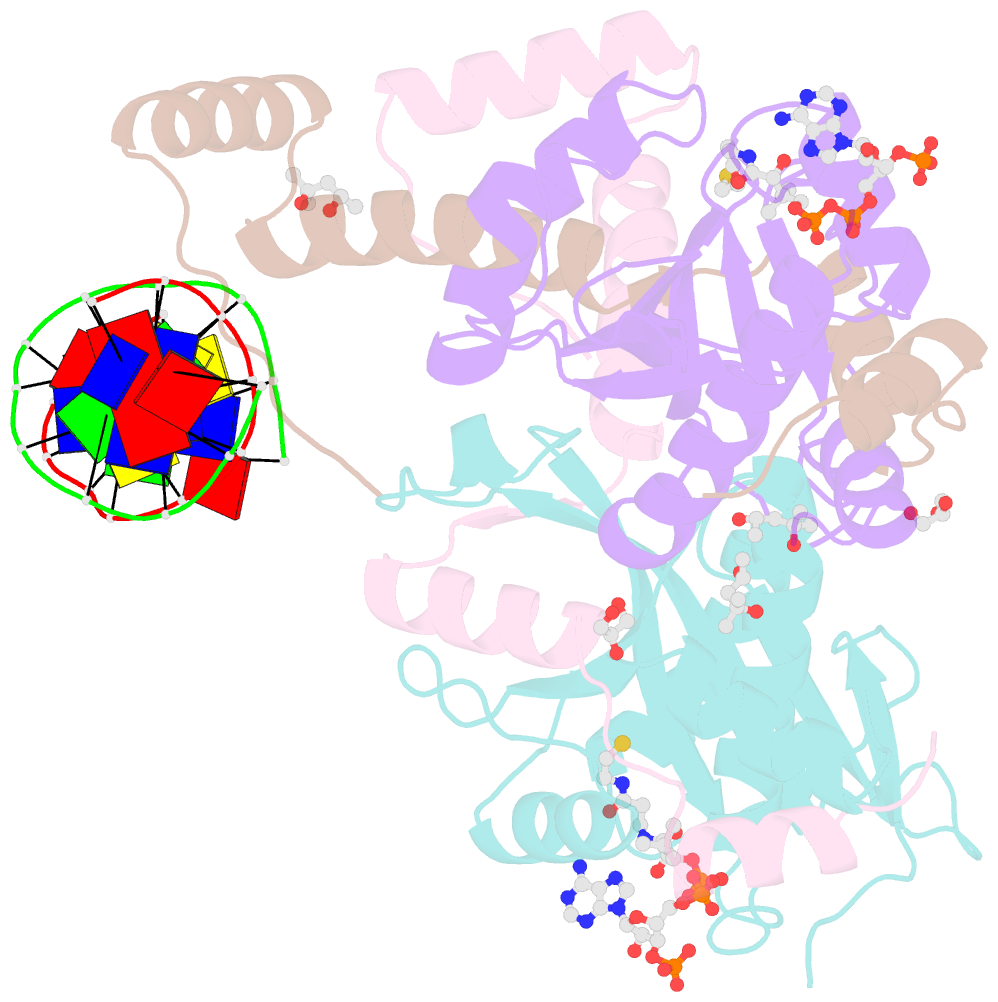
- Summary
- The crystal structure of salmonella tacat3-DNA complex
- Reference
- Grabe GJ, Giorgio RT, Wieczor M, Gollan B, Sargen M, Orozco M, Hare SA, Helaine S (2024): "Molecular stripping underpins derepression of a toxin-antitoxin system." Nat.Struct.Mol.Biol. doi: 10.1038/s41594-024-01253-2.
- Abstract
- Transcription factors control gene expression; among these, transcriptional repressors must liberate the promoter for derepression to occur. Toxin-antitoxin (TA) modules are bacterial elements that autoregulate their transcription by binding the promoter in a T:A ratio-dependent manner, known as conditional cooperativity. The molecular basis of how excess toxin triggers derepression has remained elusive, largely because monitoring the rearrangement of promoter-repressor complexes, which underpin derepression, is challenging. Here, we dissect the autoregulation of the Salmonella enterica tacAT3 module. Using a combination of assays targeting DNA binding and promoter activity, as well as structural characterization, we determine the essential TA and DNA elements required to control transcription, and we reconstitute a repression-to-derepression path. We demonstrate that excess toxin triggers molecular stripping of the repressor complex off the DNA through multiple allosteric changes causing DNA distortion and ultimately leading to derepression. Thus, our work provides important insight into the mechanisms underlying conditional cooperativity.