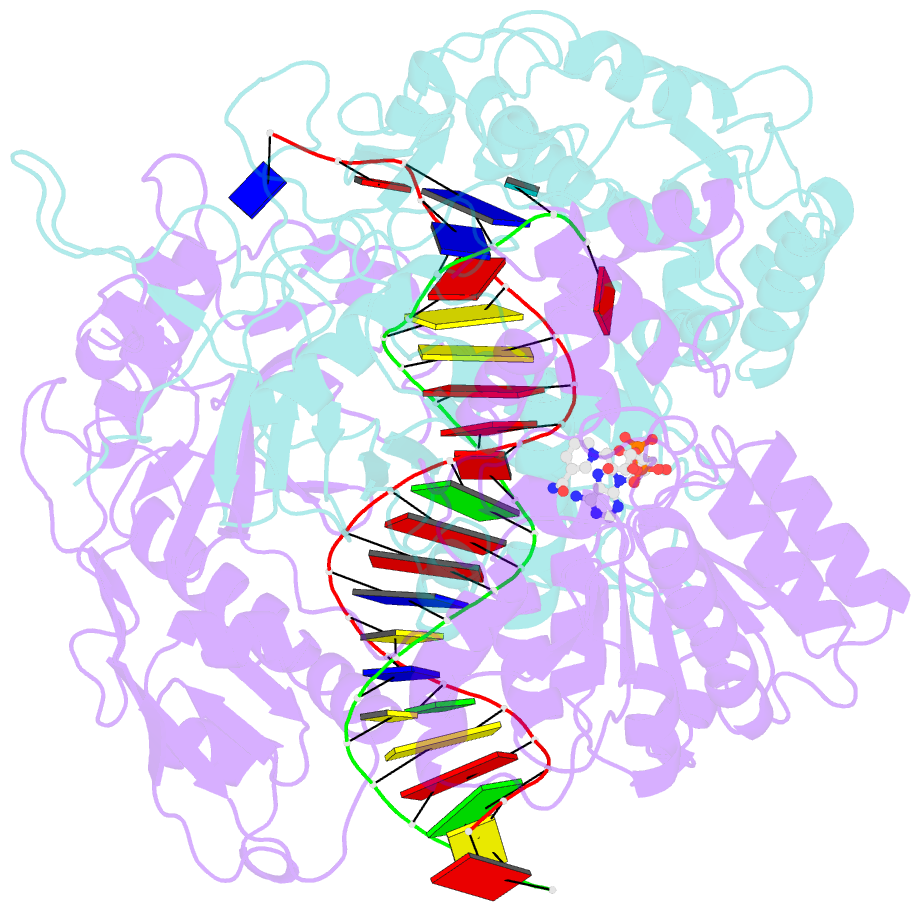
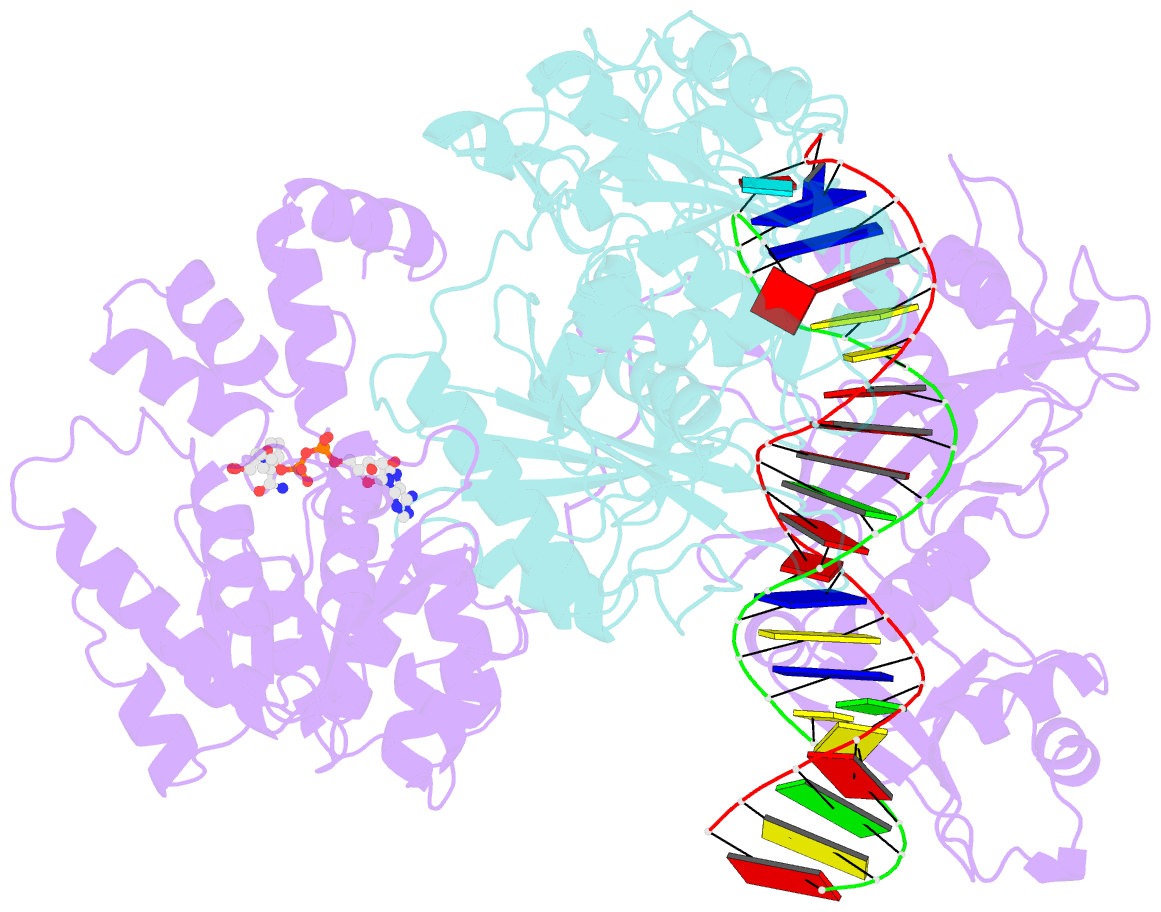
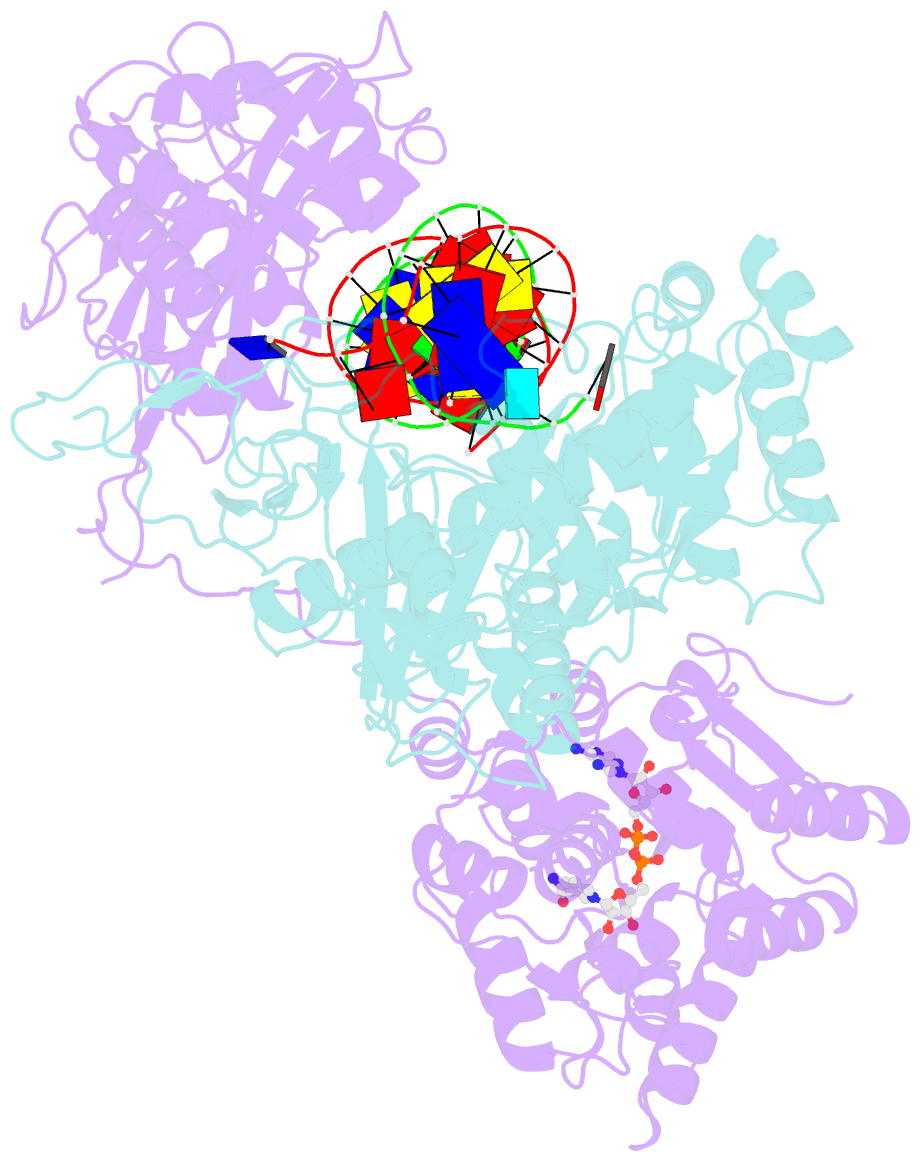
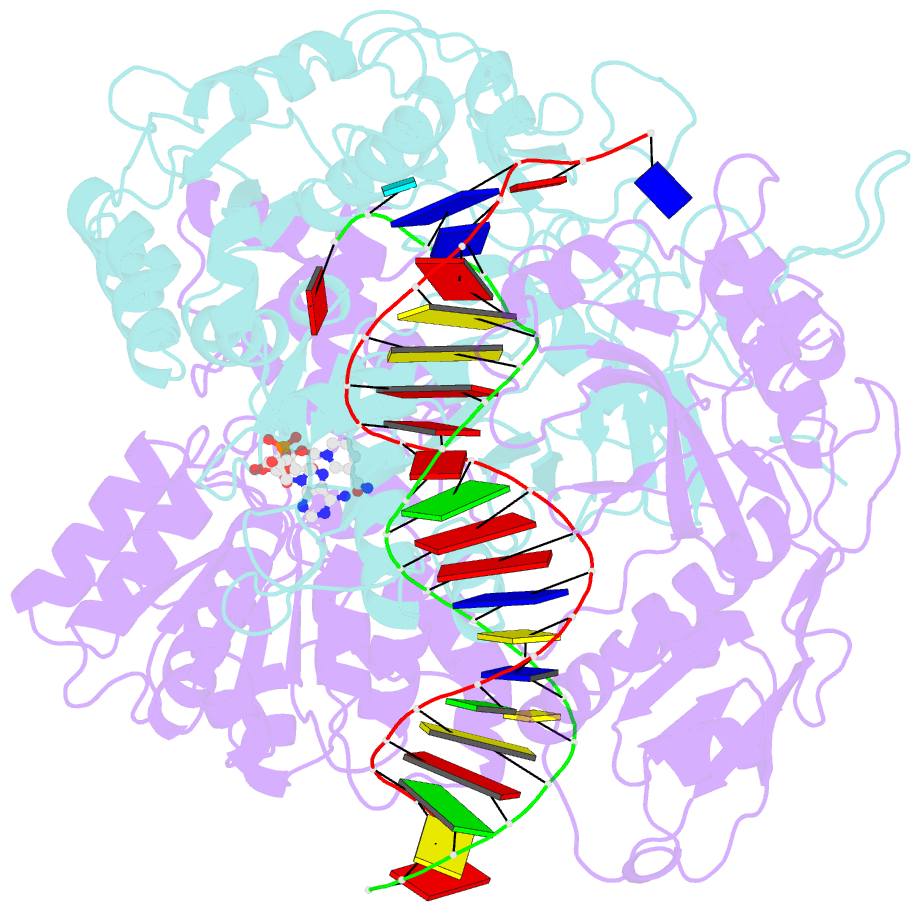
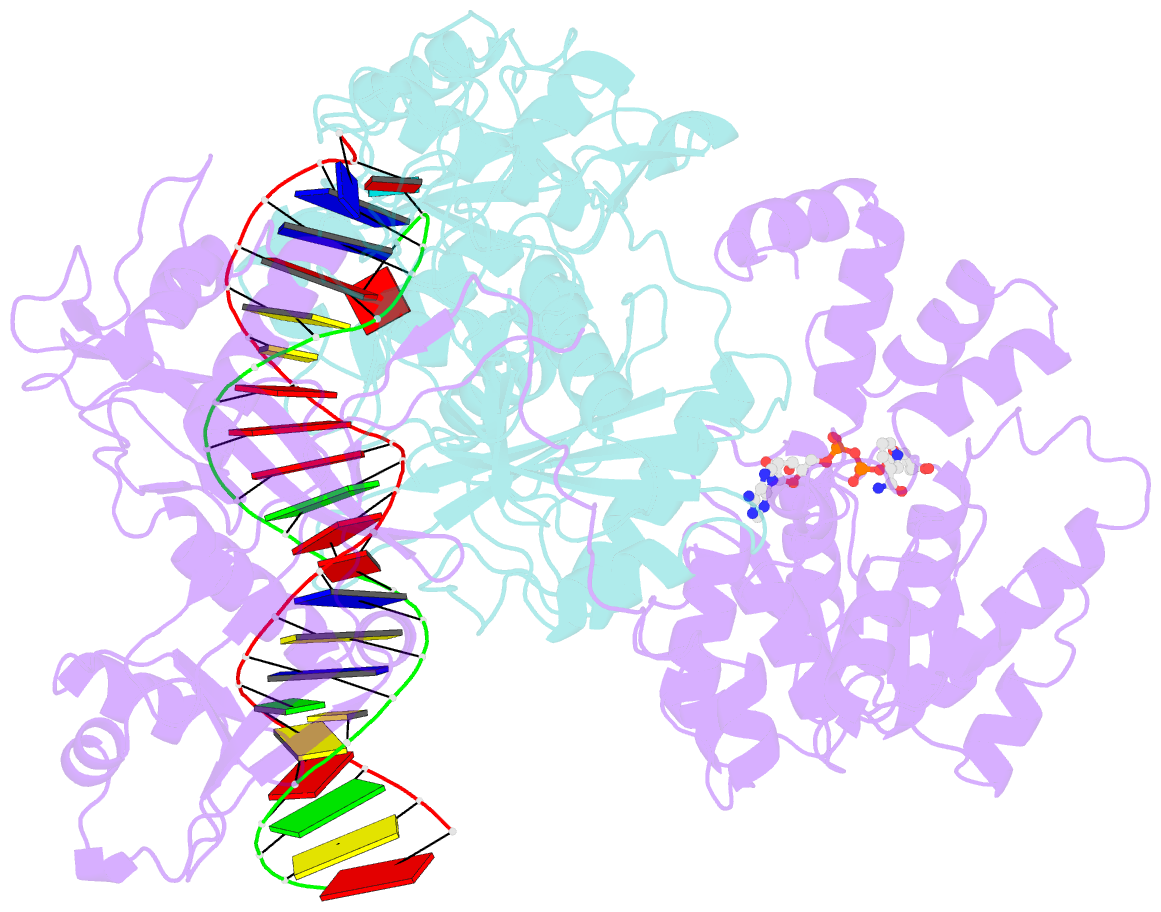
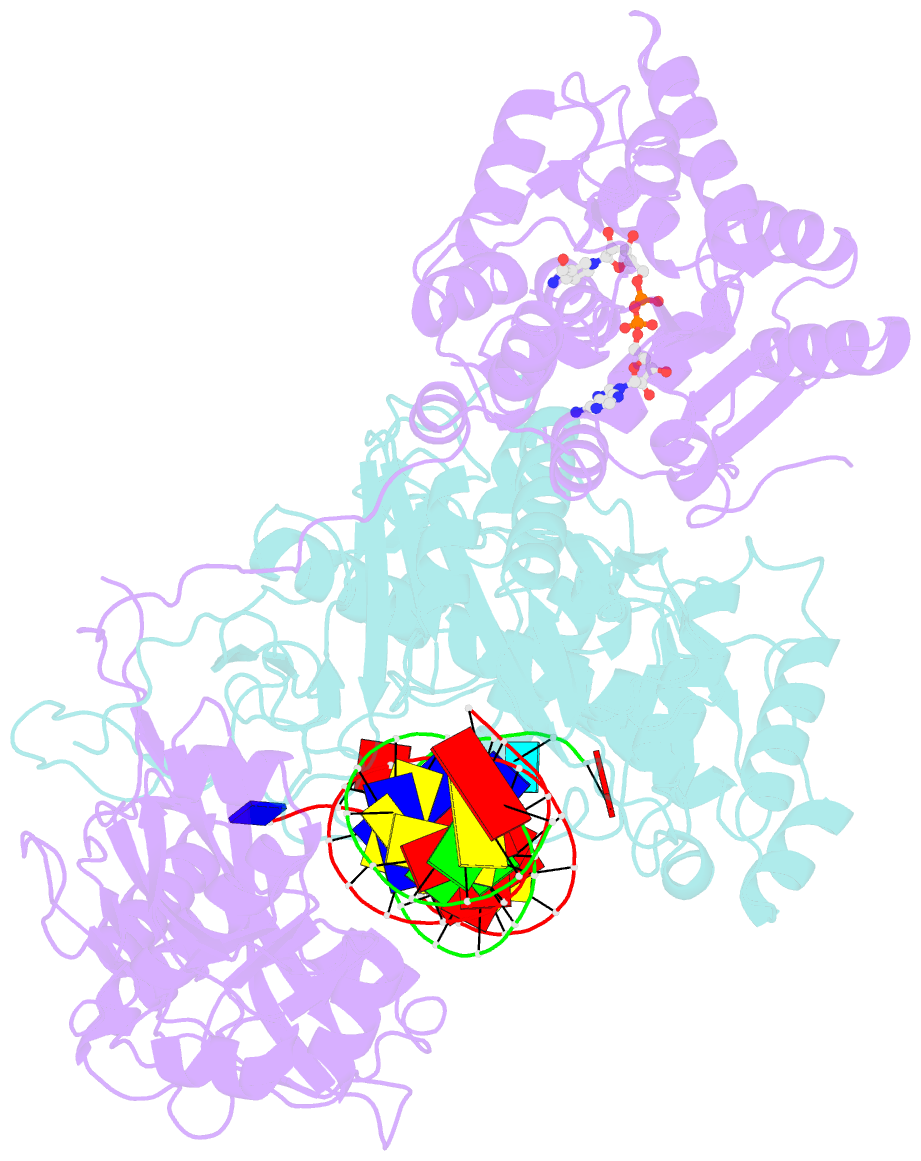
Summary information and primary citation
- PDB-id
- 8jl0; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- antiviral protein-DNA-RNA
- Method
- cryo-EM (3.1 Å)
- Summary
- cryo-EM structure of the prokaryotic sparsa system complex
- Reference
- Zhen X, Xu X, Ye L, Xie S, Huang Z, Yang S, Wang Y, Li J, Long F, Ouyang S (2024): "Structural basis of antiphage immunity generated by a prokaryotic Argonaute-associated SPARSA system." Nat Commun, 15, 450. doi: 10.1038/s41467-023-44660-7.
- Abstract
- Argonaute (Ago) proteins are ubiquitous across all kingdoms of life. Eukaryotic Agos (eAgos) use small RNAs to recognize transcripts for RNA silencing in eukaryotes. In contrast, the functions of prokaryotic counterparts (pAgo) are less well known. Recently, short pAgos in conjunction with the associated TIR or Sir2 (SPARTA or SPARSA) were found to serve as antiviral systems to combat phage infections. Herein, we present the cryo-EM structures of nicotinamide adenine dinucleotide (NAD+)-bound SPARSA with and without nucleic acids at resolutions of 3.1 Å and 3.6 Å, respectively. Our results reveal that the APAZ (Analogue of PAZ) domain and the short pAgo form a featured architecture similar to the long pAgo to accommodate nucleic acids. We further identified the key residues for NAD+ binding and elucidated the structural basis for guide RNA and target DNA recognition. Using structural comparisons, molecular dynamics simulations, and biochemical experiments, we proposed a putative mechanism for NAD+ hydrolysis in which an H186 loop mediates nucleophilic attack by catalytic water molecules. Overall, our study provides mechanistic insight into the antiphage role of the SPARSA system.