Summary information and primary citation
- PDB-id
- 9c4g; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- antibiotic
- Method
- cryo-EM (2.53 Å)
- Summary
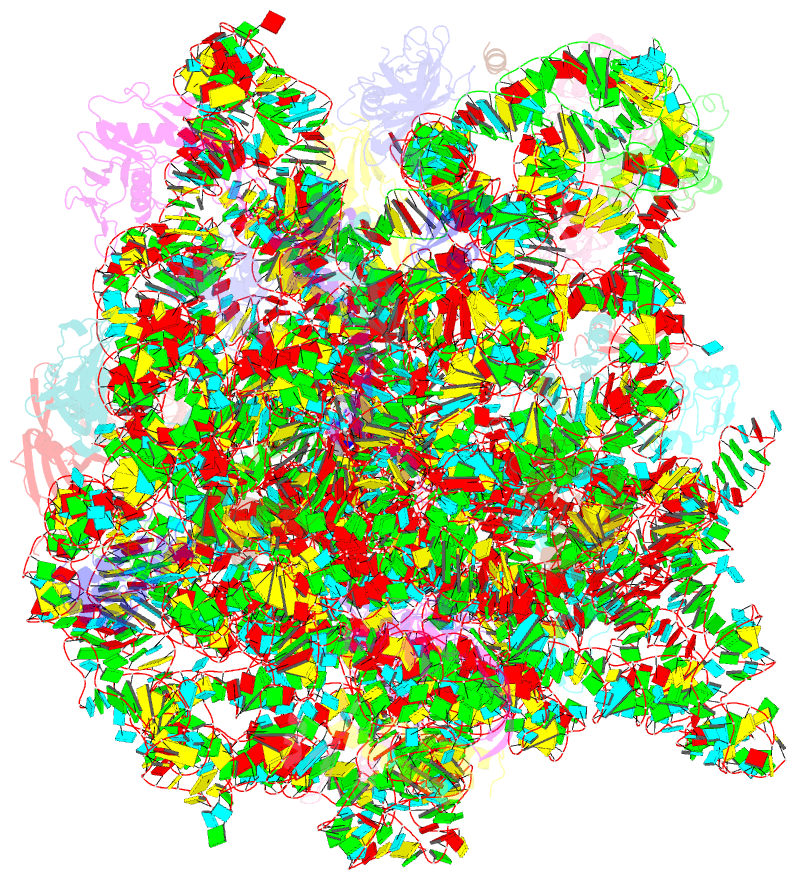
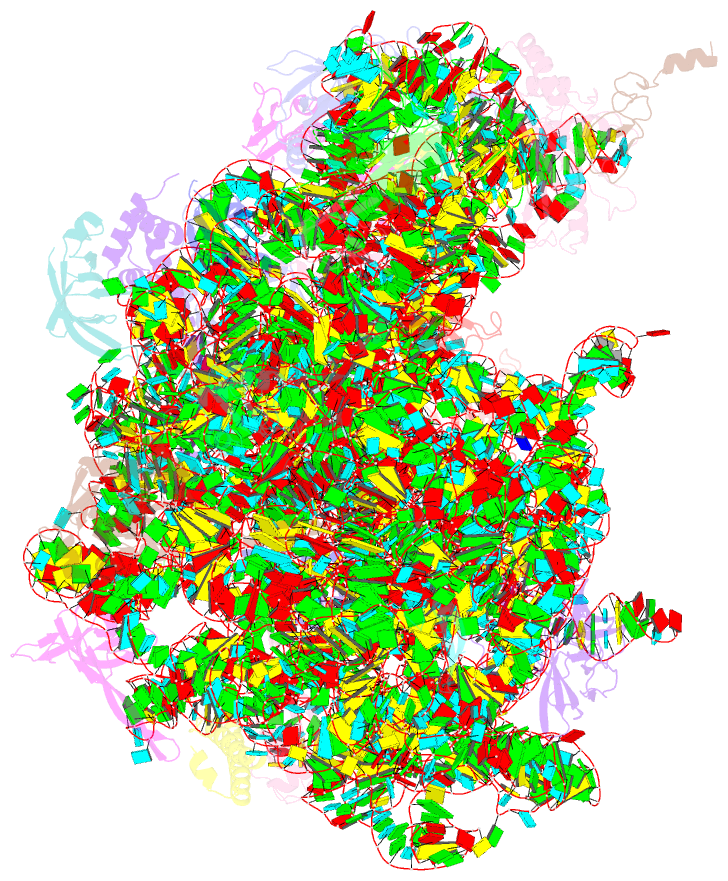
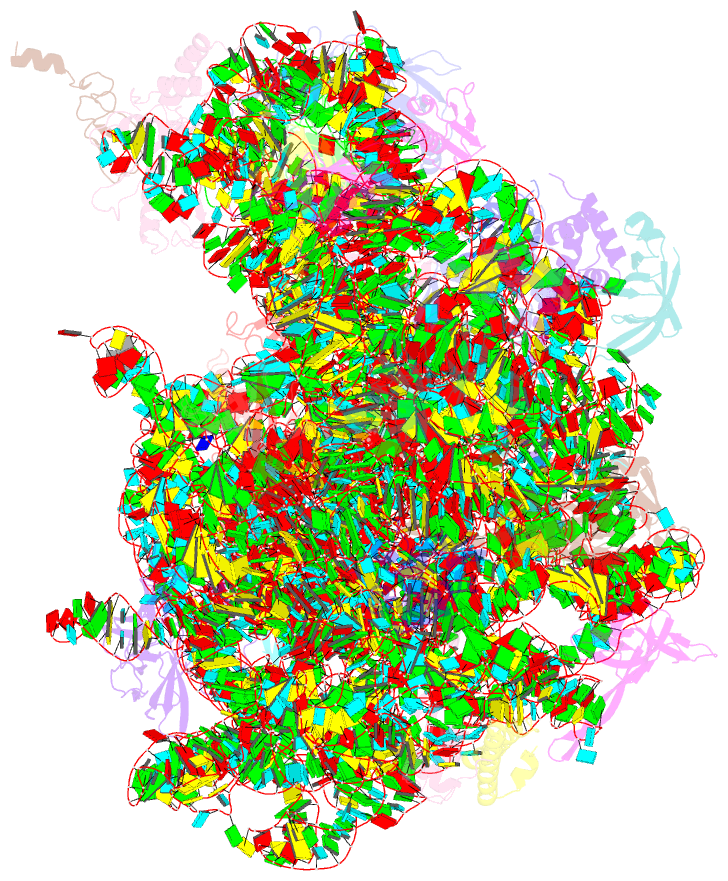
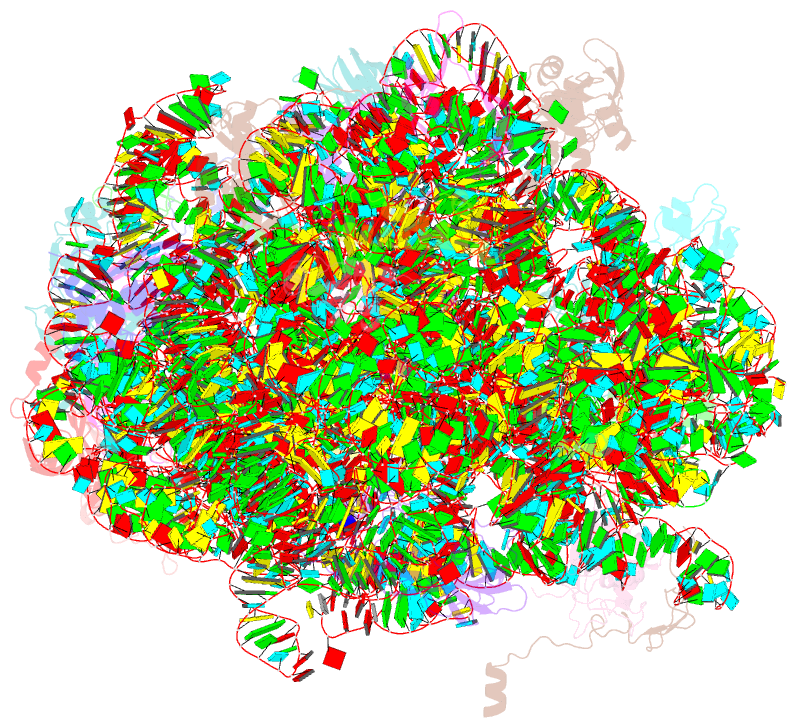
- Cutibacterium acnes 50s ribosomal subunit with clindamycin bound
- Reference
- Lomakin IB, Devarkar SC, Grada A, Bunick CG (2024): "Mechanistic Basis for the Translation Inhibition of Cutibacterium acnes by Clindamycin." J Invest Dermatol., 144, 2553-2561.e3. doi: 10.1016/j.jid.2024.07.013.
- Abstract
- Inflammation and the Gram-positive anaerobic bacterium Cutibacterium acnes, which is implicated in acne pathogenesis and pilosebaceous unit inflammation, are the main targets of antibiotic-based therapy against acne vulgaris (acne). The most widely used antibiotics in acne therapy are tetracyclines, macrolides, and lincosamides. Unfortunately, C. acnes bacteria over the past several decades have demonstrated increased resistance to these antibiotics, particularly to clindamycin. The precise knowledge of how antibiotics interact with their clinical target is needed to overcome this problem. Toward this goal, we determined the structure of clindamycin in complex with the ribosome of Cutibacterium acnes at 2.53 Å resolution using cryogenic electron microscopy. The galactose sugar moiety of clindamycin interacts with nucleotides of the 23S rRNA directly or through a conserved network of water-mediated interactions. Its propyl pyrrolidinyl group interacts with the 23S rRNA through van der Waals forces. Clindamycin binding to the Cutibacterium acnes ribosome interferes with both: proper orientation of the aminoacyl group of the A-site bound tRNA that is needed for peptide bond formation and with the extension of the nascent peptide. Our data are important for advancing understanding of antibiotic resistance and development of narrow- spectrum antibacterial drugs, which is an urgent need for contemporary antibiotic stewardship.