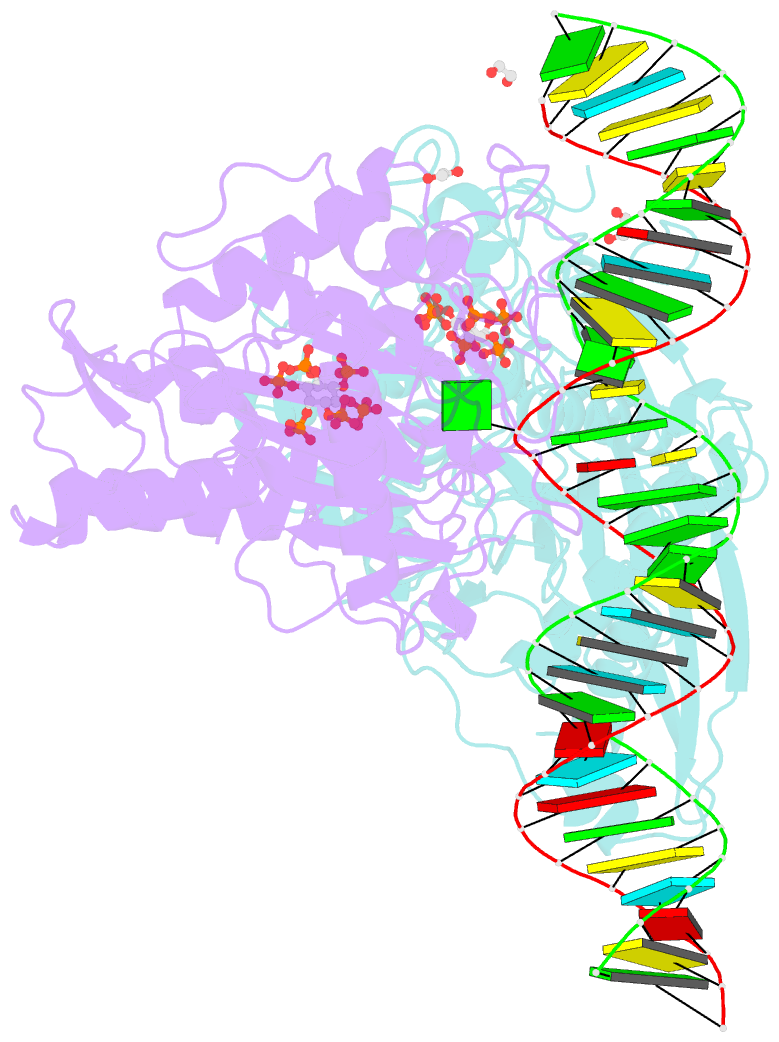
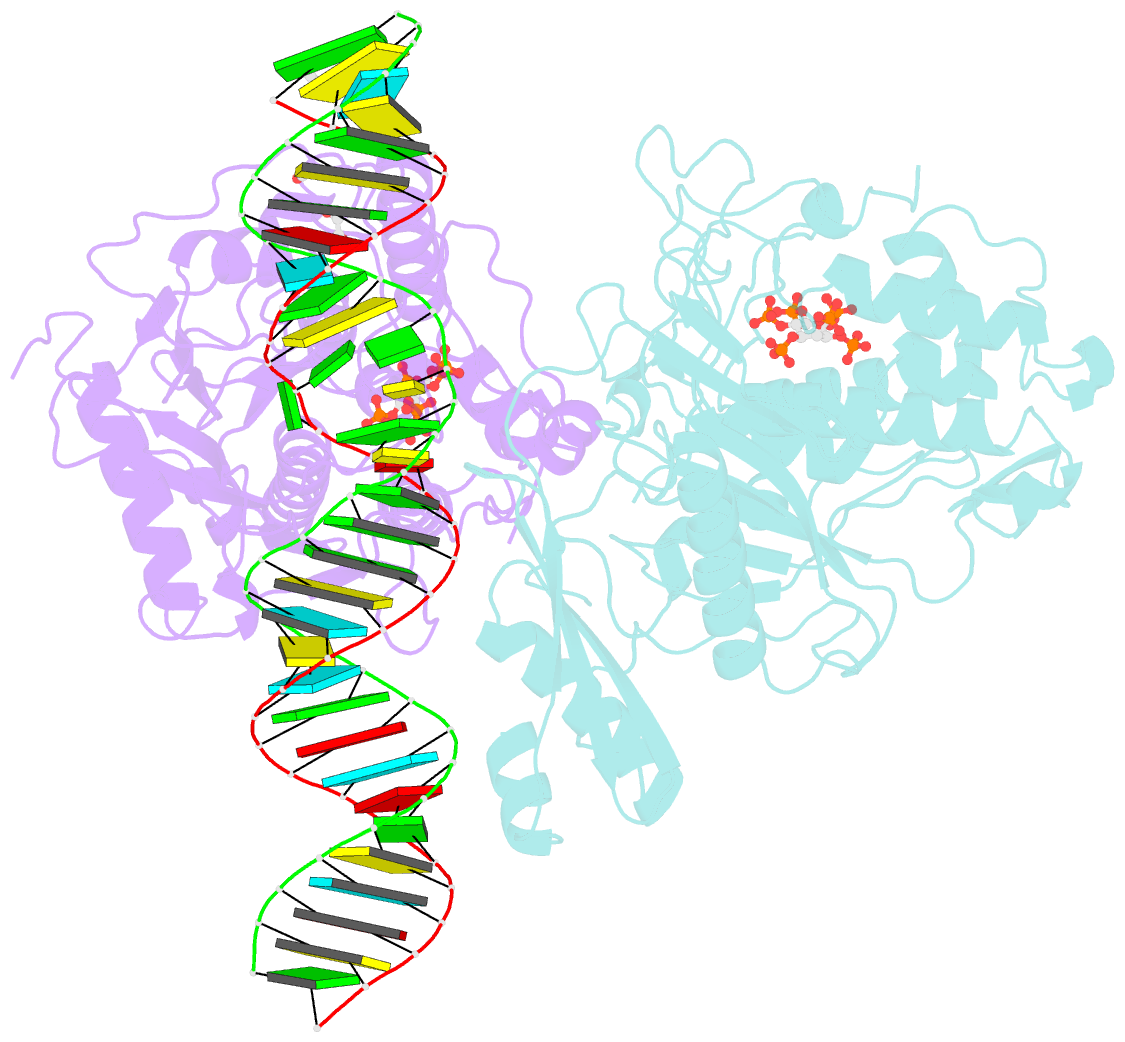
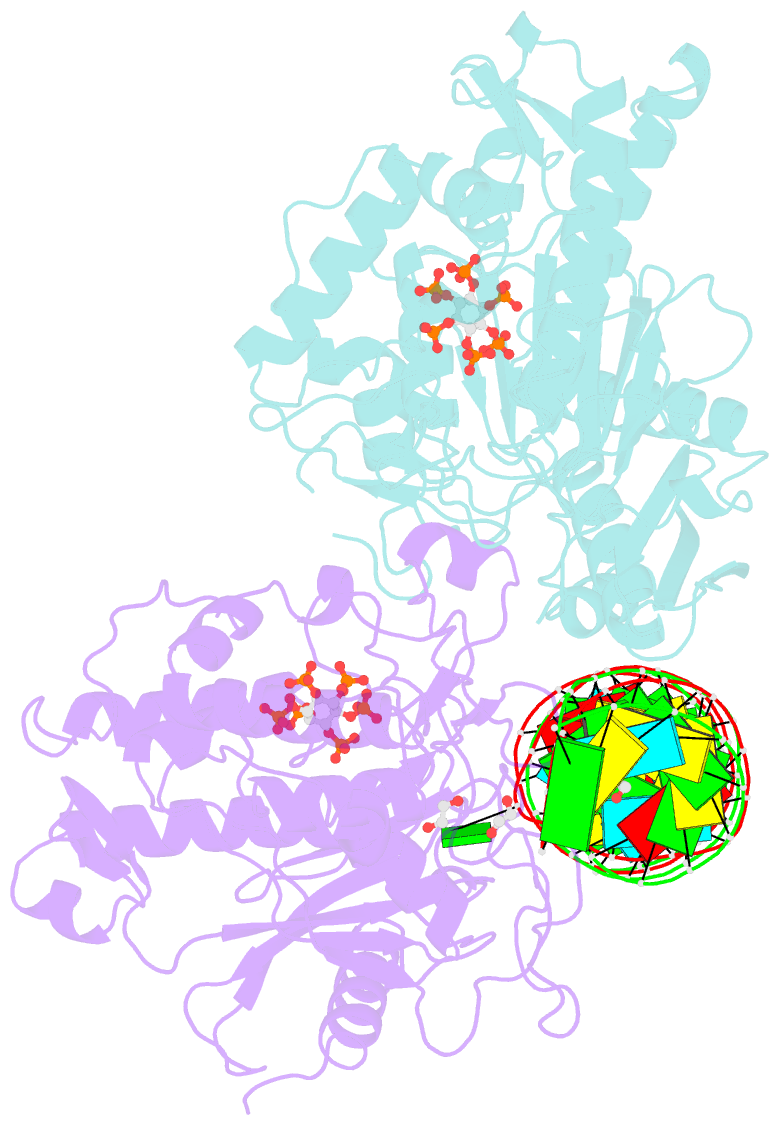
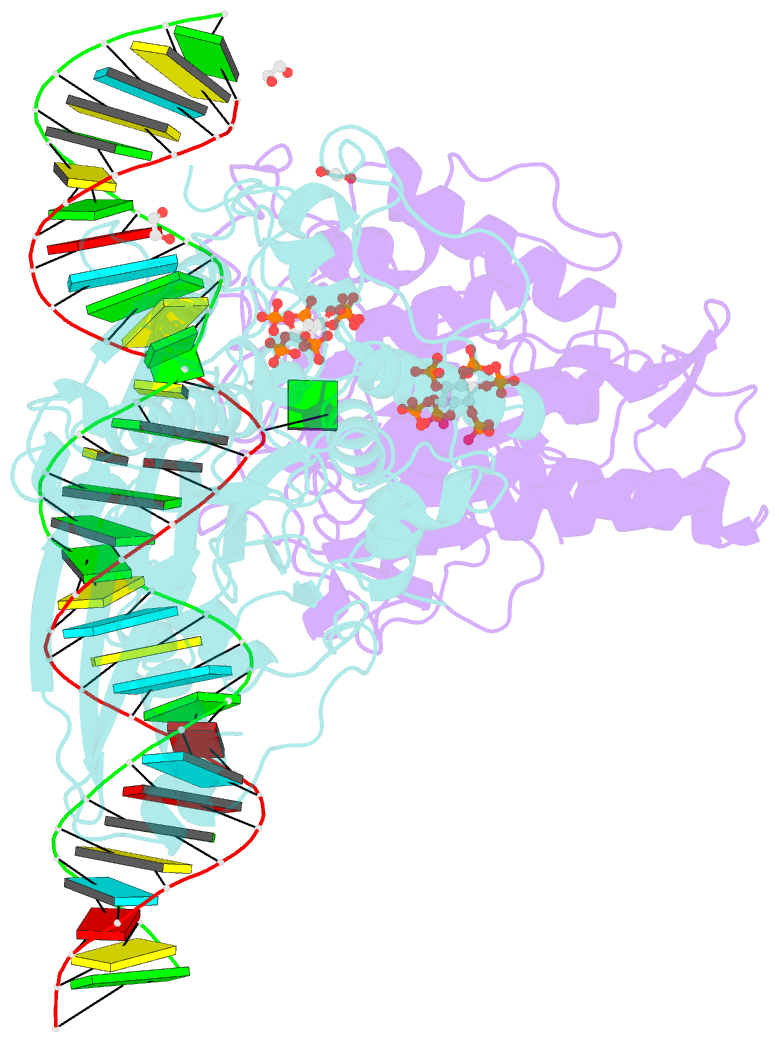
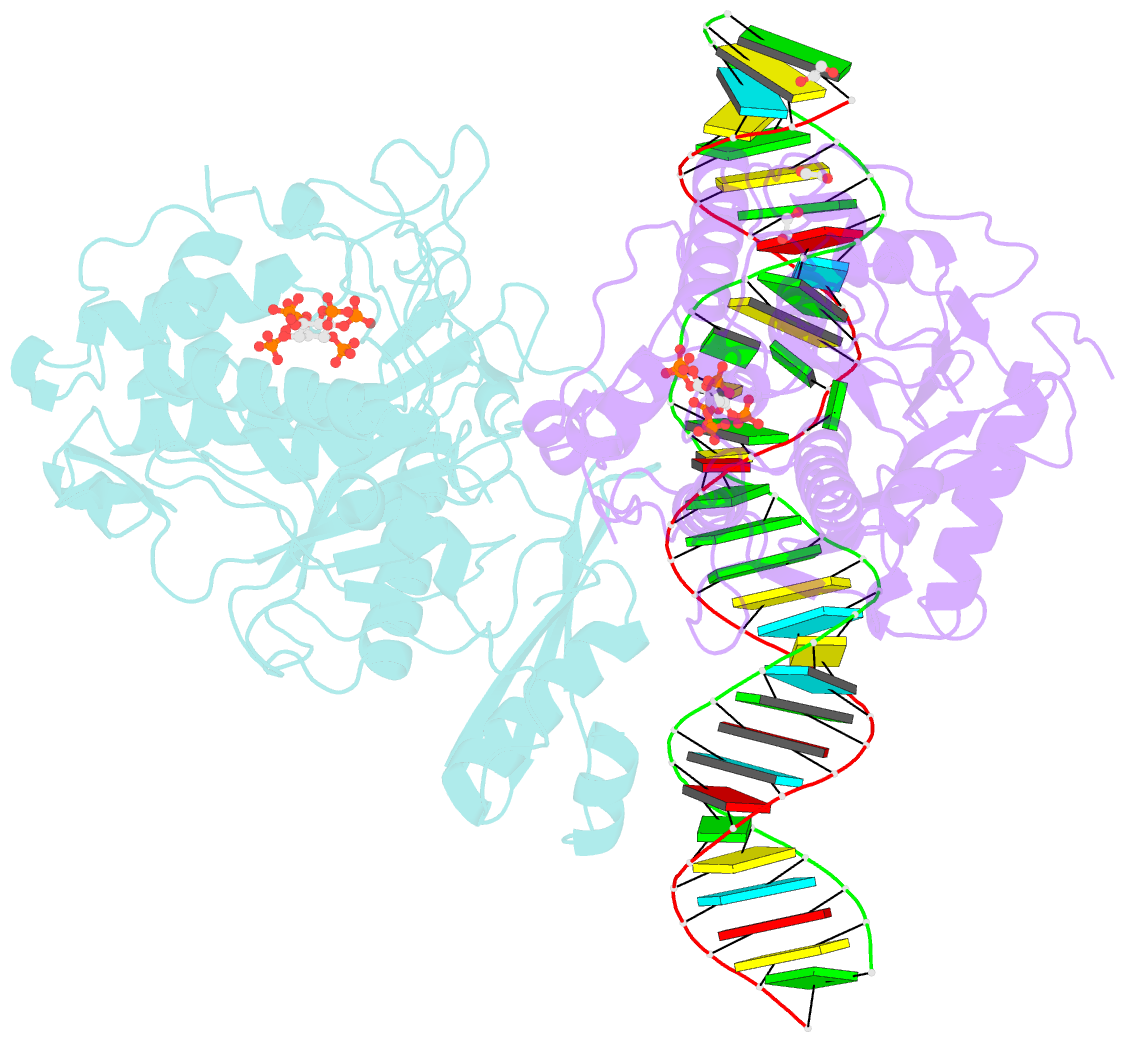
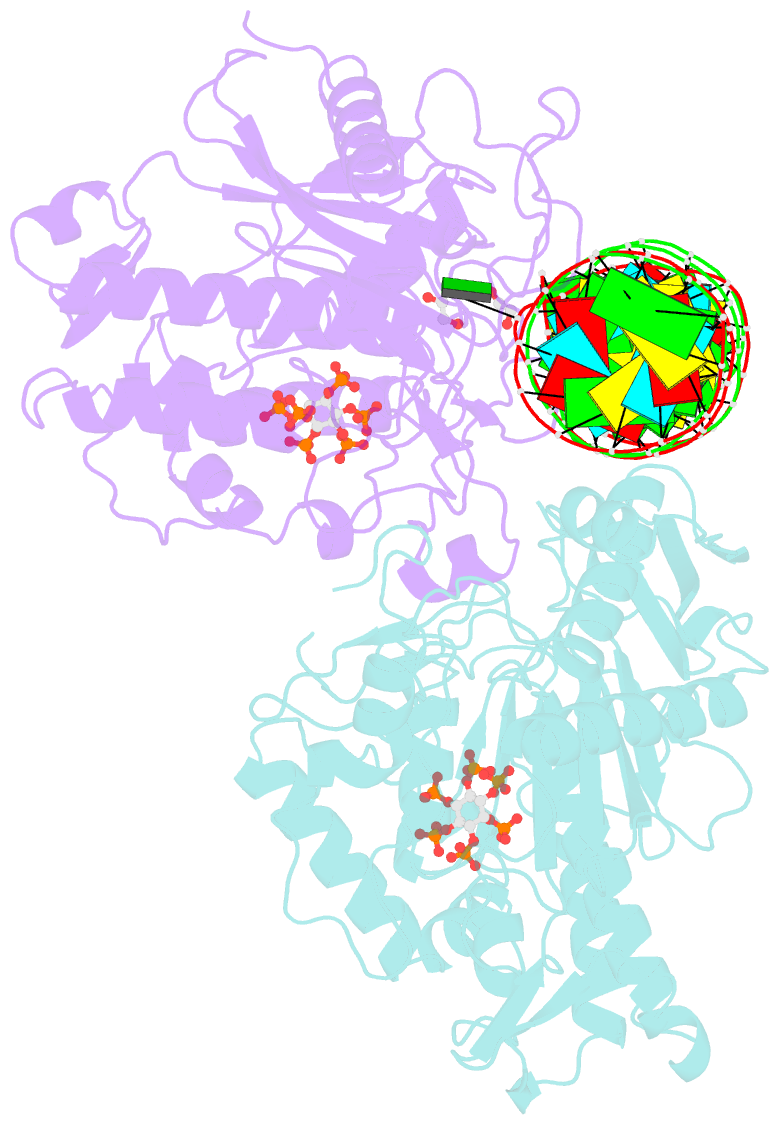
Summary information and primary citation
- PDB-id
- 9d5j; SNAP-derived features in text and JSON formats;
DNAproDB
- Class
- RNA binding protein-RNA
- Method
- X-ray (2.8 Å)
- Summary
- Human adenosine deaminase acting on dsrna (adar2-rd) bound to dsrna containing deoxyinosine at the -1 position of the guide strand
- Reference
- Manjunath A, Cheng J, Campbell KB, Jacobsen CS, Mendoza HG, Bierbaum L, Jauregui-Matos V, Doherty EE, Fisher AJ, Beal PA (2024): "Nucleoside Analogs in ADAR Guide Strands Enable Editing at 5'-G A Sites." Biomolecules, 14. doi: 10.3390/biom14101229.
- Abstract
- Adenosine Deaminases Acting on RNA (ADARs) are members of a family of RNA editing enzymes that catalyze the conversion of adenosine into inosine in double-stranded RNA (dsRNA). ADARs' selective activity on dsRNA presents the ability to correct mutations at the transcriptome level using guiding oligonucleotides. However, this approach is limited by ADARs' preference for specific sequence contexts to achieve efficient editing. Substrates with a guanosine adjacent to the target adenosine in the 5' direction (5'-GA) are edited less efficiently compared to substrates with any other canonical nucleotides at this position. Previous studies showed that a G/purine mismatch at this position results in more efficient editing than a canonical G/C pair. Herein, we investigate a series of modified oligonucleotides containing purine or size-expanded nucleoside analogs on guide strands opposite the 5'-G (-1 position). The results demonstrate that modified adenosine and inosine analogs enhance editing at 5'-GA sites. Additionally, the inclusion of a size-expanded cytidine analog at this position improves editing over a control guide bearing cytidine. High-resolution crystal structures of ADAR:/RNA substrate complexes reveal the manner by which both inosine and size-expanded cytidine are capable of activating editing at 5'-GA sites. Further modification of these altered guide sequences for metabolic stability in human cells demonstrates that the incorporation of specific purine analogs at the -1 position significantly improves editing at 5'-GA sites.