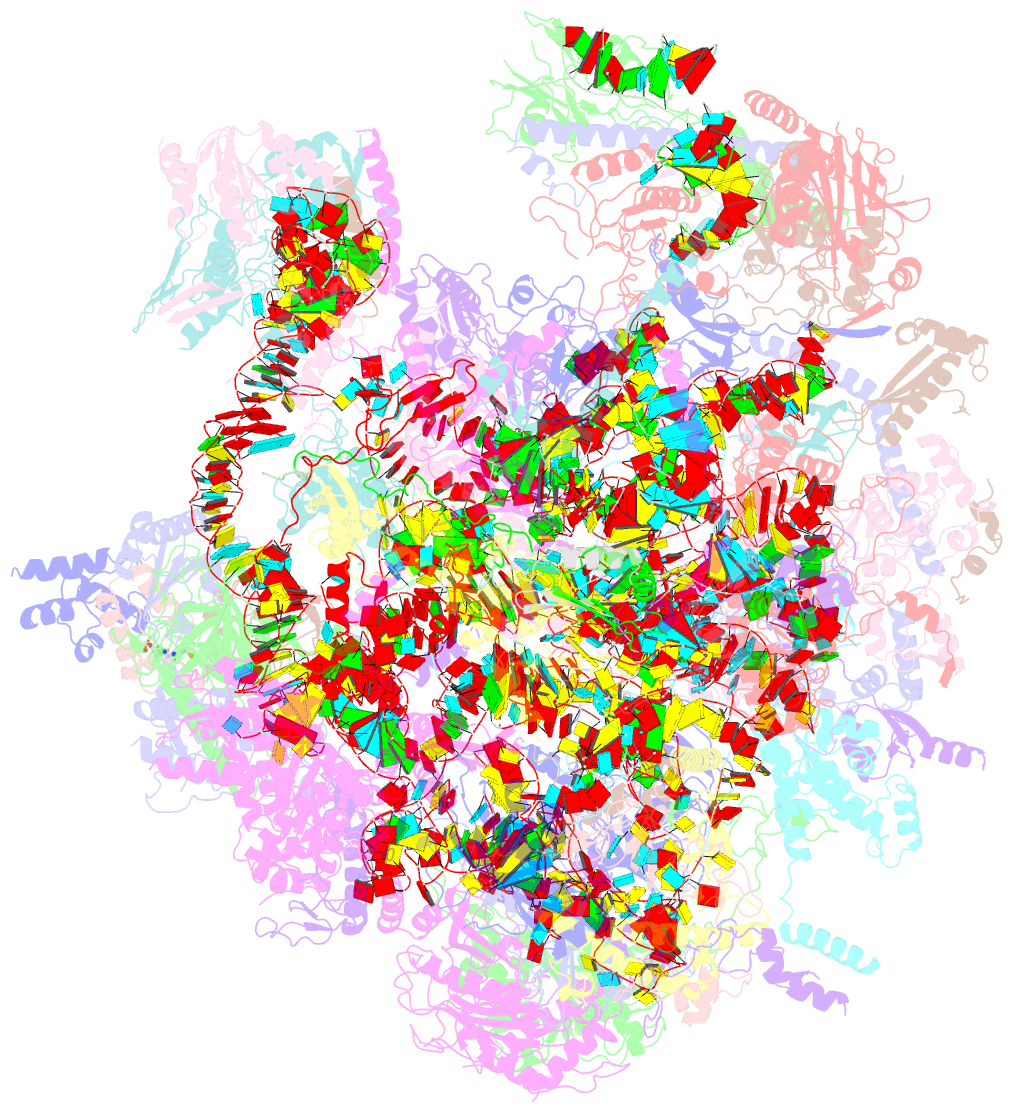
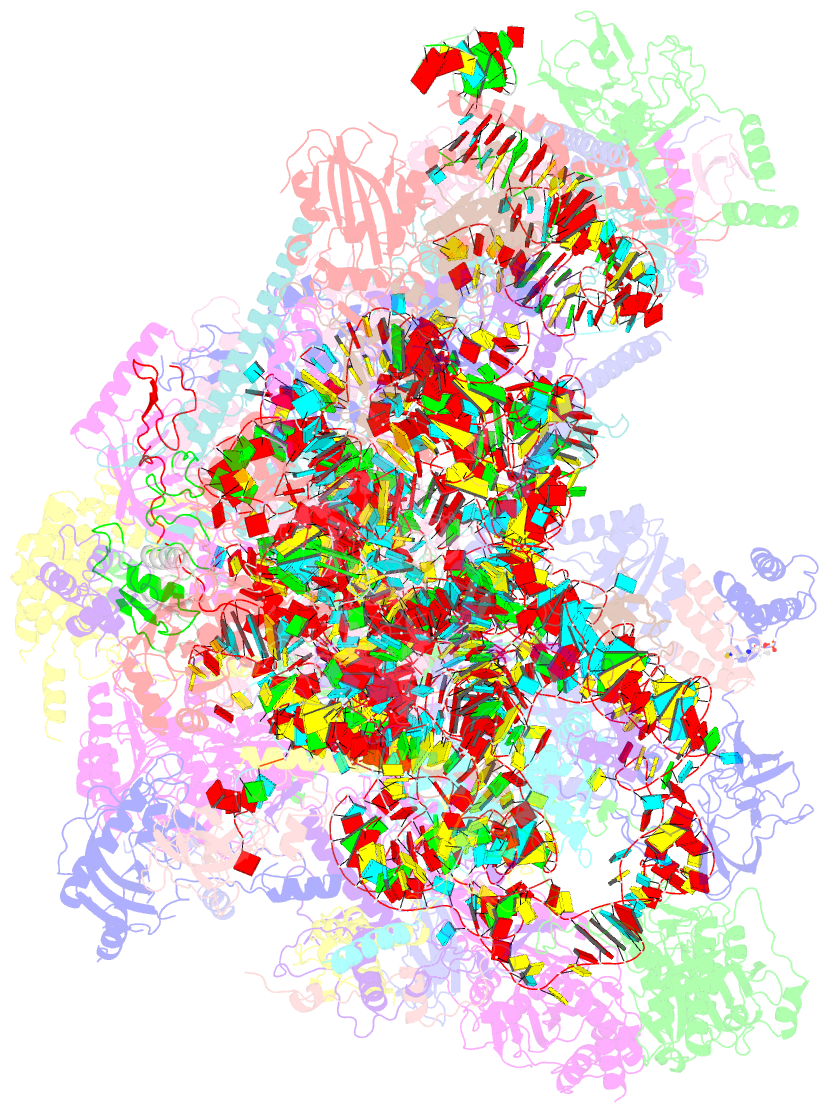
Summary information and primary citation
- PDB-id
- 8qu5; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- ribosome
- Method
- cryo-EM (2.42 Å)
- Summary
- Mt-lsu assembly intermediate in gtpbp8 knock-out cells, state 2
- Reference
- Cipullo M, Valentin Gese G, Gopalakrishna S, Krueger A, Lobo V, Pirozhkova MA, Marks J, Palenikova P, Shiriaev D, Liu Y, Misic J, Cai Y, Nguyen MD, Abdelbagi A, Li X, Minczuk M, Hafner M, Benhalevy D, Sarshad AA, Atanassov I, Hallberg BM, Rorbach J (2024): "GTPBP8 plays a role in mitoribosome formation in human mitochondria." Nat Commun, 15, 5664. doi: 10.1038/s41467-024-50011-x.
- Abstract
- Mitochondrial gene expression relies on mitoribosomes to translate mitochondrial mRNAs. The biogenesis of mitoribosomes is an intricate process involving multiple assembly factors. Among these factors, GTP-binding proteins (GTPBPs) play important roles. In bacterial systems, numerous GTPBPs are required for ribosome subunit maturation, with EngB being a GTPBP involved in the ribosomal large subunit assembly. In this study, we focus on exploring the function of GTPBP8, the human homolog of EngB. We find that ablation of GTPBP8 leads to the inhibition of mitochondrial translation, resulting in significant impairment of oxidative phosphorylation. Structural analysis of mitoribosomes from GTPBP8 knock-out cells shows the accumulation of mitoribosomal large subunit assembly intermediates that are incapable of forming functional monosomes. Furthermore, fPAR-CLIP analysis reveals that GTPBP8 is an RNA-binding protein that interacts specifically with the mitochondrial ribosome large subunit 16 S rRNA. Our study highlights the role of GTPBP8 as a component of the mitochondrial gene expression machinery involved in mitochondrial large subunit maturation.