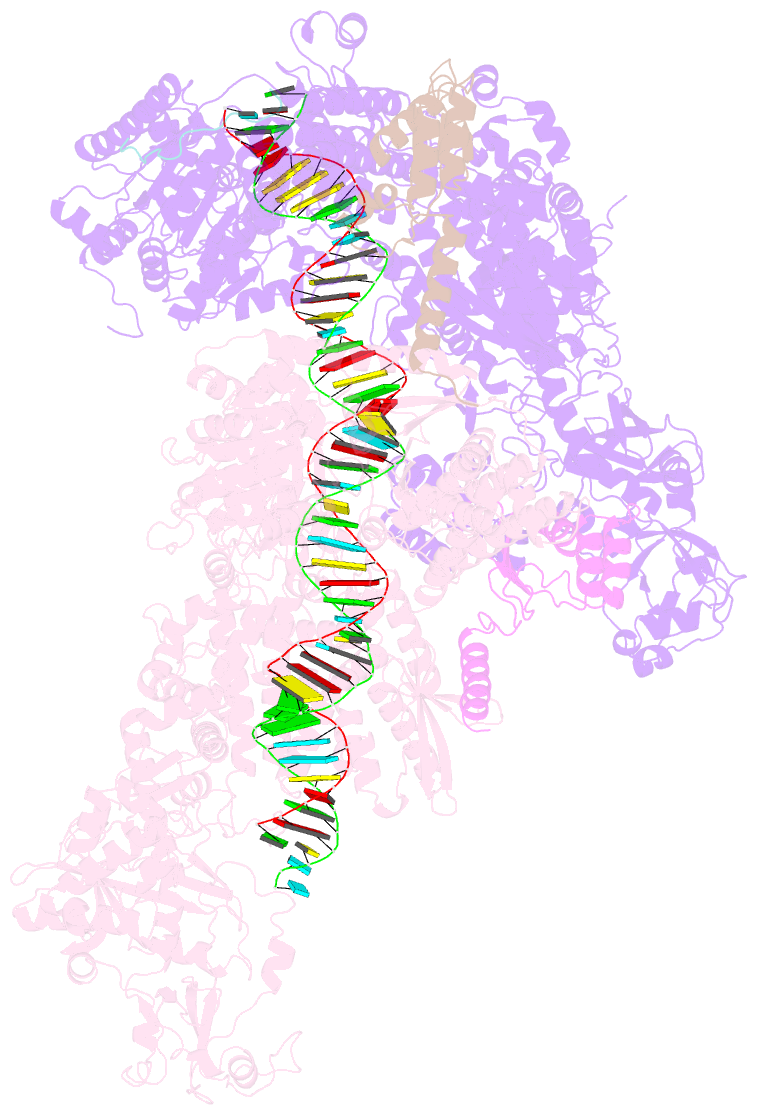
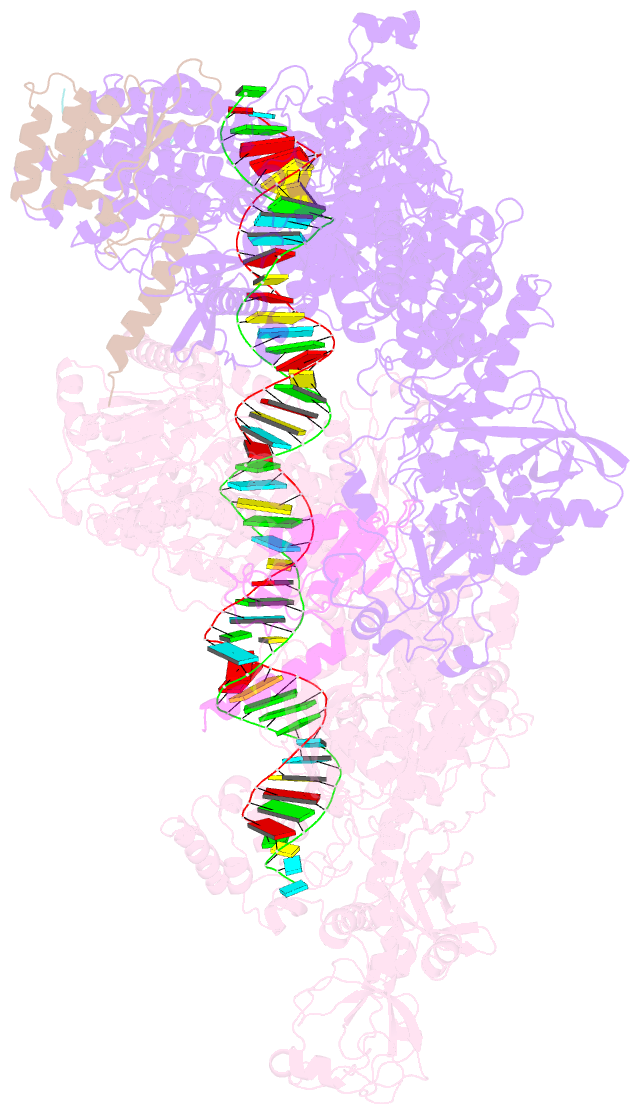
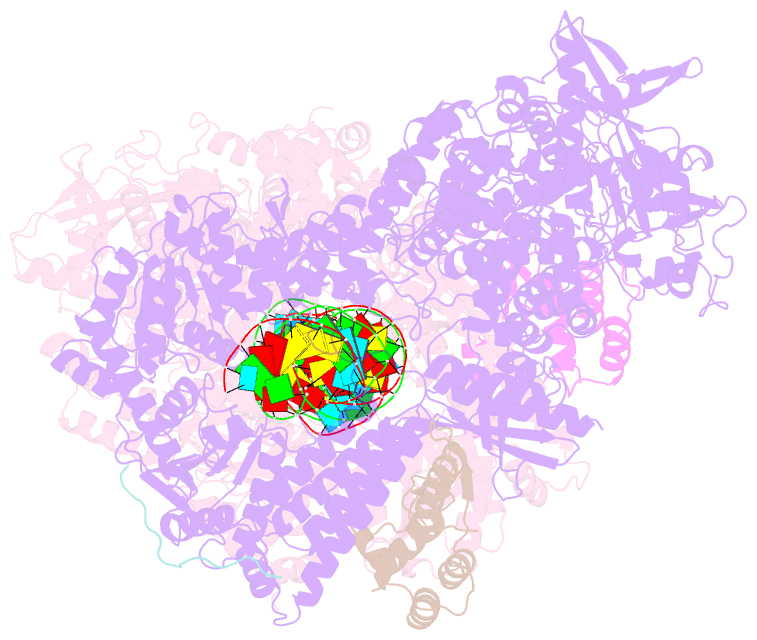
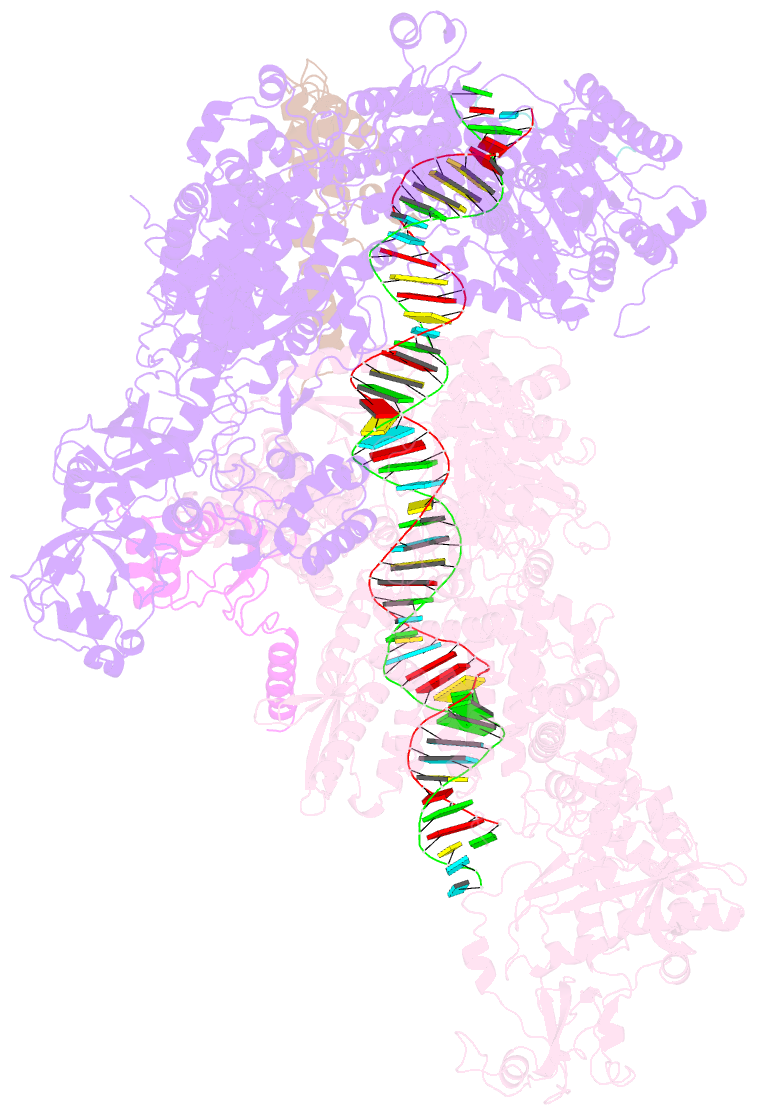
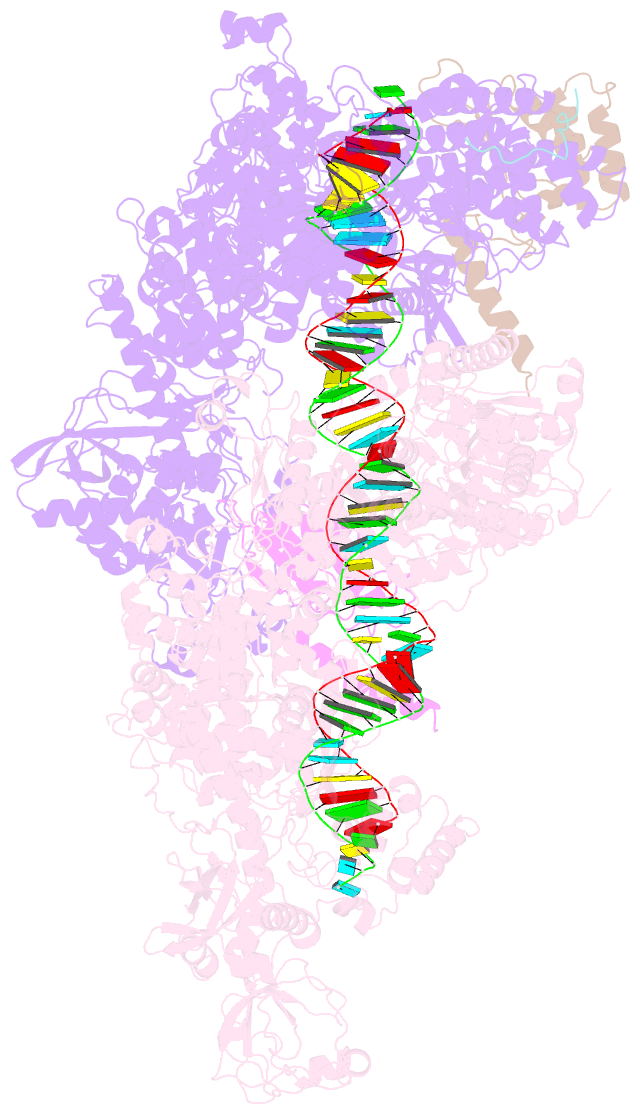
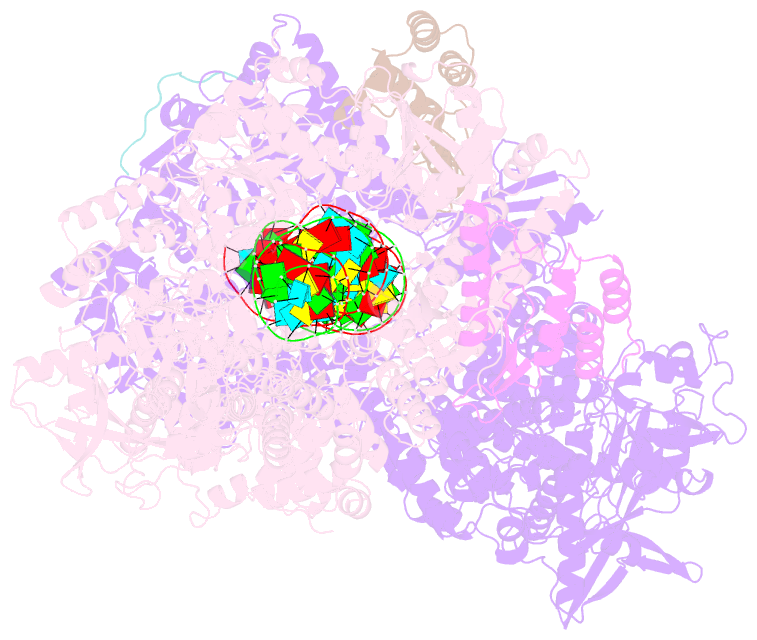
Summary information and primary citation
- PDB-id
- 8hf0; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- RNA binding protein-RNA
- Method
- cryo-EM (3.72 Å)
- Summary
- Dmdcr-2-r2d2-loqspd with 50bp-dsrna in dimer state
- Reference
- Deng T, Su S, Yuan X, He J, Huang Y, Ma J, Wang J (2023): "Structural mechanism of R2D2 and Loqs-PD synergistic modulation on DmDcr-2 oligomers." Nat Commun, 14, 5228. doi: 10.1038/s41467-023-40919-1.
- Abstract
- Small interference RNAs are the key components of RNA interference, a conserved RNA silencing or viral defense mechanism in many eukaryotes. In Drosophila melanogaster, Dicer-2 (DmDcr-2)-mediated RNAi pathway plays important roles in defending against viral infections and protecting genome integrity. During the maturation of siRNAs, two cofactors can regulate DmDcr-2's functions: Loqs-PD that is required for dsRNA processing, and R2D2 that is essential for the subsequent loading of siRNAs into effector Ago2 to form RISC complexes. However, due to the lack of structural information, it is still unclear whether R2D2 and Loqs-PD affect the functions of DmDcr-2 simultaneously. Here we present several cryo-EM structures of DmDcr-2/R2D2/Loqs-PD complex bound to dsRNAs with various lengths by the Helicase domain. These structures revealed that R2D2 and Loqs-PD can bind to different regions of DmDcr-2 without interfering with each other. Furthermore, the cryo-EM results demonstrate that these complexes can form large oligomers and assemble into fibers. The formation and depolymerization of these oligomers are associated with ATP hydrolysis. These findings provide insights into the structural mechanism of DmDcr-2 and its cofactors during siRNA processing.