Summary information and primary citation
- PDB-id
- 8uw3; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- RNA binding protein-RNA-DNA
- Method
- cryo-EM (3.2 Å)
- Summary
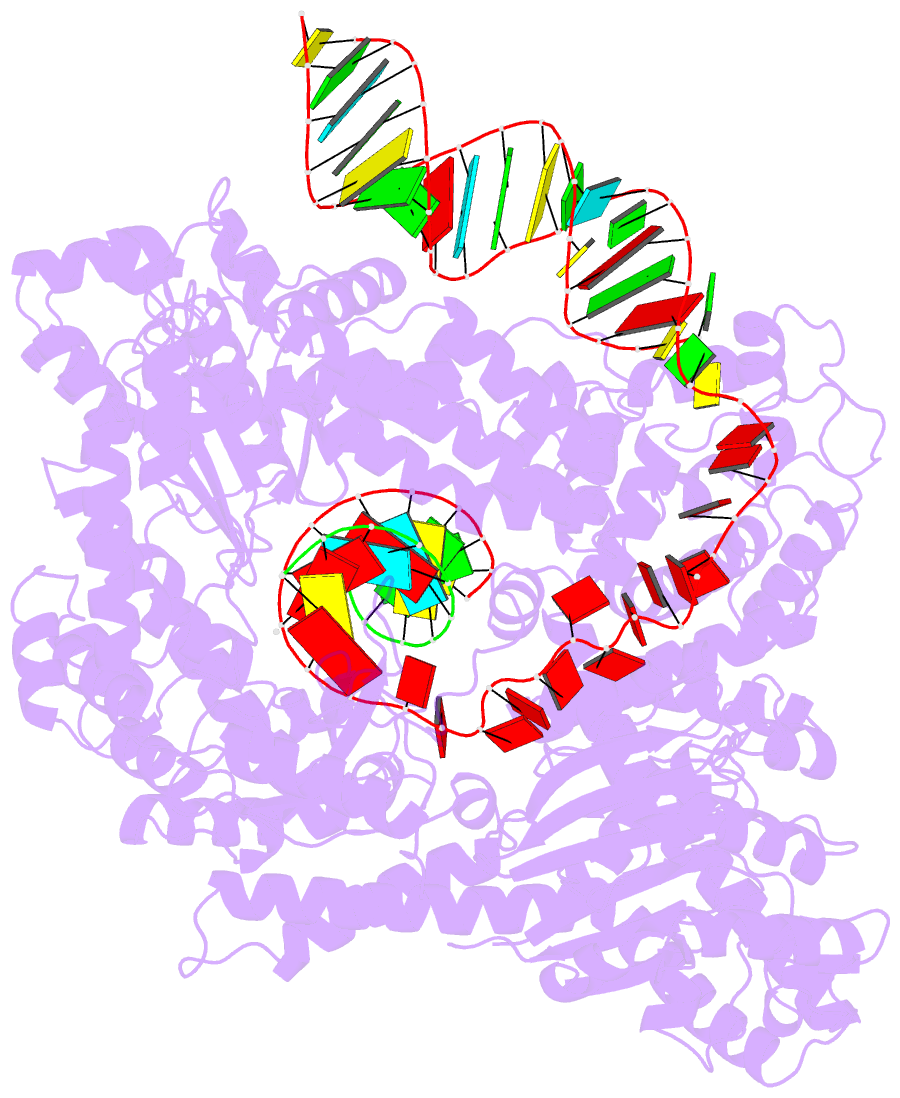
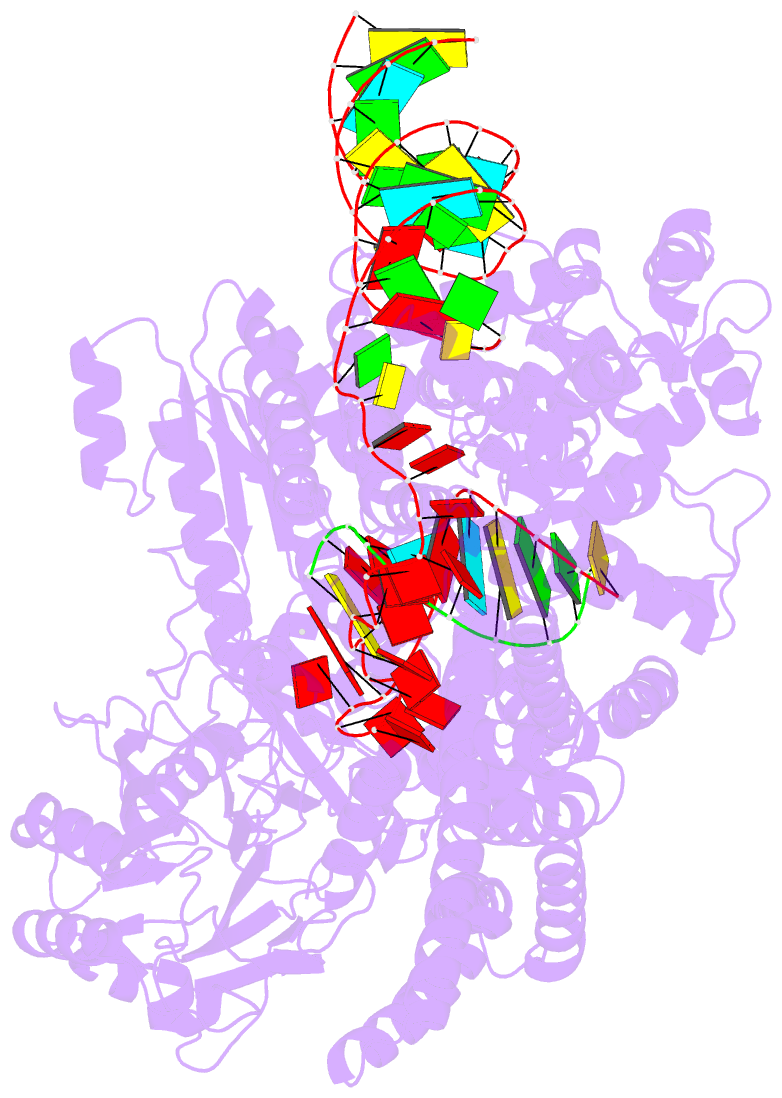
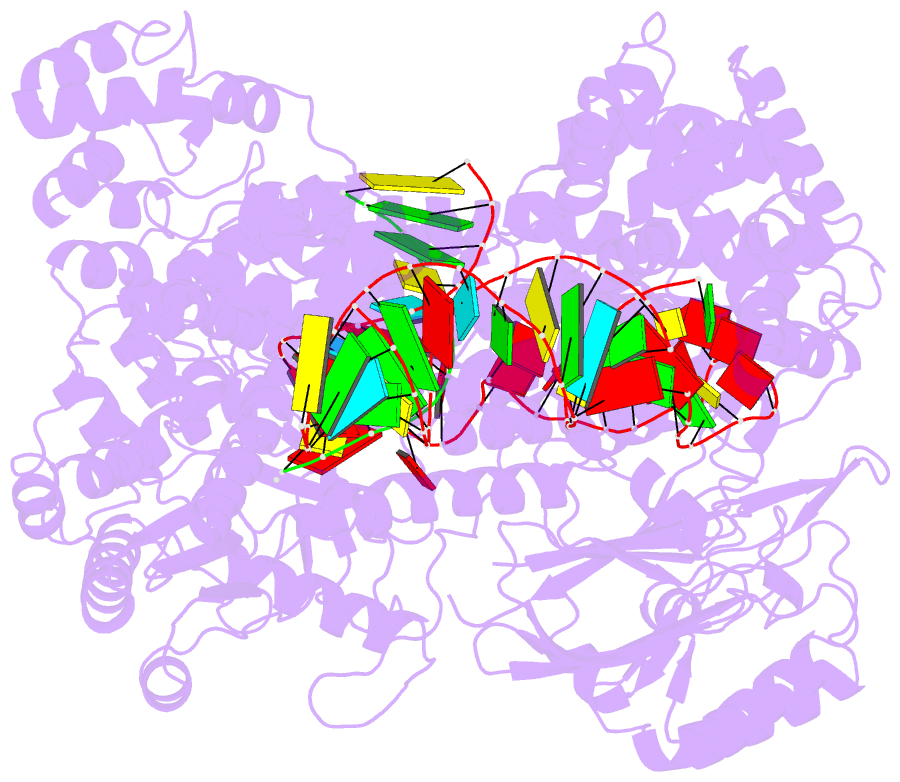
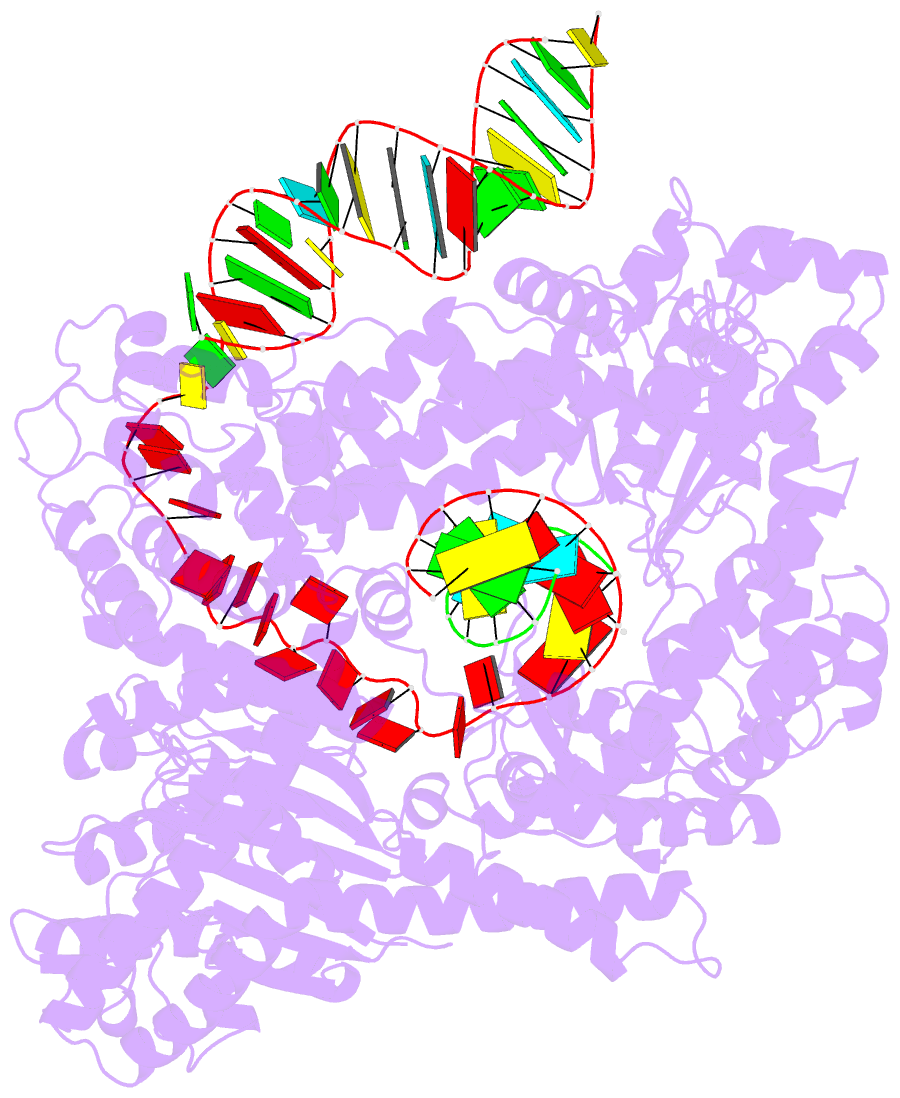
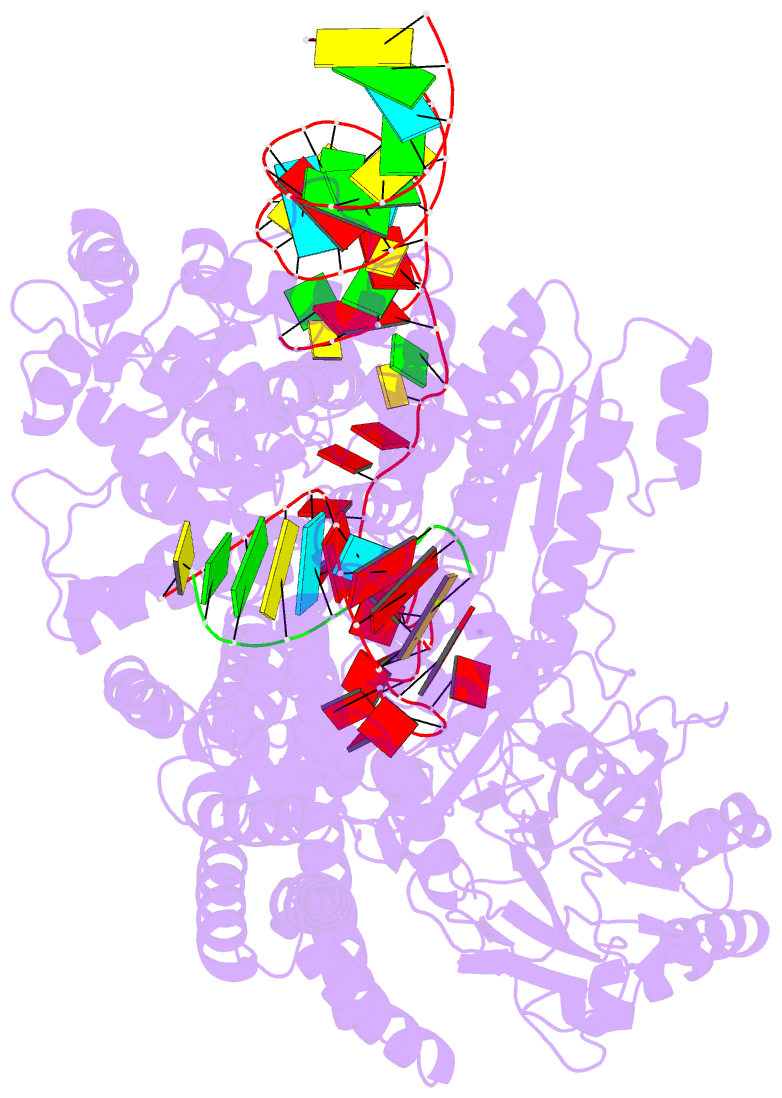
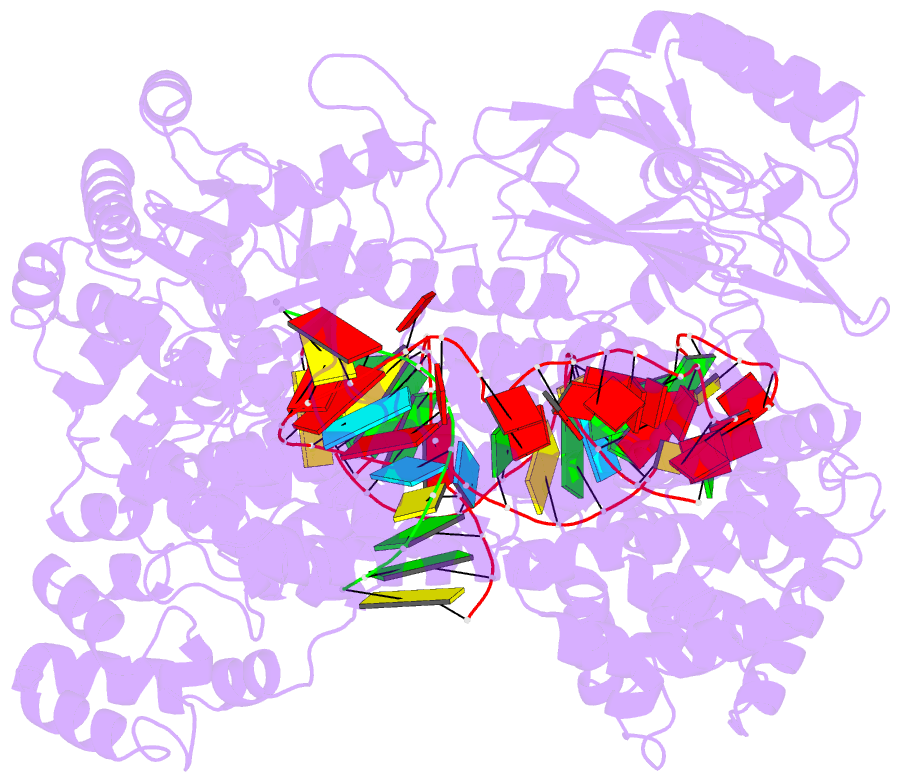
- Human line-1 retrotransposon orf2 protein engaged with template RNA in elongation state
- Reference
- Thawani A, Ariza AJF, Nogales E, Collins K (2024): "Template and target-site recognition by human LINE-1 in retrotransposition." Nature, 626, 186-193. doi: 10.1038/s41586-023-06933-5.
- Abstract
- The Long Interspersed Element-1 (L1) retrotransposon has generated nearly one-third of the human genome and serves as an active source of genetic diversity and human disease1. L1 spreads via a mechanism termed target-primed reverse transcription (TPRT), in which the encoded enzyme (ORF2p) nicks the target DNA to prime reverse transcription of its own or non-self RNAs2. Here, we purified the full-length L1 ORF2p and biochemically reconstituted robust TPRT with template RNA and target site DNA. We report cryo-electron microscopy structures of the human L1 ORF2p bound to structured template RNAs and initiating cDNA synthesis. The template polyadenosine tract is recognized in a sequence-specific manner by five distinct domains. Among them, a novel RNA-binding domain bends the template backbone to allow engagement of an RNA hairpin stem with the L1 ORF2p C-terminal segment. In addition, structure and biochemical reconstitutions demonstrate a surprising target-site requirement: L1 ORF2p relies on upstream single-stranded DNA to position adjacent duplex in the endonuclease active site for nicking of the longer DNA strand, with a single nick generating a staggered DNA break. Our work provides key insights into the mechanism of ongoing transposition in the human genome and informs the engineering of retrotransposon proteins for gene therapy.